

Greener Journal of Agricultural Sciences
ISSN: 2276-7770; ICV: 6.15
Research Article
Production of Soy yoghurt from Lactobacillus isolated from fermented African Oil Bean Seed (Ugba)
C. C. Opara, T. Kuru and I. B. Ezenwaka
Department of Chemical Engineering, University of Port Harcourt
|
ARTICLE INFO |
ABSTRACT |
|
|
Article No.: 112412293 DOI: 10.15580/GJAS.2013.2.112412293 |
An important process for stabilizing high protein agricultural products is through fermentation which uses specific microorganisms. Costs involved in the supply of such organisms can be a major component of the cost of the fermented product. Soy milk is currently produced in abundance in Nigeria and part of it is fermented into yoghurt with imported micro-organisms which makes the product expensive. In this project, a microbial consortium isolated from fermented African oil bean seed was used as substitute to the imported starter culture to ferment soy milk. Lactobacillus spp. was isolated and cultured after different tests were carried out and inoculated into the soy milk to form soy yoghurt. Yoghurt made from milk sterilized at 90oC had desirable colour, taste and titrable acidity, but very soft texture. A satisfactory product was obtained at 100OC. The study further showed that soy yoghurt can be kept at 5OC for about 90 days without any significant change in acidity, pH, and viable cell count. The most acceptable titrable acidity was 0.8% of lactic acid. When the acidity was higher than 0.9%, the yoghurt had a harsh flavour.
|
|
|
Submitted: 24/11/2012 Accepted: 20/01/2013 Published: 20/02/2013 |
||
|
*Corresponding Author Kuru T. E-mail: tukakuru@gmail.com
|
||
|
Keywords: Soy yoghurt, African oil bean seed, Soy milk, Lactobacillus
|
||
|
|
|
INTRODUCTION
Fermentation is one of the oldest and most economical methods of producing and preserving foods in developing countries (David and Aderibigbe, 2010). According to Campbell-Platt (1987), fermented foods are those foods which have been subjected to the action of micro-organisms so that desirable biochemical changes cause significant modification to the food. As one of man’s earliest form of food preservation technology, fermentation enabled the move from perishable crops to preserved products (Zilberman and Eunice, 2011). Marshall et al. (2011) reveals that extending the shelf-life of foods is one of the major objectives of fermentation, with aspects such as wholesomeness, acceptability and overall quality.
A typical product of fermentation is yoghurt; consumed by millions of people worldwide. Yogurt is originally a Turkish word referring to a slightly acid milk food fermented with a mixed culture of lactic acid bacteria (Shurtleff and Aoyagi, 2007). The bacteria used is often obtained from various sources and cultured.
African Oil bean seeds (Pentaclethra macrophylla) called Ugba in the Igbo language of Eastern Nigeria belongs to leguminous family mimosa cease. It is frequently cultivated in forest areas (Nwamarah and Madueke, 2010). The raw seed is a potential source of edible protein, energy and fatty acids (Enujiugha and Agbede, 2000; Enujiugha, 2003). The fermented African oil bean seed product is traditionally prepared in various steps for a period of three days (Enujiugha and Agbede, 2000; Enujiugha, 2003). After 3-day fermentation, it is known as ‘ugba’ which will now be ready to be consumed as snack or used as condiment in soup mixes, local salad and porridges. Previous works reported that fermentation improved the nutritional quality as compared with the unfermented seeds (Achinewhu, 1986).
Isu and Njoku (1997) studied micro-organisms associated with fermented African oil bean seed during ugba production. Only bacteria were isolated from the ugba samples used. Although the bacteria included Bacillus spp., Lactobacillus spp., Staphylococcus spp., Micrococcus spp. and members of the family Enterobacteriaceae, only the Bacillus spp. were found to ferment African oil bean seeds to ugba. It was also found that Bacillus spp. were the predominant micro-organisms present, constituting over 95% of the total microbial population density.
The fermentation process for the production of soy yoghurt from soy milk involves the inoculation of Lactobacillus, isolated from fermented Ugba, to soy milk. A microbial metabolic process occurs by the partial oxidation of carbohydrates (starchyose and raffinose) resulting in a desired form of food drink that has high commercial value.
Soy yoghurt is particularly suitable for vegens, ovo-vegetarians, people with phenylketonoria (PKU) and those with lactose intolerance or milk allergy (Wikipedia, 2012).
The digestibility of soy yoghurt is also greater due to its appetizing flavour and aroma as well as textural characteristics.
This report aims to produce soy yoghurt by fermentation of soy milk using Lactobacillus spp. isolated from fermented ugba.
The shelf-life qualities affected by protein content, sensory tests, proximate compositions, characteristics of soybean yoghurt and soybean milk subjected to varied heat treatment is also done.
Although the use of bacteria is essential, not all bacterium is used for the process thus more advanced production equipment is needed to achieve an optimal production process of the healthy non- dairy yoghurt.
MATERIALS AND METHODS
Materials
Ugba, 3ml nutrient broth tubes, 0.1% Peptone Water ( Product Number: 299111, BD), Bunsen burner (TD - 1240.2, TD MODELS AND SCIENTIFIC), 0.9% Normal saline (DANA) , Pasteur’s pipette, Incubator, slide, heater (Model 440482, BD), Gram’s iodine, Water (tap, distilled and ionized), ethanol, sugar (lactose), conical flask, Nutrient agar plates per group, pendulum scale, petroleum jelly, microscope(Model A1-10 , TD MODELS AND SCIENTIFIC) , pack of colour pencils, petri-dishes, MacConkey broth (Alpha Biosciences, Inc), Durham tubes, Eosin methylene blue agar (PA-54014.05, BD), inoculating wire loop, crystal violet stain, Simmon’s citrate agar (ISO 10273 CONDA), 5% naphthol (261192, BD), potassium hydroxide, gelatine agar, hot air oven (ACM-22066-I ACMAS), blender (Model STZ-04, TECHLAB), filter cloth, boiler, stop watch (FP601, UNIDEX), pendulum scale, sodium bicarbonate, potash, Sonifer J32A of Branson Instruments Inc., Soymilk, lactobacillus culture, water bath, thermometer (model 4400, ALPHA TECHNICS), pH meter (model 291 MKS, PYE UNICON), measuring cylinder.
Method
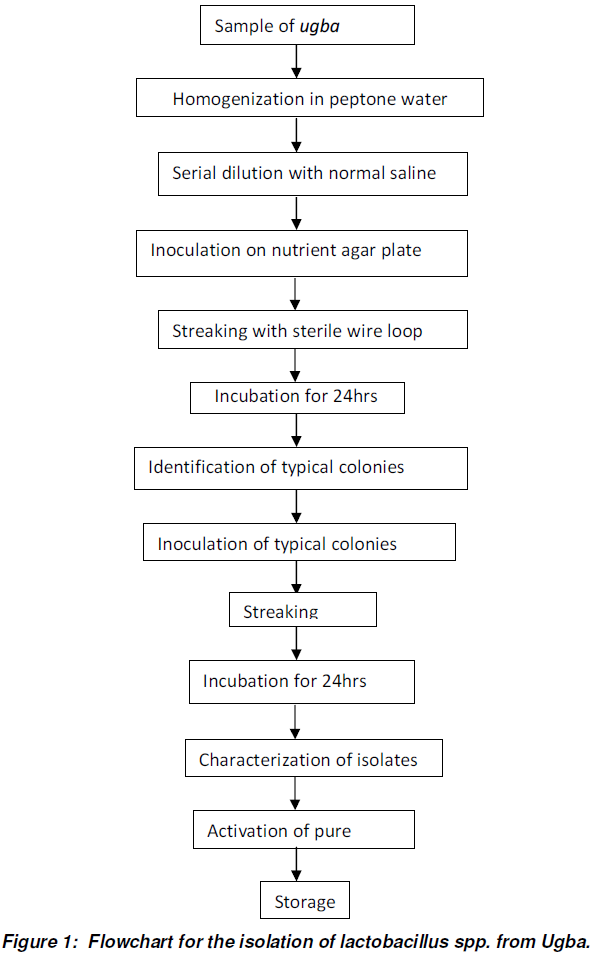
Isolation of Microbial Culture Consortium
Isolation of microbial colonies was done using the plate streak method as described in Thiery and Frachon (1997).
Gram staining
The isolates were stained using the procedure described by Prescott et al. (1996).
Characterization of Isolates
The characterization test for isolates was done following the procedure as outlined by Collins and Lyne (1976), and Skerman (1967). The isolates were identified according to the identification test procedure reported in Bergey’s manual of determinative Bacteriology (Buchanan and Gibbons, 1974). The characterization included morphological and biochemical characterization.
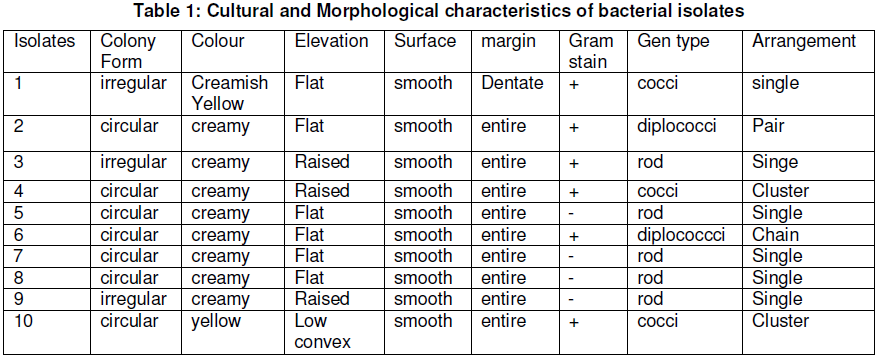
Morphological characterization was done to identify typical colonies with respect to their physical features like cell shape, size, colour, elevation, nature of surface, optical characteristics and pigmentation.
Sensory Test
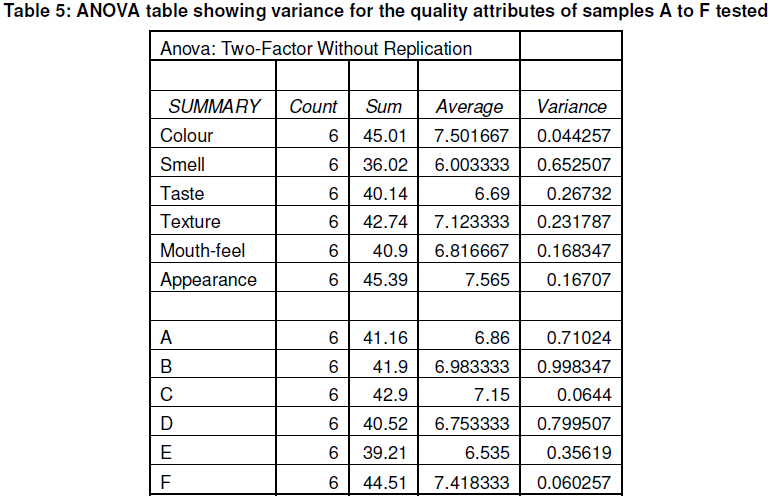
A series of enquiry was carried out on soy yoghurt produced from the various methods (A-F) among 15 panelists and the mean score of their judgements on qualities like colour, smell, taste texture, mouth-feel and appearance was recorded and graded on a 1-10 scale. An extreme dislike was assigned value 1 while an extreme like was assigned 10 with the value of 5 as midpoint.
Activation of Pure Starter Culture
Following the morphological and biochemical test, the identified sample Lactobacillus was activated by dissolving the pure starter culture in 5ml of distilled water and transferred into 10ml of sterile reconstructed low fat milk. Thorough mixing was done and the mixture was incubated at 37oC for 24hrs. The activated culture was then chilled and stored.
Soy Milk Preparation
Soybean used for the preparation of soymilk was bought at the local market in Rivers State. The Soymilk sample was prepared from clean, sorted seeds that have a creamy colour. Six different methods were used in extraction of soymilk namely; hot extraction method, cold extraction method, soaking and blanching method, delay filtration method, and experimental methods like potash processing method and sodium bicarbonate methods(which involves ultrasonic heat treatment) developed by Ogundipe and Osho (1996). The flow sheet for the six methods is shown below;

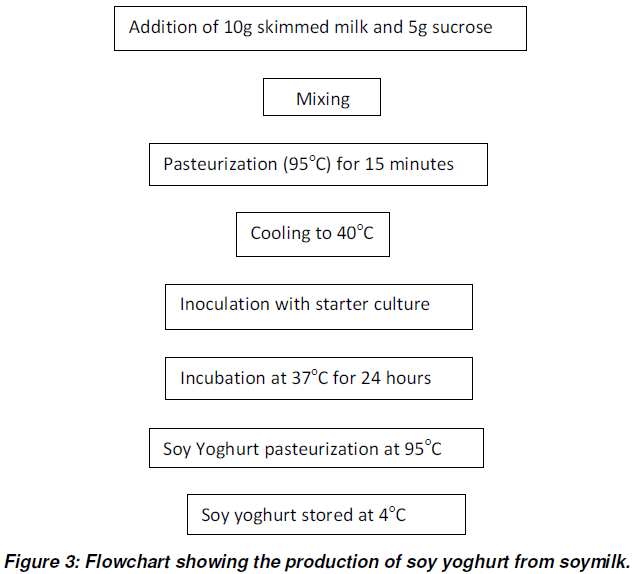
Preparation of Soy Yoghurt
The method utilized for the production of soy yoghurt was reported by Porter (1975). The sample was prepared from soymilk with the addition of 100ml of sucrose and 20g of skimmed milk. The soymilk was pasteurized at 95oC for 15 minutes. It was then cooled in a water bath to a temperature of 45oC. 1ml inoculums of the activated starter culture was added to the soymilk and the dispersion, incubated at a temperature of 37oC for 24hrs. After this period, a firm gel developed and its pH was taken using a pH meter. The soy yoghurt obtained was pasteurized at 95oC for 30 minutes and stored at 4oC until ready for use.
RESULTS AND DISCUSSION
Composition of Microbial Consortium in Ugba
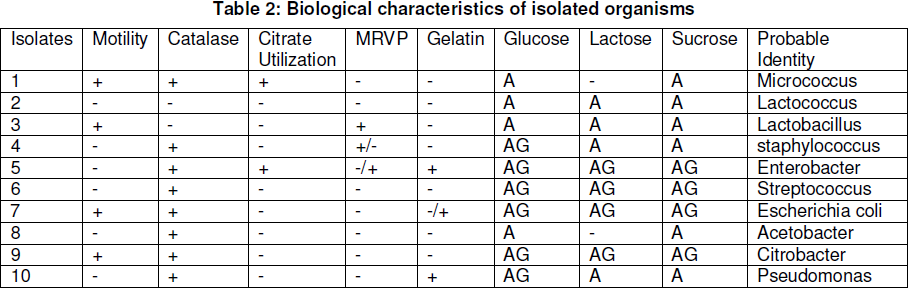
Five samples of Ugba were analysed for total heterotrophic bacterial count. And all the samples examined exhibited microbial contamination. The cultural and morphological characteristics of bacterial isolates are shown in Table 1, while the biological characteristics of isolated organisms are presented in Table 2. Based on the results of the gram reaction and biochemical test performed, the genera of bacteria identified were Micrococcus spp, Lactococcus spp, Citrobacter spp, Staphylococcus spp, Enterobacter spp, Streptococcus spp., Lactobacillus spp., Escherichia coli, Acetobacter spp., and Pseudomonas spp.
Soymilk Composition and Sensory Evaluation
The proximate composition of the soymilk samples are shown in Table 3 while the sensory properties are in Table 4.
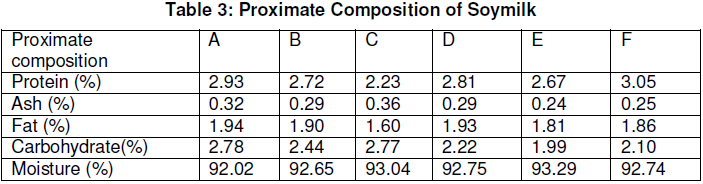
The protein content of the soymilk samples ranged from 2.23% to 3.05% for the delayed filtration and soaking in potash respectively. The high protein value for the delayed filtration is probably due to the fact that blanching had increased the breaking down of the secondary bonds holding down the molecules of the amino acid thus making the protein more soluble in the filtrate and increasing availability of the amino acids. The delay in filtration also helps to increase the solubility of the denatured protein in the filtrate such that the protein content of the soymilk produced is high.
The low value obtained for the potash method is due to the fact that potash reacts with protein and forms a complex, which reduces the protein availability and thus decreases the protein content of produced soymilk. The value for sodium bicarbonate, which is also low, is due to the same reason as that of potash. Soaking as well as blanching gave a higher protein content of soymilk because soaking gives a tender product, which results in finer slurry and thus more filtrate will pass through the filter cloth, increasing yield and subsequently the protein content of soymilk.
The fat content of hot extraction, soaking and blanching, soaking and delayed filtration were significantly different and higher than those with the use of sodium bicarbonate and potash. The high values for the methods in which the soybeans seeds are cooked is because the preheating treatment reduces viscosity of the oil and allows for easy breakdown of oil cells and release of oil.
The lower value for the delayed filtration is due to the fact that the delay period causes the formation of a complex between the fat and starch/ carbohydrate which is not soluble in ether; the normal method used for fat analysis, thus giving a low fat content. This finding is also supported by Ogundipe and Osho (1990).
The sensory evaluation shown in Table 4 shows that samples produced using potash and sodium bicarbonate have a higher preference for smell, texture and taste because the methods reduced the beany flavour to a bearable extent as reported by Liu (1997). However these samples have the lowest preference in terms of colour.
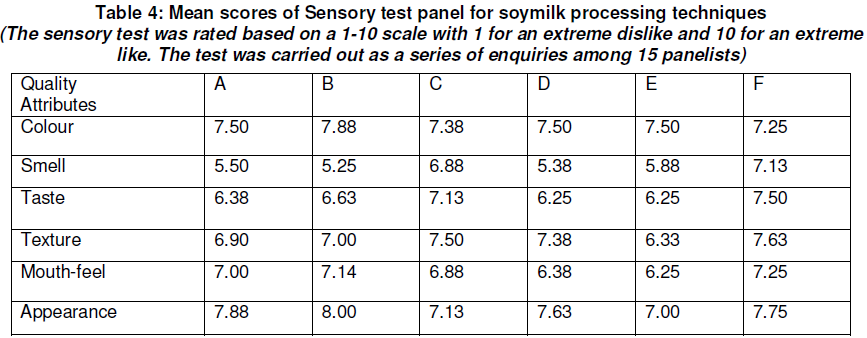
It is observed that these samples had the lowest nutritional composition while soymilk samples with high nutritional compositions had the lowest sensory quality in terms of smell, texture and taste. This shows that the beany taste of soymilk is still a problem that can hinder soymilk utilization in the country since processing methods that eliminated the beany taste had a higher preference though they had a lower nutritional value. Also, the different methods of production have different nutritional compositions and therefore the methods chosen should be determined by the important nutritional component that is needed.
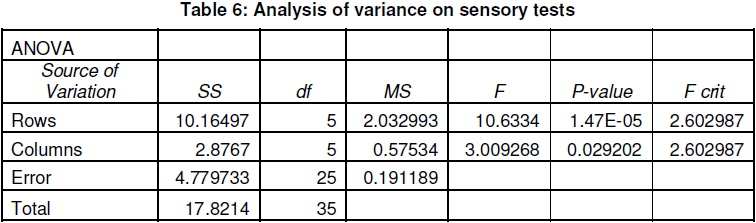
Yoghurt Production, Composition and Sensory Attributes
Lactobacillus spp. (acidophilus/delbruecki) was used for fermentation. The culture obtained from the laboratory and then transferred to soybean milk medium. The culture was incubated at 37oC for 24 hours and was serially transferred medium before using as a starter. The culture was grown singly and added to soybean milk medium for yoghurt fermentation. Although sucrose does not significantly affect the growth of Lactobacillus spp., 5 % sucrose was added to the soybean milk to give a balanced taste of sweetness and sourness to the fermented product. Sucrose-enriched soybean milk however is still low in titrable acidity after 24 hours at 37oC in starter culture fermentation. However, high temperatures could also have adverse effects on the quality of the fermented product. Hence, it is essential to find in optimum sterilization or pasteurization condition. Data in table 7 reveal that yoghurt made from soybean milk sterilized at 90oC had desirable colour, taste and titrable acidity, but very soft texture. In addition, to a slightly bitter taste, yoghurt made from soybean milk sterilized at 121oC had a dark colour and harder texture.
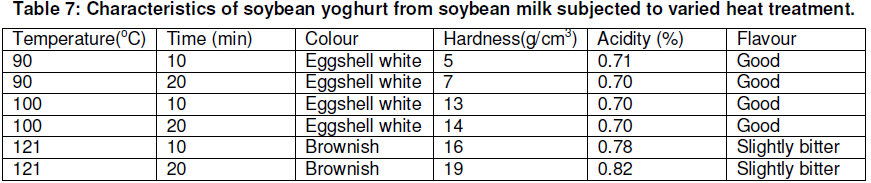
A satisfactory product, however, can be obtained from soybean milk heated at 100oC for either 10 or 20 minutes. Such flavours as vanilla, orange, strawberry, or lemon, if desired, can be added to soybean milk before inoculation. Texture, taste, and colour are important qualities of food. In the case of yogurt, viable bacterial count is also critical, because many yoghurt consumers consider the presence of active lactic acid bacteria an essential feature of yoghurt.
As indicated in Table 8, in yoghurt, the protein content, acidity, and hardness are factors that have a high value when the ratio of water extracted to bean is at 6:1 than 10:1.
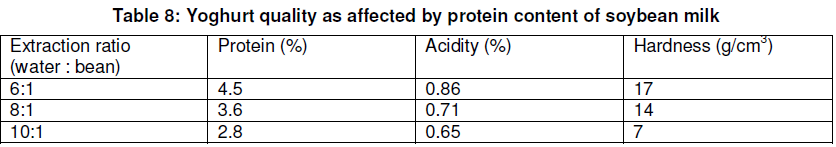
As indicated in Table 9 and Figure 4, soybean yoghurt can be kept at 5oC for about 19 days without any significant change in acidity and viable cell count. After that time, the study showed that the viable count was reduced to a point that many yoghurt consumers might consider too low. Although the viable cell count of yoghurt remained high after about 19 days at 20oC, titrable acidity was 0.8% of lactic acid. When the acidity was higher than 0.9%, the yoghurt had a harsh flavour.
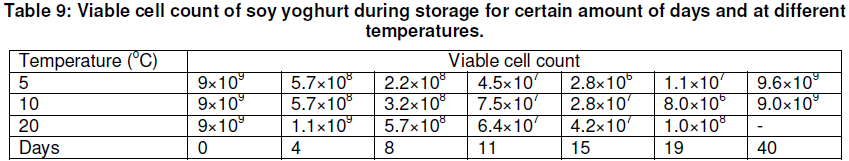
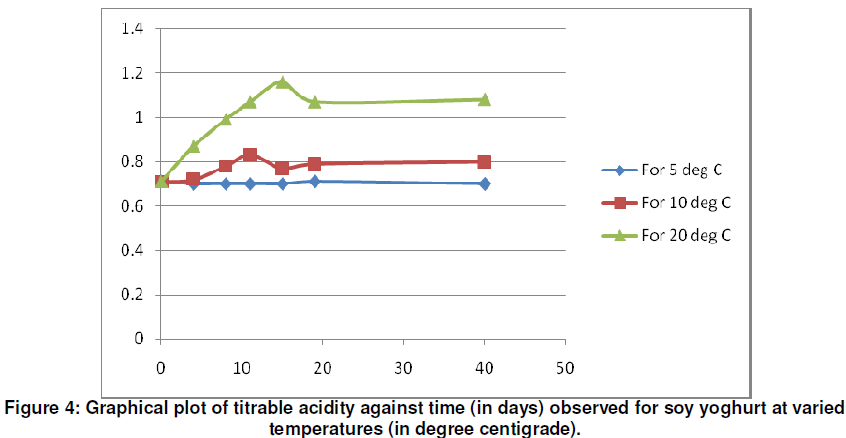
The graphical representation for titrable acidity (% lactic acid) against time (in days) as shown in figure 4 for temperatures of 5oC, 10oC and 20oC indicate that at 5oC, titrable acidity for soy yoghurt is fairly constant for the 40 days for which the investigation was carried out. At 10oC, the titrable acidity remained within acceptable limits for the 4 days considered whereas at 20oC, the titrable acidity rose to above 0.9% which indicates a harsh flavour for the yoghurt.
CONCLUSION
The different methods of production of soy yoghurt from soymilk have varied nutritional composition and therefore the method chosen should be determined by the preferred nutritional components.
Yoghurt made from soybean milk sterilized at 90oC had desirable colour and taste but very soft texture whereas yoghurt made from soybean milk sterilized at 121oC had a dark colour, harder texture and a slightly bitter taste. A satisfactory product, however, can be obtained from soybean milk heated at 100oC for either 10 or 20 minutes. Such flavours as vanilla, orange, strawberry, or lemon, if desired, can be added to soybean milk before inoculation.
Soybean yoghurt can be kept at 5oC for about 19 days without any significant change in acidity, pH and viable cell count. After that time, the study showed that the viable count was reduced to a point that many yoghurt consumers might consider too low.
Yoghurt should be prepared in a good hygienic condition so as to reduce the microbial load. Also care should be taken during processing and storage of the yoghurt samples to prevent contamination with unwanted materials and micro-organisms such as staphylococcus species (e.g. staphylococcus aureus) and coliforms (e.g. Escherichia coli).
REFERENCES
Achinewhu SC (1986). The effect of fermentation on carbohydrate and fatty acid composition of African oil bean seed (P. macrophylla). Fd. Chem., 19:105-116.
Buchanan RE and Gibbons NC (1974). In Bergey’s Manual of Determinative Bacteriology. 8th edition. Baltimore: Williams and Nilks, pp 56-73.
Campbell-Platt G (1987). Fermented foods of the world - a dictionary and guide. London, Butterworths. ISBN: 0-407-00313-4.
Collins CH and Lyne PM (1976). Microbiological Methods. 4th edition. London: Academic Press, pp 954.
Zilberman D and Eunice K (2011). The Lessons of Fermentation for the New Bio-Economy. AgBioForum, 14(3): 97-103.
David OM Aderibigbe EY (2010). Microbiology and proximate composition of ‘ogiri’, an oily paste produced from different melon seeds. New York Science Journal, 2010; 3(4): pp 18-27.
Elaine M and Danilo M (2011). Traditional fermented food and breverages for improved livelihood. Rural Infrastructure and Agro-Industries Division Food and Agriculture Organization of the United Nations, Rome 2011, 21: 15.
Enujiugha VN and Agbede (2000). Development of a new food paste from Pentaclethra species. Appl. Trop. Agri., 5:89-94.
Enujiugha VN (2003). Nutrient changes during the fermentation of African oil bean seeds (Pentadethra macrophylla benth). Pak J. Nut., 2:320-323.
Isu NR and Njoku HO (1997). An evaluation of the microflora associated with fermented African oil bean (Pentaclethra macrophylla Bentham) seeds during ugba production. Plant Foods Hum Nutr. 1997; 51(2):145-157.
Liu K (1997). Soybeans: Chemistry, Technology and Utilization. New York. Chapman and Hall. http://www.soyfoods.com
Nwamarah JU and Madueke UO (2010). Graded Fermentation Effects on Nutrient Content of Oil Bean Seed (pentaclethra macrophylta) Consumed in Umuayom Village, Awka. JHER, 13: 16-23.
Oyewole OA and Isah P (2012). Locally Fermented Foods in Nigeria and their Significance to National Economy: a Review J Rec Adv Agri, 1(4): 92-102.
Ogundipe HO and Osho SM (1990). Soybean Processing. National workshop on soybean Processing and Utilization for improved Nutrition Training manual. IDRC/IITA/IAR and soybean Utilization project, Ibadan, Nigeria.
Prescott LM, Harley JP and Klein DA (1996). Microbiology. 3rd edition. William C Brown Publishers. Dubuque, IA, USA.
Skerman VBD (1967). A Guide to the Identification of the Genera of bacteria. 2nd edition. Baltimore: William and Nilks, pp 81-99.
Thiery I and Frachon E (1997). Identification, isolation, culture and preservation of Entomopathogenic Bacteria. In: Lacey AL. ed. Manual of Techniques in Insect Pathology. Academic Press; 1997: pp. 55-73.
Wikipedia (2012). http://en.wikipedia.org/wiki/Soy_yogurt
William S and Akiko A (2007). History of Fermented Soymilk and Its Products. A Special Report on The History of Modern Dairy-like Soyfoods. A Chapter from the Unpublished Manuscript, History of Soybeans and Soyfoods: 1100 B.C. to the 1980s. Soyinfo Center, Lafayette, California.
|
Cite this Article: Opara CC, Kuru T, Ezenwaka IB (2013). Production of Soy yoghurt from Lactobacillus isolated from fermented African Oil Bean Seed (Ugba). Greener Journal of Agricultural Sciences, 3(2): 110-119, http://doi.org/10.15580/GJAS.2013.2.112412293. |