|
Greener Journal of Biochemistry and Biotechnology
Vol. 6(1), pp. 01-11, 2019
ISSN: 2384-6321
Copyright ©2019, the copyright of this article is retained by the
author(s)
DOI Link: http://doi.org/10.15580/GJBB.2019.1.070719132
http://gjournals.org/GJBB
|

|
A
Comparative Study of Secondary Metabolites, Amino acids
and Protein Profiles of the Host – Parasite Plants in the Relationship between
the African Mistletoe, Tapinanthus
bangwensis [Engl. and K.
Krause] Danser and Two of its Host species
Edagbo, David Enuwa1*;
Prof. Oyetunji, Olusola Jacob2
Plant Genetic Resources Unit, National Centre for Genetic
Resources and Biotechnology, Ibadan, Nigeria.
Email: dedagbo@ yahoo.
com;
Department of Botany, University of Ibadan,
Ibadan, Nigeria. Email: solatunji2k2@ yahoo. com
|
ARTICLE INFO
|
ABSTRACT
|
|
Article No.: 070719128
Type: Research
DOI: 10.15580/GJBB.2019.1.070719132
|
The presence of
secondary metabolites, amino acids and sugar alcohol including protein
profile in the host-parasite relations of Tapinanthus
bangwensis (Tb) on the host
plants, Citrus sinensis (Cs) and Irvingia gabonensis (Ig)
were evaluated. Secondary metabolites contained in the host-parasite plants
was observed for the Citrus-parasite
(tannin - 0.35 Tb, 0.21 Cs; phenol - 0.21 Tb, 0.14 Cs;
alkaloids - 1.42 Tb, 0.17 Cs g/100g) and Irvingia-parasite
(tannin - 0.23
Tb, 0.18 Ig; phenol - 0.19 Tb, 0.15 Ig; alkaloids - 1.34 Tb, 0.57 Ig g/100g) with comparative higher
quantities in the parasite. Free amino acids and sugar alcohol contents in
the leaves of host plants revealed the constituents of some of the groups in
the infested (tyrosine - 12.26 Cs, 14.70
Ig; aspartic acid - 12.21 Cs,
11.23 Ig ng/g) and uninfested
(tyrosine - 10.76 Cs,12.93 Ig; aspartic acid - 9.09 Cs, 9.79 Ig ng/g) which indicated significant higher values for the
infested. Protein profiling of the Citrus
leaves revealed lack of protein at 25.0 kDa band in the infested host.
Assessment of metabolites and protein features in the mistletoe-host
relationship affirmed presence of similar metabolites in the host-parasite
entity while infested hosts had increased free amino acids and there were
noticeable variations in protein banding pattern of host plants with
mistletoe incidence.
|
|
Submitted: 07/07/2019
Accepted: 14/07/2019
Published: 29/08/2019
|
|
*Corresponding
Author
Edagbo, David Enuwa
E-mail: dedagbo@ yahoo. com
|
|
Keywords: Tapinanthus bangwensis; Citrus sinensis; Irvingia
gabonensis; infested hosts
|
|
|
|
INTRODUCTION
Mistletoes are generally parasites which
have form an intimate physiological connection with their hosts often leading
to change and or adaptive variation in phenotype (and usually as well genetic
change) for them and their host plants. Mistletoe plants may become so
intrinsically connected to the host that it may seem like another branch (in
cases with strong mimicry); but mistletoe on host is separate and distinct with
its metabolic process and photosynthetic interaction for sustenance. The
various groups of mistletoes in interaction with their host gave rise to
different display of physiological dependence on host photosynthesis. The
hemiparasite autotrophs rely on water and inorganic nutrients supply of the
host through their (host) xylem while the holoparasites, in addition to
depending on water and inorganic solutes of their host make use of the photosynthate
of their host (Glatzel and Geils, 2009). Mistletoe–host compatibility is a function of host
susceptibility to infection and of mistletoe infectivity (Yan, 1993). If
mistletoes are physiologically, biochemically and physically compatible with a
tree, they will have a chance to germinate, establish and survive on that tree
(Roxburgh and Nilcolson, 2005).
Most genera of African mistletoes belong to the family Loranthaceae (Polhill and Wiens, 1998). In
West Africa and Nigeria in particular, mistletoes are found on many agronomic
tree crops which include the shea butter tree (Vitellaria paradoxa Gaertn.
f.), Citrus species; mostly sweet
orange (Citrus sinensis L.) and grape (Citrus paradisi L.), and
cocoa (Theobroma cacao L.). Different species of these hemi-parasitic
plants grow on other medicinal as well as cultivated trees such as the
brimstone tree (Morinda lucida Benth), the kola-nut tree (Cola nitida
Vent. Schot and Endl.), forest trees such as Irvingia gabonensis (Aubrey-Lecomte ex O. Rorke) Baill, Parkia biglobosa,
among a host of other tree crops (Wahab et al., 2010; Adesina et al.,
2013; Ibrahim et al., 2014).
Mistletoes are used severally in traditional medicine practice. The
group, especially of the Loranthaceae and Viscaceae are widely used by
different races and cultures in almost every continent to treat various
ailments including hypertension and diabetes, or used as a diuretic agent
(Adesina et al., 2013). The plant is
ethnomedicinally used by the different ethnic groups in Nigeria as a remedy for
several human and animal ailments such as dysentery, diarrhoea, convulsion
(Ilesanmi and Olawoye, 2011), cardiovascular diseases and gynaecology problems
(Adodo, 2004). It has been found to have anti-microbial properties against
certain multiple-drug-resistant bacterial and fungal isolates of farm animals
(Deeni and Sadiq, 2002).
There had been observed variation in secondary
metabolites among same mistletoe species occurring on different host plants
(Wahab et al., 2010; Ilesanmi and
Olawoye 2011; Umoh et al.,
2011; Ibrahim et al., 2014). The screening for phytochemical substances in
various species of the African mistletoes reflects divergent and varying
constituent of the Alkaloids, Saponin, Tannin, Phlobatanin,
Anthraquinone, Cardiac glycosides, Cardenolides, Steroidal nucleus, reducing
sugar, flavonoids among others in their composite formation.
It has been suggested that pharmacologically active compounds may pass
from the host trees to the parasitic plants. Thus, biological activities of the
parasitic plant could differ, just as the apoptosis-inducing properties of Viscum
album extract has been found to be
host dependent (Bussing and Schietzel, 1999). The influence of the host chemistry on the chemical constituents of the
parasite on different hosts might justify why the host is as important as the
parasite in pharmacognosy, ethnopharmacology and ethnomedicine, and why the use
of these mistletoes in the treatment of an ailment is often dependent on a
particular or specific host (Burkill, 1995; Snyder et al., 1996; Adodo, 2002; Olapade, 2002; Preston et al., 2010).
Parasitic
plants often connect to their host through a continuous vascular system (and
plasmodesmata) in some species like Striga
and Cuscuta. This linkage has been reported served in
part for movement of molecules between these plants (Ichihashi et al., 2015).
Macromolecules are naturally transported into the parasite from the host
plant, and several reports have implicated haustorial connections in DNA, RNA,
protein and also viruses translocation between host and parasite (Mower et al., 2004; Roney et al., 2007; David-Schwartz et
al., 2008; Ichihashi et al.,
2015).
Plant reactions and responses to either biotic or abiotic stress
conditions is a complex phenomenon involving alterations in physiological and
biochemical processes, which may result in morphological and developmental
changes (Azevedo
Neto et al., 2009). In
plants, most of the observed stress conditions are generally correlated with
enhanced proteolytic activity and increased protein turnover, leading to either
acclimation to the stress condition or to senescence and subsequent cell death
(Martinelli et al., 2007). Several
investigations have shown increased accumulation of free amino acids especially
proline, during adaptation to various environmental stresses (Simon-Sarkadi et al., 2002; Kaplan et al., 2004; Zuther et al., 2007; Kempa et al.,2008; Sanchez et al.,
2008; Usadel et al., 2008; Lugan et al., 2010; Murugan et al., 2014).
The accumulation of soluble sugars and
sugar alcohols (e.g. pinitol) is known to be another common metabolic response
of higher plants to stress. Pinitol is a common sugar alcohol present in a wide
range of plant species including legumes and some other higher plants. A
handful of the research work on plants under stress conditions of high
temperature, salinity and drought, so far revealed the potential physiological
acclimation of such plants through a means of increase in pinitol accumulation
(Murakeözy et al., 2002; Nayyar 2003;
Griffin et al., 2004; Li‐Xia et al., 2008).
Considering the extent of work done on
African mistletoes, there are few staggered investigations on presence of
secondary metabolites in this group of parasites with even scanty efforts on
amino acids and protein profiles of the parasites; especially with respect to
their host plants. Most of the available works have not extensively addressed
the host-parasite relationship along the path of products of their secondary
metabolites, amino acids and protein profiles. Therefore, this research was undertaken to investigate the likelihood of
correlation in the output of secondary metabolites from mistletoe and its host
plants; and to also determine if the presence of mistletoe has influence on the
amino acids and protein profiles of the host species.
MATERIALS
AND METHODS
Site of the study
Stems and leaves of mistletoe on Citrus and Irvingia as
well as same from host plants were collected, cleaned and dried in the sun. The
dried materials were ground to powder and preserved in air tight containers for
the necessary analyses. The samples were collected from plantation
fields at Moor Plantation, Apata, Ibadan, South-Western, Nigeria (located at
latitude, 0703872’ - 0703860’N; longitude, 00308420’
- 0030 8415’E; and at an altitude of 3 m). The laboratory works were
conducted at the Central Laboratory / Biotechnology Centre, Federal University
of Agriculture, Abeokuta (FUAAB) and the Institute of Agricultural Research and
Training (IAR&T).
Phytochemical Screening of Secondary Metabolites
Qualitative Analysis
Test for the presence of tannins, phenols, saponins, alkaloids,
flavonoids, oxalate, phytate, terpenes,
and steroids content of the plant were determined by the methods described by
Trease and Evans (1989), Early
and De Turk (1944) and Sofowora (1982).
Quantitative Analysis
Determination of
Tannic Acid (Tannin)
One gramme (1g) of each sample of the mistletoe and host plants was
weighed into a beaker. A solvent mixture containing 80 ml acetone and 20 ml
glacial acetic acid was used to extract tannin by soaking each sample for 5
hours. Filter paper with double layer used to filter the samples and the
filtrates obtained were then removed. A set of standard solution for tannic
acid ranging from 10 ppm to 50 ppm was prepared. With the use of spectronic 20,
absorbances of the standard solution and filtrates were read at 500 nm
wavelength.
Percentage tannin was obtained with the calculation below:
% Tannin= Absorbance X Average
gradient X Dilution factor
10, 000
Determination of
Total Phenol
One gramme (1g) of each sample of the mistletoe and respective
(infested) host plants was weighed and transferred into 250 millilitres conical
flask. Twenty millilitres (20 ml) volume of distilled water was used in soaking
each sample. These samples were filtered after 4 days and each filtrate
measured was marked up with distilled water to 100 ml in volumetric flask. A
(one) 1 ml volume of the filtrate from each sample was transferred into a test
tube and 3 ml each of 0.01 N Iron (III) chloride and 0.008 N Potassium
hexacyanoferrate (III) were added into each filtrate. Absorbance of the
filtrate from each sample was noted after 10 minutes at 760 nm. Total
polyphenol percentage was measured as below:
% Total polyphenol
= Absorbance X Average
gradient X Dilution factor
Weight of sample X 10, 000
Determination of
Saponin
Twenty grammes (20 g)
each of the samples of mistletoe and host plants was weighed and transferred
into a conical flask and 100 ml of 20% isobutylalcohol (octanol) was added thus
heated over a hot water bath for 4 h at about 55°C with continuous stir for
thorough mixing in order to obtain a uniform solution. Filtration was then
performed on the mixture and the residue re-extracted using another 200 ml 20%
ethanol. Reduction of the combined extracts to 40 ml over a water bath was
undertaken at about 90°C. The concentrate was transferred into a 250 ml
separatory funnel and 20 ml of diethyl ether was added and shaken vigorously.
The ether layer was discarded while the aqueous layer was recovered and a
repeat of the purification process was done. 60 ml of n – butanol was added and
the combined n – butanol extracts were washed twice with 10 ml of 5% aqueous
sodium chloride. The remaining solution was then heated in a water bath after
evaporation; the samples were dried in the oven to a constant weight.
Percentage saponin was obtained with the calculation below:
Saponin in % =
Residue weight X 100
Sample weight
taken
Determination of
Total Alkaloids
Five gramme (5 g)
each of the samples of mistletoe and host plants (stems and leaves) was weighed
and transferred into beaker of 250 ml volumetric capacity into
which was added 200ml of 10% solvent of acetic acid in
ethanol. The beaker containing the mixture was covered (to check evaporations
of solvent) and allowed to stand for 4 hours. This was filtered and the extract was concentrated
on a water bath to one – quarter of the original volume. The whole solution was
allowed to settle and the precipitate was collected and washed using dilute
ammonium hydroxide which was ultimately subjected to filtration. The precipitate residue which was dried and weighed is the
alkaloid.
Weight of
total alkaloids: W2 – W1 g,
W3
% Yields of
Alkaloid: W2 – W1 X 100
W3
Where,
W1 = crucible weight, W2 = crucible
and alkaloids weight, W3 = plant sample
initial weight taken for estimation.
Determination of
Flavonoids
One gramme of the powdered stem and leaf samples of the mistletoe and
infested host plants (Citrus and Irvingia) was weighed into 250 ml
flask. Warm distilled water of 20 ml was added to the sample and placed inside
water bath for 10 minutes at 1000C. Subsequently, filtration was
done. With the use of a pipette, 1 ml of the filtrate was collected and emptied
into a clean test-tube while 1 ml of 0.5 N NaOH was added. Distilled water of
about 8 ml was added and allowed to stand for 10 mins. Using spectronic 21D,
absorbance was measured at 410 nm wavelength. The standards were prepared using
2.5 ppm, 1.5 ppm, 1 ppm and 0.5 ppm.
Calculation: absorbance x average gradient x dilution factor
Determination of
Oxalate
Extract of samples (obtained from powdered stem and leaf) of the
mistletoe (Tapinanthus bangwensis)
and host plants (Citrus sinensis and Irvingia gabonensis) each weighing one
gramme was placed into 250 ml conical flask soaked in 100 ml distilled water.
The soaked samples after 3 hours were each filtered through a filter paper of
double layer. A standard solution of oxalic acid was prepared which comprised
10 ppm, 20 ppm, 30 ppm, 40 ppm and 50 ppm. The absorbance was then measured on
spectrophotometer at 420 nm wavelength. Absorbance of filtrate obtained from
each sample was also measured on the spectronic 20. The percentage oxalate was
obtained using the expression:
% oxalate = sample absorbance X average gradient
from the curve for standard X dilution factor
10, 000
Determination of
Phytic Acid (Phytate)
Two (2) grammes of the sample of each plant materials (the mistletoe and
associated host plants which include the Citrus
and Irvingia) was weighed and
transferred into 250 ml conical flask. Each sample soaked in 100 ml volume of
2% conc. Hcl poured into a conical flask was left for 3 hours after which time
a hardened filter paper with double layer was used for filtration. With 250 ml
beaker into which has been poured 50 ml of each filtrate a 107 ml volume
distilled water was added in each case for proper acidity. To each solution was
added an indicator containing 10 ml solution of 0.3% ammonium thiocynate. With
the use of standard Iron (III) chloride solution containing 0.0019 g iron per
millilitre, the solution was titrated. The slightly brownish – yellow end point
which persisted for about 5 minutes was the positive indicator. Percentage
phytic acid was obtained with the calculation below:
% Phytic acid = X
X 1.19 X
100
2
Where X =
Titre value X 0.00195
Determination of
Terpene
With a 50 ml conical flask in place and weighing 0.50 g therein
(powdered stem and leaf samples each obtained from the mistletoe and attached
hosts), 20 ml volume of 2:1 chloroform-methanol mixture was added and
thoroughly shaken before allowing the whole mixture to stand for 15 minutes.
The mixture was subsequently centrifuged for yet another 15 minutes. Hereupon,
another 20 ml chloroform-methanol mixture was used to rewash the precipitate
got for re-centrifugation while the supernatant obtained was discarded.
A solution of 40 ml volume of 10% Sodium deodocyl sulphate was used to
dissolve the resultant precipitate. One millilitre (1 ml) of 0.01M Ferric
chloride solution was added to the above at 30 seconds interval shaken well and
allowed to stand for 30 minutes. Using a stock of 100 mg/l terpenes solution,
concentration range of 0-5 mg/ml standard terpenes was prepared. Absorbances
for sample as well as standard concentrations of terpenes were read on a
Digital Spectrophotometer at a wavelength of 510 nm. Percentage terpene
was obtained with the calculation below:
Absorbance of sample X dilution factor X gradient factor
Wt. of
sample X 10, 000
Determination of
Steriods
A Half gramme (0.50 g) extract, each obtained from powdered sample (stem
and leaf) of Tapinanthus and the
infested hosts, Citrus and Irvingia was weighed and transferred
into a 100 ml beaker. To dissolve the extract, a mixture of 20 ml
chloroform-methanol (2:1) was added; then placed on a shaker and shaken for 30
minutes. The whole mixture was then filtered through a Whatman No. 1 filter
paper into another 100 ml conical flask that was clean and dry.
Resultant residue was rendered steroids-free by repeated treatment with
chloroform-methanol mixture. To achieving a homogenous mixture, 1 ml of
filtrate got with a pipette was poured into a 30 ml test tube then 5 ml volume
of alcoholic KOH was further added and thoroughly shaken. The mixture was
subsequently set in a water bath which had been fixed at 370C – 400C
for 90 minutes, afterwards allowed to cool to ambient temperature and 10 ml
Petroleum ether added with further 5 ml of distilled water also added. This
mixture left in a water bath was heated to dryness. To the residue in dry
bottle was added 6 ml Liebermann Burchard reagent and absorbance taken at
wavelength of 620nm on Spectronic 21D digital spectrophotometer.
Prepared from 100 mg/ml stock of standard steroids solution were
standard steroids of concentration range between 0-4 mg/ml and treated similarly
like sample as above.
% Steroid was calculated as below:
Absorbance of sample X Gradient X Dilution
Factor
Wt of
sample X 10000
Determination of
Trypsin Inhibitor
A sample (powdered stem and leaf) of 0.2g of the mistletoe and host
plants was transferred into a screw cap centrifuge tube with 10 ml 0.1M
phosphate buffer addition and the content was shaken at room temperature on a
UDY shaker for 1 hour. The suspension obtained was filtered through whatman No.
42 filter paper after centrifugation at 5000 rpm for 5 mins. With phosphate
buffer, the volume of each filtrate was adjusted to 2ml. The test tubes were
placed in water bath, maintained at 37oC. Six millilitres (6ml) of
5% TCA solution was added to one of the tubes previously kept at 37oC.
These were incubated for 20 mins. The reaction process was stopped after 20
mins by adding 6ml of TCA solution to the experimental tubes and shaken. The
reaction was allowed to proceed for 1 hour at room temperature. Filtration of
the mixture obtained was carried out using Whatman No. 42 filter paper. The
filtrate from sample and trypsin solutions was read at 280 nm for their
absorbance. The trypsin inhibitor measured in mg/g sample was obtained as
below:
T. I. mg/g = A std
- A sample X
Dilution factor
0.19 X
sample wt in g 1000 X
sample size
Determination of Free
Amino Acids
The ninhydrin colorimetric analysis method of Rosen (1957) was adopted
for the (free) amino acids test. Extracts were got from leaf samples of Citrus sinensis and Irvingia gabonensis with each diluted as appropriate. To 1 ml of
the diluted leaf extract in methyl cellosolve were added 0.5 ml of 3% ninhydrin
and 0.5 ml Cyanide acetate buffer. Using water bath, the mixture was heated for
15 mins at 1000C. A further addition of 5 ml isopropyl alcohol water
mixture with vigorous shaking was soon carried out. The colour of the mixture
was read in colorimeter at 570 nm after it had been left to cool. Based on the
known concentration of various amino acids, concentrations of the amino acids
were calculated and obtained from a standard graph.
SDS-Page Test for
Extract of Soluble Proteins from Leaf Specimens
800µl. 0.1M Tris-Hcl of pH 7.6 was added to 0.3g of the leaf specimen
(obtained from the infested and uninfested host plants of Citrus and Irvingia)
ground to powder which was subsequently subjected to vortex for 1 min; then
spun down at the speed of 10, 000 rpm for 5 minutes. Resultant supernatant collected was placed in
a new Enpendorff tube and kept at 4oC. The gel was prepared at 12%
separating gel and 4% stack gel; the test material and the standard were thus
loaded for the gel to be run at 150v for 45 minutes. The resultant product was
stained in commassie blue for 45 minutes and later de-stained with de-staining
solution with several changes until bands showed clearly.
Statistical Analysis
Data obtained was considered statistically by using the SPSS 21
Statistics Program. Statistical analysis was done using one way analysis of
variance (ANOVA) followed by Duncan’s Multiple Range Test (DMRT).
RESULTS
Table I
showed the
status of secondary metabolites as obtained in the mistletoe (Tapinanthus bangwensis) in relation to
its two hosts; Citrus sinensis and Irvingia gabonensis. Tests for tannin
indicated strongly positive presence (+++) in the mistletoe on Citrus while the host plant had positive
(++) outlook. Indication of presence of tannin in the Irvingia–mistletoe association was positive in the host while it
was strongly positive in the parasite. Phenol presence in the mistletoe and the
two hosts (Citrus and Irvingia) was positive. Examination for
saponin in both mistletoe and its Citrus
host yielded positive presence. Also saponin in the mistletoe on Irvingia was positive (++) compared to
the trace (+) indication of saponin presence in the Irvingia. Alkaloid was strongly positive (+++) for the mistletoe on
Citrus in comparison to its positive
indication (++) in the host. However, alkaloids presence was positive for both
the mistletoe on Irvingia and the
host. The mistletoe on Citrus and the
host manifested positive presence of flavonoid. The presence of flavonoid
observed in the mistletoe on Irvingia
was positive (++) but trace (+) for the host plant. The mistletoe on Citrus and Irvingia together with the two hosts gave indications of positive
presence of oxalate. Phytate presence in the mistletoe on Citrus and Irvingia and
the two hosts as well was positive. It was a reflection of trace presence of
terpenes in the mistletoe on Citrus
and the host but the mistletoe on
Irvingia showed trace presence compared to the positive presence observed
in its host. Trace presence of steroids was observed in the mistletoe on Citrus while steroid presence was
positive in the host. The Irvingia–mistletoe
association reflected trace presence of steroids for the parasite and host.
TABLE I: Secondary Metabolites presence in Tapinanthus
bangwensis and its two hosts
Sample Tannin
Phenol Saponin Alkaloids
Flavonoids Oxalate Phytate
Terpenes Steroids
T. bangwensis +++ ++ ++ +++ ++ ++ ++
+ +
on Citrus sinensis
Citrus sinensis ++ ++ ++ ++ ++ ++ ++ + ++
T. bangwensis +++ ++
++ ++ ++ ++ ++ + +
on Irvingia gabonensis
Irvingia gabonensis ++
++ + ++ + ++ ++ ++ +
|
KEY: + = TRACE; ++ = POSITIVE; +++ = STRONGLY POSITIVE
|
The quantitative array of secondary
metabolites present per each host species in relation to its attached mistletoe
is shown in Table II. The observed amount of tannin in the mistletoe (0.35
g/100g) on Citrus was significantly
higher (α = 0.05) than the amount obtained in the host.
The mistletoe on Irvingia also had
higher tannin content than its host. The phenol content of Tapinanthus on the Citrus
and Irvingia hosts was higher than
the observed content in each host. Saponin in the mistletoe on Citrus and that of its host was not
statistically different but the mistletoe on Irvingia had significantly higher amount than what was available in
the host. The alkaloid in the mistletoe (1.42 g/100g) on Citrus was significantly higher (α = 0.05) than
that in the host and similarly the mistletoe on Irvingia had higher alkaloid content than the host. Flavonoid in
the mistletoe (1.45 g/100g) on Citrus
was significantly higher than that of the host but the Irvingia host plant however had significantly higher quantity (0.31
g/100g) of flavonoid than its parasite. The quantity of the oxalate in Citrus host was significantly higher
than its parasite while the parasite on Irvingia
had higher amount than in its host. Phytate in the mistletoe (0.41 g/100g) on Citrus was significantly higher than
that in the host and the phytate content in the mistletoe (0.52 g/100g) on Irvingia was also higher than in its
host. The observed amount of terpenes present in the mistletoe on the Citrus and Irvingia hosts was significantly higher than the respective host.
Steroid obtained in the mistletoe on Citrus
and Irvingia hosts was in higher
amounts than that available in the hosts. Trypsin inhibitor content of the
mistletoe and both Citrus and Irvingia hosts were statistically
similar.
TABLE II: Secondary
Metabolites contents in Tapinanthus
bangwensis and its two hosts
Sample Tannin
Phenol Saponin Alkaloids
Flavonoids Oxalate Phytate
Terpenes Steroids Trypsin
g/100g
g/100g g/100g g/100g g/100g g/100g g/100g
g/100g g/100g inhibitor
g/100g
T. bangwensis 0.35a 0.21a 0.18c 1.42a 1.45a 0.50c 0.41b 0.08b 0.11a 0.13a
on Citrus sinensis
Citrus sinensis 0.21c 0.14d 0.19b 0.17d 0.22c 1.24a 0.12d 0.02d 0.01c 0.13a
T. bangwensis 0.23b 0.19b 0.80a 1.34b 0.18d 0.62b 0.52a 0.09a 0.02b 0.13a
on Irvingia gabonensis
Irvingia gabonensis 0.18d 0.15c 0.19b 0.57c 0.31b 0.45d 0.33c 0.03c 0.01c
0.11b
|
Means followed by the same letter in each column are not significantly
different by DMRT at α = 0.05
|
Shown in Table III were the results on content of
the free amino acids and stress metabolites in the leaf specimens of infested
and uninfested Citrus and Irvingia host plants.
Tyrosine in the infested leaf of Citrus (12.26 ng/g) was significantly higher (α = 0.05) than the uninfested. And the infested leaf of Irvingia (14.70 ng/g)
also possessed significantly higher quantity of tyrosine than the uninfested. Phenylalanine in the uninfested Citrus (14.15 ng/g) was significantly higher than the infested but the infested leaf of Irvingia (16.97 ng/g) had significantly higher
phenylalanine content than the uninfested. The serine
assessment showed that the infested leaves of the Citrus (11.49 ng/g) and
Irvingia (13.49 ng/g) hosts had higher contents of the amino
acid than the uninfested. Glycine in the Citrus and Irvingia hosts showed contrasting outcomes as the uninfested Citrus (10.08 ng/g) had higher glycine than the infested while the infested Irvingia (11.07 ng/g) had higher value than the uninfested. The Aspartic acid
content in Citrus revealed the infested leaf had significantly higher quantity (α = 0.05) than the
uninfested and the same it was for the Irvingia leaf. Proline content of the infested Citrus leaf was significantly higher than the
uninfested while it was similar in the infested and uninfested Irvingia leaves. Cysteine in the infested Citrus (9.16 ng/g)
was significantly higher than the uninfested whereas the infested and
uninfested Irvingia possessed cysteine content that was not significantly different.
Isoleucine (7.97 ng/g) in the infested Citrus was
significantly higher than the uninfested but Irvingia had similar isoleucine content in both the infested and uninfested
state. Pinitol was significantly higher in the infested leaves of the Citrus and Irvingia hosts.
TABLE III: Free Amino acids and sugar alcohol in the infested and uninfested Citrus
sinensis and Irvingia gabonensis hosts
Sample Tyrosine
Phenylalanine Serine Glycine
Aspartic Acid Proline Cysteine
Isoleucine Pinitol
ng/g
ng/g ng/g ng/g ng/g ng/g ng/g ng/g ng/g
Infested Leaf 12.26b 12.81c 11.49b 9.34c 12.21a 8.91a 9.16b 7.97c 6.78b
of Citrus
sinensis
Uninfested Leaf 10.76c 14.15b 10.55c 10.08b 9.09c 6.88c 8.27c
6.24d 5.76c
of Citrus sinensis
Infested Leaf 14.70a 16.97a 13.49a 11.07a 11.23b 7.89b 10.83a 8.83a 7.98a
of Irvingia gabonensis
Uninfested Leaf 12.93b 12.71c 11.95b 10.31b 9.79c 7.42bc 10.34a 8.27b 6.87b
of Irvingia gabonensis
|
Means followed by the same letter in each column are not significantly
different by DMRT at α = 0.05
|
|
|
SDS- PAGE electrophoretic bands in Plate 1 ranged
from 14.7 – 150 kDa molecular weights. Sample 1 lacked protein at the 20.0 kDa
band. Samples 2 and 3 lacked protein at the 20.0 and 25.0 kDa bands. Samples 4
– 6 displayed similar features as they lacked protein at 20.0 kDa band. The
protein bands in the samples 1 - 3 (infested Citrus) were less densely expressed than those in samples 4 - 6
(uninfested Citrus). Generally the
leaf specimens of Citrus (samples 1 -
6) lacked protein at the 20.0 kDa band. In addition however, the infested Citrus leaf specimens lacked protein at 25.0 kDa band.
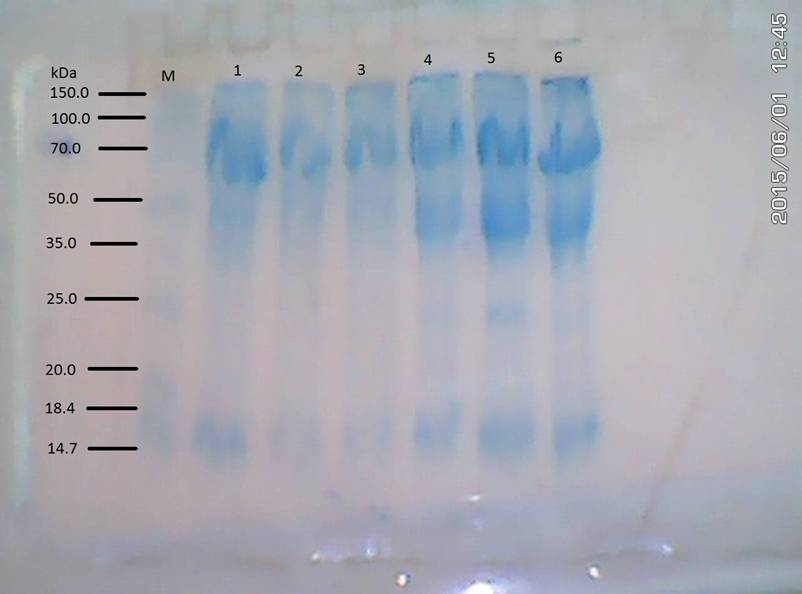
PLATE I: Electrophoregram of the leaf of Citrus sinensis
M: Standard; 1, 2 & 3: leaves of infested Citrus; 4, 5, & 6: leaves of
uninfested Citrus
DISCUSSION
Preliminary
phytochemical screening of secondary metabolites as well as the quantitative
determination of same metabolites from mistletoe and its hosts revealed
variations stemming from each host-parasite association. Generally, the
mistletoe possessed higher quantities of secondary metabolites when compared
with its Citrus
and
Irvingia hosts. In a comparative assessment of the constituent metabolites
in each mistletoe – host pair vis-a-vis the Citrus
and Irvingia hosts; metabolites such
as tannin, phenol, alkaloids, flavonoids and steroids were found to be of
significantly higher quantities in the mistletoe on Citrus while the other metabolites such as saponin, oxalate and
phytate were available in higher quantities in the mistletoe on Irvingia. Similarly, distributions of
secondary metabolites, within the two host plants were similar. The available
metabolites in the mistletoe which are mainly
host dependent have been the major reason for strong consideration of
host source in the utilization of mistletoe for pharmacognosy and ethnomedicine
in places where adopted. Differential accumulation of secondary metabolites in
the mistletoe relative to host source is often thought to avail such mistletoe
the desired potentials which favour their selection in treatment of certain
health challenges (Adodo, 2004; Guimaraes et al., 2007). The output of
secondary metabolites from T. bangwensis
on the basis of its association with the Citrus
and Irvingia hosts were
consistent with the observation made by some researchers (Wahab et al., 2010; Ilesanmi and Olawoye 2011;
Umoh et al., 2011; Ibrahim et al., 2014) who
noted that variations existed in the content of secondary metabolites from same
mistletoe species occurring on different host plants. Again this observation
supports the suggestion of the translocation of pharmacologically active
substances from host plants to parasites via their vascular connecting systems
at structures such as the haustorium (Bussing and Schietzel, 1999).
Evaluation
of the free amino acids and stress metabolite contents of the host-parasite
plants showed that the presence and interaction of mistletoe with the host
plants do exert some measure of influence on the cumulative content of metabolites
in the host plants. Hence, the infested host plants of Citrus and Irvingia which bore the presence of mistletoe accrued
significantly higher quantities of amino acids and stress metabolites when
compared with the uninfested of the same host plants. Although for
phenylalanine and glycine in Citrus the results were in contrast. The free amino acids and stress
metabolite released were distinctly higher for the infested hosts in the Citrus and Irvingia host-parasite
association. This result is in support of similar investigations by researchers
such as Griffin et al., (2004), Liu et al., (2008), Usadel et al., (2008), Lugan et al., (2010) and Murugan et al., (2014) which revealed increased
accumulation of free amino acids, soluble sugar and sugar alcohols in plants as
metabolic responses or otherwise physiological acclimation to various
environmental stresses (both abiotic and biotic). The higher free amino acids
content in the infested samples is a cue that the activities of the mistletoe
might have caused denaturation or breakdown of proteins and bound amino acids,
resulting in enhanced free amino acids content of the host tissues.
Sodium dodecyl sulfate–polyacrylamide
gel electrophoresis (SDS-PAGE) for soluble proteins carried out on the leaves
of infested and uninfested Citrus
showed products of distinct protein profile expressions relative to the
different class of the host plant. The protein banding pattern of the infested
host differed sharply from the uninfested. The protein bands of the infested were
less densely expressed. The uninfested only lacked Protein at 20.0KDa while
protein of 20.0KDa and 25.0KDa were lacking in the infested. This result thus
supported the claim of variations in number and intensity of SDS
electrophoretic bands of proteins observed between infected and healthy plants
(Sharaf et al., 2009; Murugan et al., 2014). The mistletoe’s presence
and activities which could be said to induce perturbation, metabolic reaction
and proteolytic realignment in the sampled plants is by extension seen to have
physico-chemical implication for the host- parasite association. However, no protein bands were noticed in the
outcome of the SDS-PAGE process on the leaves of Irvingia gabonensis. This does not preclude the existence of
soluble proteins in the sample but such might result from the interplay of some
intrinsic factors such as high water content, absence of reducing agents
(dithiothreitol or β-mercaptoethanol buffer) that breaks disulfide bonds,
protein structure, post transitional modification and amino acid composition
which minimizes the effect of secondary structure on migration (Osawaru et
al., 2012).
CONCLUSION
Secondary
metabolites observed in the African mistletoe, Tapinanthus bangwensis and its hosts were similar; this gives
credence to the suggestion of movement of pharmacologically active compounds in
the host-parasite association and as such it is therefore logical to state that
there are intercellular exchanges of macromolecular substances between
mistletoe and its hosts. Increased accumulation of free amino acids as the
observed incidence in the infested hosts in comparison with the uninfested
implicates the activities of mistletoe in the breakdown of bound amino acids or
proteins in the notable variation of protein bands between the infested and
uninfested. This observed change in the protein and amino acids status of the
infested hosts is a part of plant response to stress which therefore implies
that the presence of Tapinanthus bangwensis on host plants imposes biotic stress on such host.
REFERENCES
Adesina S. K., Illoh H. C.,
Imoh I. J. and Imo E. J. (2013). African
Mistletoes (Loranthaceae); Ethnopharmacology, Chemistry and Medicinal Values:
An Update. Afr J Tradit Complement Altern Med. 10(3):161-170. http://dx.doi.org/10.4314/ ajtcam.v10i4.26
Adodo A. (2002). Nature Power: A Christian approach to Herbal Medicine.
D. M. Bosco Training Center, Akure, Nigeria. p. 207.
Adodo A. (2004). Nature power, A
Christian Approach to Herbal Medicine. Decency printers Ilorin, Nigeria,
pp. 103-110.
Azevedo Neto André
D., José T. Prisco and Enéas Gomes-Filho (2009). Changes in soluble amino-N,
soluble proteins and free amino acids in leaves and roots of salt-stressed
maize genotypes, Journal of Plant
Interactions, 4 (2): 137-144, DOI: 10.1080/17429140902866954.
Burkill H. M. (1995). The Useful Plants of West Tropical Africa. Edition
2. Vol. 3. Families J-L Royal Botanic Gardens. Kew. p. 857.
Bussing A. and Schietzel M. (1999). Apoptosis-inducing properties of Viscum
album L. extracts from different host trees, correlate with their content
of toxic mistletoe lectins. Anticancer
Res. 19(1A): 23-28.
David-Schwartz, R.,
Runo, S., Townsley, B., Machuka, J., and Sinha, N. (2008). Long-distance
transport of mRNA via parenchyma cells and phloem across the host–parasite
junction in Cuscuta. New Phytol. 179:
1133–1141.
Deeni Y. Y. and Sadiq N. M. (2002). Antimicrobial property and
phytochemical constituents of leaves of African mistletoe. J. Ethnopharmacol., 831: 235-240.
Earley E. B. and
DeTurk E. E. (1944). Time and rate of synthesis of phytin in corn grain during
the reproductive period. J. Am. Soc.
Agron. 36: 803 – 814.
Glatzel, G. and B. W. Geils. (2009).
Mistletoe ecophysiology: host–parasite Interactions. Botany 87:
10–15. Published by NRC Research Press.
Griffin Jason J.,
Thomas G. Ranney and D. Mason Pharr (2004). Heat and Drought Influence
Photosynthesis, Water Relations and Soluble Carbohydrate of Two Ecotypes Redbud
(Cercis canadensis). J. Amer. Soc. Hort. Sci. 129 (4): 497 –
502.
Guimaraes K. C., Kustar R.
M., Amaral A. C. F., Ferreira J. L. P. and Sinia A. C. (2007). Histological
study of the leaf and stem of Amazonian medicinal mistletoe Cladocolea micrantha (Loranthaceae). Int. J. Bot. 3(2): 218 -221.
Ibrahim J. A., Egharevba H. O., Iliya I., Tarfa F. and Ayodele A. E. (2014). Chemical Profile as
chemotaxonomic tools for Loranthaceae in Nigeria. African Journal of Plant Science, 8 (7): 343- 352. DOI:
10.5897/AJPS2014.1161.
Ichihashi Yasunori,
J. Musembi Mutuku, Satoko Yoshida and Ken Shirasu (2015). Transcriptomics
exposes the uniqueness of parasitic plants. Briefings
in Functional Genomics, 1–8 doi: 10.1093/bfgp/elv001
Ilesanmi Funmilayo Florence and T. l. Olawoye
(2011). A
preliminary comparative phytochemistry of metabolites of orange (Citrus
sinensis) and guava (Psidium guajava) mistletoes and their host
plants. Journal of Medicinal
Plants Research, 5 (3): 340-343.
Kaplan F., Kopka J.,
Haskell D. W., Zhao W., Schiller K. C., Gatzke N., ....... Guy C. L. (2004).
Exploring the temperature-stress metabolome of Arabidopsis. Plant Physiology 136: 4159–4168.
Kempa S., Krasensky
J., Dal Santo S., Kopka J., Jonak C. (2008). A central role of abscisic acid in
stress-regulated carbohydrate metabolism. PLoS
One 3, e3935.
Liu Li‐Xia,
Shou‐Mtn Xu , De‐Li Wang and K. C. Woo
(2008). Accumulation of pinitol and other soluble sugars in water‐stressed
phyllodes of tropical Acacia
auriculiformis in northern Australia, New
Zealand Journal of Botany, 46 (2): 119-126, DOI: 10.1080/00288250809509759.
Lugan R., Niogret M.
F, Leport L., Guegan J. P., Larher F. R., Savoure A, ......Bouchereau A.
(2010). Metabolome and water homeostasis
analysis of Thellungiella salsuginea
suggests that dehydration tolerance is a key response to osmotic stress in this
halophyte. The Plant Journal 64,
215–229.
Martinelli T.,
Whittaker A., Bochicchio A., Vazzana C., Suzuki A. and Masclaux-Daubresse C.
(2007). Amino acid pattern and glutamate metabolism during dehydration stress
in the ‘resurrection’ plant Sporobolus
stapfianus: a comparison between desiccation-sensitive and
desiccation-tolerant leaves. Journal of
Experimental Botany, 58 (11): 3037–3046. doi:10.1093/jxb/erm161
Mower, J. P.,
Stefanovi_C, S., Young, G. J., and Palmer, J. D. (2004). Plant genetics: Gene
transfer from parasitic to host plants. Nature
432: 165–166.
Murakeözy E. P.,
Smirnoff N., Nagy Z. and Tuba Z. (2002). Seasonal accumulation pattern of
pinitol and other carbohydrates in Limonium gmelinii subsp. hungarica.
Journal of Plant Physiology 159:
485-490.
Murugan K., Pradeep D. P., Meenu K. V. G., Aswathy
J. M., Greeshma M., Greeshma G. M.,.…. Lubaina A. S. (2014). Interaction
between Hemiparasitic- Dendrophthoe falcata (l.) Ettingsh. on Mangifera
Indica Linn. – Some Observations. World Journal of Pharmacy and
Pharmaceutical Sciences, 3
(6): 585-607.
Nayyar H. (2003).
Accumulation of osmolytes and osmotic adjustment in water-stressed wheat (Triticum
astivum) and maize (Zea mays) as affected by calcium and its
antagonists. Environmental and Experimental
Botany 50: 253-264.
Olapade E. O. (2002). The herbs for good health: the 50th Anniversary
Lecture of the University of Ibadan. NARL Specialist Clinic, Ibadan, Nigeria.
p. 230.
Osawaru, M. E., Ogwu, M. C., Chime, A.O. and
Amorighoye, A. R. (2012). Morphological evaluation and protein profiling of
three accessions of Nigerian Corchorus linn. Species. Bayero Journal of Pure and Applied Sciences, 5 (1): 26 – 32. http://dx.doi.org/10.4314/bajopas.v5i1.6
Polhill Roger and Delbert Wiens. (1998).
Mistletoes of Africa, The Royal
Botanic Gardens, Kew Ed. ISBN. pp. 370.
Preston A. L., An M. and Watson D. M. (2010). Chemical profile
differences in the endemic parasitic weeds: a study of host-parasite chemical
profile in select mistletoe and Eucalyptus
species. Seventeenth Australasian Weeds Conference.
Roney, J. K.,
Khatibi, P. A., and Westwood, J. H. (2007). Cross-species translocation of mRNA
from host plants into the parasitic plant dodder. Plant Physiol. 143: 1037–1043.
Rosen C. (1957).
Higher sugar content of extracts interferes with colorimetric determination of
free amino acids and free proline. Analyst
Biochem. 200: 115 – 118.
Roxburgh, L. and Nicolson, S. W. (2005). Patterns of host use in two
African mistletoes: the importance of mistletoe-host compatibility and avian
disperser behaviour. Functional Ecology 19: 865-873.
Sanchez D. H.,
Siahpoosh M. R., Roessner U., Udvardi M., Kopka J. (2008). Plant metabolomics
reveals conserved and divergent metabolic responses to salinity. Physiologia Plantarum 132, 209–219.
Simon-Sarkadi Livia,
Gábor Kocsy, Zoltán Sebestyén (2002). Effect of salt stress on free amino acid
and polyamine content in cereals. Acta
Biologica Szegediensis, 46 (3-4):73-75, Proceedings of the 7th Hungarian
Congress on Plant Physiology.
Snyder, M.A., Fineschi, B., Linhart,
Y.B., and Smith, R.H. (1996). Multivariate discrimination of host use by dwarf
mistletoe Arceuthobium vaginatum subsp. cryptopodum: Inter- and
intraspecific comparisons. J. Chem. Ecol.
22: 295–305. doi:10.1007/BF02055100.
Sofowora, A. (1982). Screening Plants
for Bioactive Agents. In: A. Sofowora, Medicinal
Plants and Traditional Medicine in Africa. Spectrum Books Limited. pp. 128
– 161.
Trease G. E., and Evans W. C. (1989). Pharmacognosy (13th edn).
Bailliere Tindall: London; 683-684.
Umoh U. F., Ekpo B. A. J., Bala D. N.,
Udobang J. A., Cocobassey M. and Etim E. I. (2011). Phytochemical
and comparative antidiabetic studies of leaf extracts of Viscum album from
different plant hosts. Int. J. Biol. Chem. Sci. 5 (4): 1448-1454. DOI:
http://dx.doi.org/10.4314/ijbcs.v5i4.11
Usadel B., Blasing O.
E., Gibon Y., Poree F., Hohne M., Gunter M., …… Stitt M. (2008). Multilevel
genomic analysis of the response of transcripts, enzyme activities and
metabolites in Arabidopsis rosettes to a progressive decrease of temperature in
the non-freezing range. Plant, Cell and
Environment 31: 518–547.
Wahab, O. M., Ayodele A. E. and Moody J. O. (2010). TLC phytochemical screening in some
Nigerian Loranthaceae. Journal of Pharmacognosy and Phytotherapy, 2(5): 64-70.
Yan, Z. (1993). Germination and seedling development
of two mistletoes, Amyema preissii and Lysiana exocarpi: host
specificity and mistletoe–host compatibility. Australian Journal of Ecology 18: 419–429.
Zuther E., Koehl K.
and Kopka J. (2007). Comparative metabolome analysis of the salt response in
breeding cultivars of rice. In: Jenks M. A., Hasegawa P. M., Jain S. M., eds: Advances in molecular breeding toward
drought and salt tolerant crops. Netherlands: Springer, 285–315.
|
Cite
this Article: Edagbo, DE; Oyetunji, OJ (2019). A Comparative Study of Secondary
Metabolites, Amino acids and Protein Profiles of the Host – Parasite Plants
in the Relationship between the African Mistletoe, Tapinanthus bangwensis
[Engl. and K. Krause] Danser and Two of its Host species. Greener
Journal of Biochemistry and Biotechnology, 6(1): 01-11, http://doi.org/10.15580/GJBB.2019.1.070719132.
|

