|
Greener Trends in Plant Pathology and Entomology
Vol. 3(1), pp. 01-17, 2020
ISSN: 2672-4510
Copyright ©2020, the copyright of this article is
retained by the author(s)
https://gjournals.org/GTPPE
|

|
Postharvest Spoilage and Management of Fruits and Vegetables: A
Perspective on Small-Holder Agricultural Systems of the Tropics
DN
Enyiukwu1; IN Bassey2; GA Nwaogu1; LA
Chukwu3; JO Maranzu4
1Department of Plant Health
Management, Michael Okpara University of Agriculture,
Umudike PMB 7267 Umuahia, Abia State. *Corresponding
author email:enyidave2003@ gmail.
com
2Department of Botany and
Ecological studies, University of Uyo, Ikpa Road Uyo, PMB 1017 Uyo, Akwa Ibom
State, Nigeria
3Department of Agricultural
Technology, Faculty of Agriculture, Akanu Ibiam Federal Polytechnic Uwana, Ebonyi State, Nigeria
4National Environmental
Standards Regulation and Enforcement Agency (NESREA), Owerri,
Imo State,
Nigeria
|
ARTICLE INFO
|
ABSTRACT
|
|
Article No.: 070519128
Type: Review
|
Fruits and vegetables are important components
of human diets all over the world. They are noted for their high moisture
contents, and for providing good measures of amino acids, minerals and vitamins
to their consumers. They are reported to afford their best benefits
especially when consumed fresh. However, damages incurred during harvesting
or handling, physiological changes and pathological attacks during storage
are known to reduce their shelf-lives, affect their nutrient quality and
possible health benefits. Herein we reviewed available literature on the
subject matter from 1997-2018; and information obtained from local growers
or grocers of agro-produce in southeast Nigeria..
Bacteria and fungi represent the highest numbers of incidents of
post-harvest microbial diseases on agro-produce which often times are
predisposed by abrasions due to poor harvesting and handling techniques
and/or poor storage facilities especially at village level farmsteads,
farm-families and marketing chains. Microbes-induced rots and deteriorations
usually begin around wound sites and are exacerbated by moisture and insect
migration into store houses. Between 10-50 % agricultural produce losses and
food wastages are reported to occur post-harvest. Losses are more severe in
the developing than developed economies and the warm humid agro-ecology
coupled with poor storage structures which characterize tropical
agricultural landscapes amongst other factors have been implicated for these
huge losses. Simple techniques of
curing harvested tubers, avoidance of wounds on produce, wrapping leafy
vegetables with old newspapers, avoiding direct sun rays on produce, not
packing fruits and vegetables tightly during shipment and storage; and
fumigating storehouses with bio-pesticides (tropical plant extracts) are
some of the post-harvest practices to prolong shelf-lives of
agro-commodities compatible with low-input agriculture. These low input
techniques of handling and storing agro-produce after harvesting are hereby
discussed.
|
|
Accepted: 08/07/2019
Published: 31/01/2020
|
|
*Corresponding Author
DN Enyiukwu
E-mail: enyidave2003@
gmail.com
|
|
Keywords: Postharvest
storage; rots and deteriorations; fruits; vegetables; agro-produce.
|
|
|
|
INTRODUCTION
1.1 Why
store agricultural produce?
Agricultural productions are seasonal, and require certain
time lags to produce crops. We store agricultural produce for use at a later
date usually during off-seasons or in places far removed from areas with
competitive advantage to produce such crops. However, in order to grow, develop
and build their own protoplasm, pathogenic microorganisms have been man’s
greatest rival competing with and depriving him of stored food calories and
nutrient-rich produce (Amadioha, 2012; Amadioha and Enyiukwu, 2019a).
Ensuring global food security hence is
one of the greatest challenges facing humanity especially in this 21st
century; and worldwide fungi and bacteria pose the largest number of crop
destroying pathogens both in the field and storage with fruits and vegetables
being the worst hit (Ajibade and Amusa,
2001; Meyer et al., 2016; Kasso and Bekele, 2018).
Phyto-pathogenic fungi, bacteria and oomycetes
are agents of rots and deterioration of stored agro-produce leading to
postharvest losses and wasted foods around the world (Enyiukwu
et al., 2014a; Kasso
and Bekele, 2018). Association of yams, cassava,
potato, wheat, cowpea, soybean, tomato and water melon for instance with Rhizopus spp., Mucor sp., Curvularia sp., Aspergillus sp., Erwinia sp.
and Fusarium sp. resulted in rots and storage
deterioration of these crops in Nigeria and around the world (Mahovic and Bartz, 2019;
Mohammed, 2013; Enyiukwu et al., 2014b). During pathogenesis, hyphae of these pathogens
elaborate from their spores, conidia or chlamydospores,
combining armies of enzymes and/or mechanical pressure to compromise and
overcome host cuticle and defense mechanisms; to colonize and ramify the
susceptible tissues (Amadioha 1994a, b).
In thus doing, these pathogens deprive the
host plant of calories and other vital growth factors and use same to build
their own protoplasm (Qi et al.,
2013; Markson et
al., 2014; Amadioha and Enyiukwu,
2019b). In Nigeria, postharvest studies conducted on Sweet, Irish and Hausa
potatoes as well as tomato and cowpea showed that losses of protein, lipids,
depletion of starch granules, and reduction of minerals such as calcium,
phosphorus, potassium, magnesium, zinc and iron attend these pathogenic attacks
on susceptible agro-produce in the country (Amadioha,
2004; Markson et
al., 2014 Nwaneri, 2017; Amadioha
and Enyiukwu, 2019a, b).
Because of the increasing human population the world
over, there is need therefore not only to increase food production or at least
sustain current production rate but more importantly to reduce to barest
minimum postharvest wastages at the current production scale. In developing
countries such as ours, crop yields are generally low due amongst other factors
to high on-farm pest and microbial disease attacks; with some of these diseases
continuing their ravages in stored produce. Postharvest food spoilages are
therefore tremendously threatening to food security (Madrid, 2011; Kumar and Bekele, 2016) and have been adjudged the greatest form of
losses encountered in farming concerns (Enyiukwu et al., 2014b). So far it is reported
that in the developing countries the bulk of research efforts on agriculture
(95 %) is directed at pre-harvest field studies whilst only few research
attempts (5 %) are geared at postharvest studies (Madrid, 2011)
1.2 Objectives of this study
Therefore, this article examined the postharvest
spoilages and management techniques of
fast perishing agricultural commodities (fruits and vegetables) in typical
smallholder agricultural systems of remote tropical locations with a view to
presenting compatible techniques and methods that can help enhance shelf-lives
of produce and hence food security and safety in rural settings.
2.0 METHODOLOGY
2.1 Data
generation and papers consideration
Apriori data
generated from searches on the subject matter from literature conducted in the databases
of ResearchGate, Google and Google Scholar based on
the methodology adopted by Mgbeahuruike et al. (2017; 2018) and Enyiukwu (2019) was used in this study. The search terms
included top-rate bacterial and fungal microbes of field crops, tunes of post-harvest
losses in cereals, tubers, fruits and vegetable; global and local estimates
of losses of post-harvest produce,
post-harvest microbial spoilage, storage forms of post-harvest produce,
post-harvest management of cereals, tubers, fruits and vegetable; in the humid
topics. Published papers and other
useful materials on some URLs from 1997-2018 were considered for inclusion for
review in this work. However, papers and other materials not written in English
or written in English prior to the period designated for consideration of
materials for review for this work were excluded from considerations. In
addition to these, information obtained from the interaction of the lead
researcher with 20 growers and marketers of grocers at the farm-gates and weekly
Ndoru and Ariam markets
respectively in Ikwuano Local Government Area (LGA)
of Abia state, Nigeria during the wet
(April-September) and dry (October-December) seasons of 2018 were also
incorporated into the review (Appendix 1).
3.0 DISCUSSION
3.1 Environmental
influences on storability of agro-produce
Temperature and relative humidity are the two most
important parameters that determine the storage life and produce quality (Tan,
2016). Agro-produce are living things which are
actively respiring and transpiring, losing water in the process. Proper control
of these two factors elongates the shelf-lives and quality of produce. By lowering storage temperature and
increasing relative humidity around postharvest produce, their rate of
respiration, water loss, ethylene production and microbial development on them
are reduced. Therefore temperature and humidity play active roles in the
attainment of safe moisture content for stored produce. Safe moisture content varies with
agro-produce and is attained when equilibrium is struck between the humidity
surrounding atmospheric air and the moisture content of the specific produce.
At the safe moisture content, the produce could store better and longer and the
danger of shrinkage, insect damage and attacks of noxious spoilage bacteria and
moulds becomes negligible (TSGC, 2019).
However, due to poor handling of agro-produce during
harvesting, they suffer abrasions and bruises. The consequence being increased
rate of respiration of the bruised produce leading to increased output of water
vapour. This condition is exacerbated by high ambient
temperatures which in concert with high relative humidity around stored produce
encourage microbial growth and development (Tan, 2016).. As a result of
increased rate of respiration and lowered tissue defense mechanisms of the
affected produce to microbial attacks, mouldiness and
spoilage begin to set in; and this could be worsened by surface moisture
accumulation on produce (TSGC, 2019). Control of postharvest agro-produce spoilage
and wastages usually involves the careful alteration of the physics of the
store atmosphere to seriously disadvantage or disfavour
development of spoilage-inducing microorganisms (Tan, 2016).
3.2 Qualitative
impacts of microbial attacks on agro-produce: A brief highlight
Besides quantitative deteriorations, qualitative losses
on produce such as anthracnose of banana, mango, guava, avocado and papaya on
the other hand, could render agro-produce unsightly and thus reduce their
marketable values (FRM, 2015). Some microbial pathogens associated with rots
and spoilage of agro-produce include Aspergillus spp., Penicillium spp., Botrydiplodia spp., Fusarium
spp., etc.; in some instances these
pathogens especially those of fungal origin produce toxins (mycotoxins)
such as ochratoxin A, aflatoxins,
T-2 toxins, H-2 toxin, vomiticin (DON), zearalenone, and ergot which contaminate agro-produce and
when such contaminated produce is consumed they interfere with functions of
enzymes and hormones, impede organs such as kidney, liver and the central
nervous system (CNS). Mycotoxins have been ascribed
as cause of various cancers, allergies, immune-dysfunction and even death in
humans (Shephard, 2008; Enyiukwu
et al., 2014b; 2018). In stored oily
products attacks from postharvest moulds alter their
taste making them rancid, of reduced value and poor marketability in
international trade due to their accumulation of free fatty acids (FFAs) (Enyiukwu et al.,
2014b). Insect infestations on the other hand, damage stored produce and
increase their respiration rate, creating hotspots and building up
moisture-spots in stores which in the end add to and make worse pathogenic
attacks and deterioration of stored agro-produce (www.ento.psu.edu/extension/t..).
Given the enormous nature of microbes-induced postharvest
agro-losses and their effects, the pertinent question now therefore is how do
we consciously checkmate losses due to microbial attacks on stored produce and
reduce wasted foods? Two scientific principles to this effect have been
advanced which involve to:
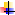 Kill the spoilage causing moulds
out-rightly or
Kill the spoilage causing moulds
out-rightly or
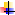 Actively suppress their activity.
Actively suppress their activity.
The later could be done through altering the environment
of storage to disfavour the metabolism of the moulds and bacteria; and this could be achieved through:
o Low temperature storage involving refrigeration of
agro-produce at 11-14oC where costs permit (Tan, 2016).
o Reduction of moisture content of produce to some safe levels
through sun or air-drying so that the produce keeps well for 1 week to up to 1 year (www.knowledgebank.irri,org/n; TSGC, 2019).
o Addition of preservatives which are synthetic or
chemicals of plant origin that ward-off microbes and/or prevent their
proliferation and eventual spoilage of agro-produce. However care must be taken
in adopting this method since some of these substances are potentially
hazardous to human health. Plant protection experts or their trained extension
agents must be involved in training farmers in this regard (Enyiukwu
et al., 2014a, b, c).
o Processing agro-produce quickly into other products or
food-forms in the crop’s value chain that have inherent ability to store better
and longer (www.ug.edu.gh/iast/node/5).
3.3 Quantitative
Post-harvest microbial deteriorations: A grouping
Spoilage refers to deterioration in agricultural produce which
makes it to lose nutritional value, taste bad, rotten or mouldy
and thus becomes a carrier of potentially hazardous and disease causing biota
or toxins. It may also refer to a loss in quality, vigour,
colour and consistency of agro-produce due to pathogenic
or non pathogenic factors (Garcha,
2018). Other factors which predispose or constitute spoilage of agro-produce
include physical damages during harvesting, threshing, storage or shipment,
physiological damage such as sprouting and pathological deteriorations and
contaminations with microbial toxins (FFTC, 2018; Enyiukwu
et al., 2018):
3.3.1
Physical damage of ago-produce during threshing, storage
and transit
Physical damage could cause or contribute considerably to
postharvest losses in stored produce or predispose them to enhanced
physiological degradation. Physical damage may result from rough harvesting, or
rough threshing of grains or due to poor handling of produce in storage,
transit or value chain lines. It may occur as well from impacts of extremes of
temperature on produce. For example temperatures below 12oC cause
chilling injury in tubers whereas temperature range of 30-45oC may
cause black heart characterized by discolouration at
the center of the tuber (Fig. 1) and mahogany browning in potato due to
asphyxiation from too low oxygen or too high carbon dioxide (CO2)
tension in the soil or store environment. In some cases it may occur in tubers
growing in waterlogged areas for the same reasons (Ephytia,
2019).

Figure 1: Black heart of Irish potato
Source: www.ephytia.inra.fr/en/G/21136/Potato-Black-heart
3.3.2
Physiological damage of agro-produce during storage
and transit
This may be as a result of metabolic losses in the form of
wilting or respiration. It may also be due to sprouting particularly in tubers
such as yam. In any case sprouting reduces the marketability of produce.
Cassava for example suffers from serious postharvest physiological
deterioration (PPD) commonly known as vascular streaking which occurs some
hours (within 24-48 h) after harvesting and can result in loss of 40-60 % total
expected economic value of the tubers. This internal blue-black discoloration
usually starts at sites of damage especially around the central vascular
bundles near to sites of damage of the root tuber and spreads to the adjacent storage parenchyma, and subsequently
making the starch stored therein to undergo structural changes (Fig. 2); and
thus unpalatable and unmarketable (Njoku, 2009)..
Other forms of physiological changes in quality may occur frequently in all
root crops in storage due to changes in starch-sugar equilibrium. In general
low temperatures increase the sugar content of tubers leading to sweetening and
may tend also to increase the rate of sprouting. Though sweetening is welcome
in sweet potatoes or yams, in most other tubers it is reported to be
undesirable.

Figure 2: Longitudinal section of cassava tuber showing vascular
streaking
Photo: DN Enyiukwu
3.3.3
Pathological deteriorations of agro-produce during
storage and transit
The most serious cause of postharvest losses in
agro-produce is probably pathogenic attacks though however, they may be
predisposed by physical or physiological damages (Okigbo,
2004; kumar and Kalita,
2017; Kasso and Bekele,
2018). Most microbes that participate in postharvest losses are low grade,
wound requiring organisms that attack produce at sites of injury incurred
during harvesting, post-harvest handling, transit or storage especially
vegetables, fruits and tubers (Fig. 3). Less frequently though, the pattern of
attack may also involve pre-harvest systemic infection which blossoms during
protracted storage especially where there are uncured postharvest wounds and
abrasions in tubers (Okigbo, 2003; 2004; Enyiukwu et al.,
2014b). In most cases attack leads to
fermentation, tuber softening and discharge of offensive odour,
hardening or making its matrix brittle (Amadioha and Markson, 2007; Njoku, 2009; Markson et al.,
2014; Nwaner, 2017).
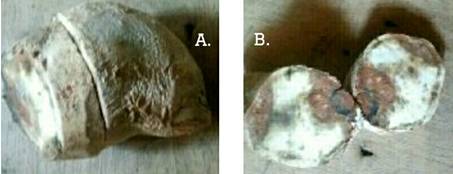

Figure 3: Microbes-rotted sweet potato tuber (A) and its transverse sections (B) and transverse section of microbes rotted cassava tuber (C) Photo:
DN Enyiukwu
3.4 Financial
estimates of microbes-induced postharvest produce losses in crops
Fruits and vegetables represent the most perishable
agricultural commodities and in addition to decimations suffered from pests and
diseases attacks in the field; postharvest losses are reported to be very
severe in these classes of produce especially in the warm humid tropics owing
to lack of adequate storage facilities and poor produce handling techniques (Salau and Shehu, 2015; Kumar and Kalita, 2017; Kasso and Bekele, 2018). Globally, besides the possibility of total
crop failure (100%) in severe cases due to plant diseases occasioned by
microbial attacks in the field, growers suffer tremendous postharvest losses
ranging from 5-70 % of actual yield of several agricultural crops (Madrid, 2011;
Begum et al., 2008; 2013).
In fruits and vegetables losses up to 25 % and 40 %
respectively have been documented (Olayemi et al., 2012; Kasso
and Bekele, 2018). In most Asian countries
postharvest losses of agricultural produce are comparatively high; ranging from
10-50 %; peaking in high-moisture containing produce such as fruits and
vegetables. In India losses of 20-35 % of tomato produced in that country
occurred postharvest. Out of the 17 million MT of vegetables produced in Japan
in 1991, 10 % was reportedly lost during postharvest handling and distribution
(Madrid, 2011; FFTC, 2019). FFTC (2019) further noted that as much as 32 % loss
of Chinese cabbage and radish both major constituents of Korean diets are lost
postharvest per annum in both Japan and Taiwan. Also, in Thailand losses due to
fruit rot of 60 % for mango, tomato and cabbage; 50 % for head lettuce and
cauliflower; 30 % for bell flower and 17 % for Chinese cabbage have been
reported (FFTC, 2019). The USA, Canada, New Zealand and Australia are not left
out, in these economies on the average, 52 % fruits and vegetables and 38 %
grains among other commodities are lost or wasted every year from farm-gates to
dining tables (Fig. 4).
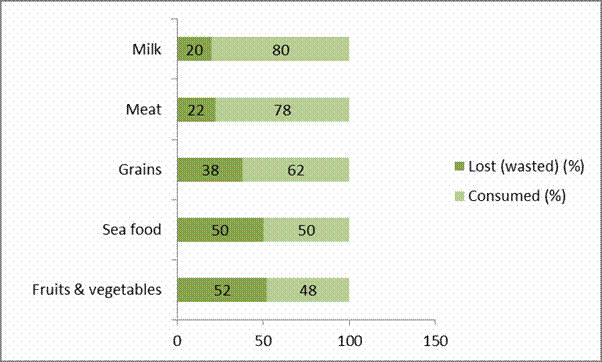
Figure 4: Estimates of foods spoilt, lost or wasted annually in
USA, Canada, Australia and New Land
Source: Plumer (2012)
Diverse ranges of agricultural produce including corms,
tubers and fish are reported to be lost or wasted after harvest on annual basis
in SSA (Oluyemi et
al., 2012; Enyiukwu et al., 2014b). Surveys conducted in Nigeria on postharvest
wastages in yam, cassava, maize, plantain and vegetables by Oluyemi
et al. (2012) revealed loss profiles
of 33 %, 27 %, 20.33 %, 27.0 % and 33.0 % respectively. A similar study in Sokoto State, Nigeria by Salau
and Shehu (2015) showed that 43 % of vegetables
produces in the State were lost to microbial maceration and degradation
encouraged by poor harvesting, handling and storage of produce. Out of the 134
million Mt of sweet potato produced around the world, 34 million MT were
reportedly estimated to be lost to postharvest spoilages. Yam another important
tuber crop in tropical Africa according to this source recorded a loss profile
of 5 million Mt out of a total global production figure of 19 million MT (Enyiukwu et al.,
2014b).
Estimates by the Imperial College London (2012) suggest
that each year fungal diseases alone destroy not less than 125 million MT of
protein-calorie providing food crops such as rice, wheat, maize, potatoes and
soybean; and thus dispossess 600 million people around the world of valuable
sources of energy and growth factors. In
three (3) cereal crops (rice, wheat and maize) alone global agriculture is
reported to lose not less than USD 60 billion to fungal diseases on annual
basis (Imperial College, 2012). Similarly, bacterial diseases are not left out
in the hunt to dispossess men of food and their economic investment in
agriculture. For instance, R. solanacearum is an extremely damaging pathogen having
wide geographic distribution and host range. Estimates suggest that USD 1
billion is lost each year around the world due to attacks by the pathogen complex in
susceptible crops (Manifield et al., 2012).
Worldwide statistics in money terms, for postharvest
losses in fruits and vegetables reveal that growers of horticultural crops lose
not less than USD 625 million per annum (Pandy and Pandy, 2012). In
Latin America alone, it is estimated that microbes-induced postharvest losses
of major crops totaled about 38 million MT per annum. Estimates further showed
that postharvest losses of agricultural produce in India and Latin America
amounted to USD 66 million per year. In the EU countries, the postharvest
losses in fruits and vegetables have been estimated at a whooping
4.0 billion EUR every year (Madrid, 2011). In the USA, a report at the
Washington Post by Plumer (2012) noted that the
country loses, wastes or spoils fruits and vegetables, sea foods, grains and
other commodities estimated at USD 165 billion every year due in part to
postharvest handling practices and high aesthetic standards or requirements for
foods especially fruits and vegetables. Africa south of the Sahara with its
high ambient temperatures and relative humidity coupled with poor storage and
transit systems is not left out of huge post-harvest food wastages, Total
post-harvest losses in perishable agro-produce ranging 20-40 % estimated to
amount to about 4 billion dollars loss of revenue to growers has been
documented in the sub-region (Hailu and Derberi, 2015).
The possibility of these postharvest losses and wasted
foods increasing or worsening in the foreseeable future is high especially in
southern Asia and tropical sub-Saharan Africa (Sadiku
and Sadiku, 2011; Enyiukwu et al., 2014b). Experts suggest that
these losses occurred largely because of inappropriate storage at traditional
village level storage systems, poor produce handling and marketing
infrastructure (Hailu and Derberi,
2015; FFTC, 2019). The reason for this is to a large extent, due to microbial
spoilage or deteriorations of moisture-rich fresh agro-produce stored in these
systems in locations characterized by high environmental temperatures and
humidity; which without doubt have been made worse by climate change effects from
heightened values of these parameters
(Salami and Salami, 2011; Enyiukwu et al., 2014b, c; FFTC, 2019). As a way
of assessing impacts of low-input storage materials in a trial, replacing
conventional traditional bamboo baskets commonly used in packaging in some
Asian settings with improved rubber crates was found to significantly reduce
postharvest spoilage and wastage of head lettuce during shipment from Chang Mai
to Bangkok from 35.8 % to 12.6 % (FFTC, 2019).
4.0
Postharvest storage and management of loss-prone
agricultural produce in the tropics
4.1
General management of physical damages
Ensure you avoid poorly drained or water-logged soils
when planting tuber crops or during tuber crops development (Mahovic and Bartz, 2019). Make sure you do not harvest potato tubers
during very hot weather (over 30oC), and ensure you shade-dry them
(other tubers inclusive) quickly to avoid moisture condensation on them. It is
recommended that during transit or storage adequate ventilation of tubers
should be maintained. Also, tubers should not be packed too tightly in enclosed
containers during shipment so as to avoid abrasions (Ephytia,
2019). In fruits such as tomato,
eggplant and mango, do not pack ripe fruits in enclosed containers and ensure
harvesting and storage only in cool dry weather (Personal communicatioms,
2018).
4.2 General management of Postharvest
Physiological Diseases (PPDs)
In High-tech societies, vacuum packaging is used to delay
onset of PPDs, however, this is impractical given our peculiar circumstances.
Conventional breeding or even biotechnology-assisted breeding is used to breed
varieties whose roots store longer without developing vascular streaking (Njoku, 2009). In smallholder farming systems, it is advisable
not to harvest cassava right after a heavy rain (Njoku,
2009). Harvest only what is needed for
food or processing, ensuring you avoid wounding the cassava roots and store the
harvested roots in cool dry well ventilated places preferably under tree shade
or underground trenches with the tubers still attached to their stems (Personal
communication, 2018; Magsasaka, 2016).
4.2
General management of pathological diseases
Several management options are open for use by growers
against microbial deteriorations in agricultural produce. These range from good
agronomic practices, chemical control, applying botanical measures and using
bio-agents to manipulate storage environments to disfavor pathogenic infection,
growth and development. These approaches have been well reviewed by Enyiukwu et al.
(2014a).
5.0
Specific management of postharvest microbes
induced diseases of stored produce
5.1
Fruits and vegetables
5.1.1 Rhizopus soft rot
of tomato
Vegetables are plant organs high in moisture content and
consist of succulent tissues which may be eaten raw or cooked as part of the
main dish. Their nutritional profiles are high supplying essential vitamins and
minerals in diets (Afolabi et al., 2012). Because of their high content of moisture,
vegetables are very highly perishable, and hence do not keep well in store
after harvest. In fact it is estimated that one-third (1/3) of all perishable
agro-produce is lost or wasted between farm-gates and usage at kitchens (www.msugreenalliance.wordpress.com/2017...).
Many vegetables including tomato are attacked by Rhizopus soft rot
caused by the fungus Rhizopus stolonifer in
stores, transit or market. The fungus is necrotrophic.
It secretes pectolytic enzymes which breakdown the
cellulose cell wall of the mature cells of the fruit tissues, invades and kills
them first before feeding on them. Infected host tissues become discoloured and liquefied (Nelson, 2009). Mycelia of the
fungus can be seen on the fruit a few days on the fruit post-infection even
without a hand lens. The organism requires an optimum temperature of 25oC
and relative humidity (RH) range of 75-85% to unleash attacks on susceptible
tomato varieties. Generally, the organism is a wound requiring parasite and
usually overwinters unfavourable
conditions as zygospores that may survive for many
months. The intensity of attacks of the pathogen on tomato fruits varies with
cultivar of the crop, temperature, relative humidity, amount of inoculum present and
the amount of wounding at harvest, threshing or storage. Usually losses are
greater in a few days and decreases with time (Mahovic
and Bartz, 2019).
Management of Rhizopus soft rot:
It is pertinent to note that resistant varieties to this
disease and several other post-harvest fungi or bacteria-induced diseases of
vegetables thus far are unavailable (Mahovic and Bartz, 2019). Generally, in managing fungal or bacterial soft
rot, it is essential you avoid wounding the fleshy fruit of tomato during
harvesting, handling, transit or marketing (Nelson, 2009). In the event this
happened, the wounded fruits should be discarded or ensure that they are
consumed immediately or better still be processed by dehydration. Do not
harvest tomato fruits in wet weather, because dampness of fruit will encourage
susceptibility to fungal rot pathogens and development of soft rots (Mahovic and Bartz, 2019).
In urban or sub-urban settings, sound fruits should be
stored in storage facilities free of crevices, roof-leaks, debris, rodents and
insects. Disinfect all such facilities to ensure freedom from insects and pest
before storage and after sale of all existing stocks of produce. Ensure also
that no weight is placed over the fruits during transit or storage. Before storage,
wrap fruits with paper impregnated with specific synthetic or botanical
fungicides such as Botran 75W (Dicloran
75W). Lastly, do not store fruits for a very long time (Mahovic
and Bartz, 2019) especially when narrow spectrum
fungicides with low residual activity is used as preservative. Make sure that
such tomato fruits are converted to other longer storing forms in the crop’s
value chains.
In local farm-settings store only sound fruits in bamboo
baskets impregnated with Tectonia
or banana leaves to cushion the fruits against shock, prevent abrasions and
absorb undue moisture (Personal communication, 2019). In smallholder households
the ground or whole fruits dehydrated by boiling and preserved with few pints
of vegetable oil store well at room temperature for up to 1-2 month (Personal
communication, 2019).
5.1.2 Storage
spoilage of onion bulbs
Sprouting is one of the major constraints limiting
storage life of onion bulbs. After prolonged storage periods mature onion bulbs
begin to sprout either during transit, marketing or in store. This generally,
reduces the market acceptability and value of the bulbs. Traditionally, onions
are stored in simple structures, insulated from undue temperature and humidity
build-up through maximum allowance of air circulation. This is achieved by
keeping the depth of the pile at minimum, orienting the store house to allow
maximum passage of prevailing wind and stacking the bulbs on a mesh of wood or
cane with an air space below or in jute bags. The maximum length of storage
depends on the agronomic practices adopted by the farmer in the field,
cultivar, store condition, and bulbs quality before storage (Wani and Taskeen-un-Nisa, 2011; Nischnitz et al., 2013).
Longer storing cultivars are generally characterized by
high dry matter contents, high refractive index, low water loss rate, strong
pungency and hence less liable to microbes incited rots. Onions can be stored
at relative humidity of 70-80% and 0oC for 8-9 months. Higher
temperatures progressively decrease the shelf-life of this valuable spice. At
room temperature in the tropics most cultivars desiccate highly and rot
uneconomically. Attacks by insects and mites on bulbs seem exclusive to fields,
however, the nematode Ditylenchus dipsaci which
has infected the crop in the field could continue systemic degradation of
stored bulbs causing them to be puffy and light weight. Fungi such as Alternaria spp., Sclerotium spp,
Fusarium spp., Rhizopus spp, Pernorospora spp., Botrytis
spp. and Colletotrichum
spp. among others have been found to be associated with onion rots (Awurum et al.,
2016). Neck rot caused by B. allii and B. aclada is commonly recognized as the most widely
distributed storage disease pathogen of onion bulbs. Infection occurs in the
field when the crop blooms and is carried over to the stores. The fungus causes
softening of the scales which take on water soaked appearance. Neck rot prevail
severely when moist conditions precedes harvest and while onion is being dried
in the field. Excessive late applications of nitrogen fertilizers and
irrigation (i.e. post bulb initiation) have been reported to exacerbate the
disease (Nischnitz et al., 2013; Awurum et al., 2016)
Massive storage losses in onion have been reported in
many countries including India. Aspergillus niger
has been documented as the cause of black mould rot
of stored onions. The fungus is widespread in distribution in onion growing
locations, and has been reported as one of the most destructrive
diseases of the crop in storage (Wani and Taskeen-un-Nisa, 2011). Evidence
of its presence is huge masses of black powdery spores on both the exterior and
between bulb scales of infected samples. High N-fertilizers predisposes the
crop to the disease. Downy mildew caused
by Perenospora destructor is also associated with the
rot of onions in storage. The disease is systemic and infected bulbs are soft
and shriveled or may sprout prematurely (Awurum et al., 2016). Basal stem rot on the
other hand is a wound requiring disease of onion caused by F. oxysporum f.sp cepae. This fungus is soil-borne, and the attendant
disease on the crop is exacerbated by high storage temperature and relative
humidity. Similarly Sclerotium
cepivorum
another soil-borne disease of the crop is systemic and late infection leads to high
storage decays in the crop (Awurum et al., 2016). Bacterial pathogens such
as Erwinia carotovora ssp carotovora, Pseudomonas cepacia and Bacillus coagulans are also reported to
cause varying degrees of rot of the produce in the store (Nischwitz
et al., 2013; TAMU, 2019; PNW Plant
Disease, 2019).
Managenent of onion
rot
Seeds or bulbs should be dressed with fungicides such as benomyl (1 g/kg seed) before sowing. In the field, ensure
that pre-mature onion bulbs are sprayed with maleic hydrazide
(MH-36% a.i) 5.5 liters in 553-675 liters of water
per ha to suppress untimely sprouting of matured bulbs during storage. Also,
ensure that bulbs are well matured before harvesting. Do not harvest mature
crops during and immediately after rains. Ensure that the bulbs do not suffer
bruises, wounds or injuries during harvesting as these could become portals of
entry to soil-dwelling rot causing bacteria and fungi such as F. oxysporum
which is common in most tropical agricultural soils.
Air-dry bulbs quickly after harvest to reduce their
moisture content (Nischwitz et al., 2013). Warehouses should be fumigated to rid them of
insects with sprays of neem kernel oil and their
roofs properly mended to prevent leaks which wiould
increase humidity and hence build up of pathogenic
fungi (Enyiukwu et
al., 2014a).
Where artificial ventilation is the order in large scale
production systems stores, store bulbs in jute bags and where not store in
local bamboo baskets or cartoned floors; and don’t
stack them above 3 meters (Nischwitz et al., 2013). Where possible in large
scale onion production systems, spray stored bulbs with botanical fungicides such as neem oil or synthetic ones such as carbendazim,
mancozeb, hexaconazole and captan (Wani and Taskeen-un-Nisa, 2011). In
smallholder farm unit stored bulbs should be admixed with lime or lemon fruits
to ward off microbial degradation (Personal communication, 2018)
5.1.3 Bacterial
soft rot of carrot
Soft rot is one of the most destructive diseases of
carrot (root crop) and other vegetables during transit and storage. It leads to
economically important losses in the form of huge rots and decreased product
vitality; as well as reduced product marketability (Green Life, 2019). In
susceptible varieties, the disease occurs as characteristic soft decays of the
fleshy vegetable. It begins as small water-soaked spots which enlarge rapidly.
Affected tissues become mushy, cream coloured and
leak water. Soft rot is caused by a non-spore forming Gram negative bacilli
called Erwinia carotovora pv carotovora (Green Life, 2019). The
organism grows well at a wide range of temperatures but thrives better at
optimum ambient temperature value of 25oC. The pathogen has wide host range affecting
potatoes, onions, tomatoes, cucumbers and other fleshy vegetables
(www.gadeningknowhow.com/soft_rot.....). It is a
cosmopolitan organism surviving in a wide range of soils and aquatic habitats.
In fields, rogue and destroy affected plants by burning since the disease is
difficult to control. Post harvest, the pathogen
usually invades bruises on the crop, and secretes pectolytic
enzymes with which it attacks and degrades cementing substances of the middle
lamella, cell walls and then being assisted by other enzymes the cell contents.
Erwinia carotovora pv carotovora
overwinters unfavourable conditions on the crop
debris, seeds, and in soils. Harvesting bruises, freeze (or chill) injury, and wounds by insect increase the chances of attacks
of the disease on susceptible cultivars of carrot. Abundant moisture at wound
sites and high atmospheric humidity also encourage the development of the
disease. In any case damaging rots ensue following attacks by the bacterium
which weather and store condition could exacerbate (Nischwitz
et al., 2013).
Management: Carrot is the enlarged tap root of the crop which we eat.
Ensure that the flesh surfaces of the produce are dry and free from moisture.
Also avoid bruising them when harvesting, transplanting and storing carrots.
Maintain good air flow in storage areas and store houses. In high-tech farms;
cool carrots to 4-6oC on arrival from shipment before storing. Never
store wet, wounded or decaying carrots. Ensure that you store at optimal
relative humidity, and keep stores clean, leaks and insect free (Green Life,
2019). In low input farming systems store carrots in bamboo baskets impregnated
with dried absorbent plant leaves (Personal Communication, 2018)
5.1.4 Leafy
and other vegetables
Telferia occidentalis (Fluted
pumpkin) and Amaranthus
esculentus (Amaranth) are choice leafy vegetables
in West Africa especially Nigeria (Afolabi et al., 2012). Fluted pumkin is an important fast growing indigenous medicinal
and nutrient rich seed and leafy vegetable in very high demand in Nigeria (Odiaka et al.,
2013). In addition to spicing up the kitchen, its phosphorus and protein-rich
seeds are widely used in ethno-medicine as tonics, to protect against
cardio-vascular diseases; for treating anaemia,
convulsions and malaria (Ajuru and Nmom, 2017). Seeds of the crop furthermore are used in
boosting sexual libido and as panacea against low sperm counts in human males (Munaya, 2016). Also, nursing mothers consume the seeds for
its lactation-inducing properties. While the seeds oil is reported to possess
potential activity against kidney or badder stone
disease and have ameliorated quinine-induced testicular damage in a trial (Akang et al..,
2010).
Harvesting of leaves of the crop is done at maturity when
the leaves are luxuriant while fruits are deferred till the end of the cropping
season (November-December). Leaf harvesting should be done in cool weather
either early in the morning or late in the evening when the sun has set.
Harvested vegetables should be kept under shades away from scorching sunlight
(Personal communication, 2018). After harvesting, the produce should be graded
and tied into bunches with ropes, and wrapped with jute bags in the case of
amaranths. Shipment to points of sale
must be as quick as possible as dehydration reduces the marketability of the produce.
At points of sale these leaf vegetables should be sprinkled with water
periodically to keep them moist and remain maximally attractive. Nevertheless,
fresh vegetables may be contaminated with rot causing organisms from soil or
water-borne fecal matter or manure between the farmstead and points of their
sale. Recent surveys conducted at points of sale of vegetables in Iran showed
contamination of fresh and minimally processed vegetables by species of Fusaium, Geotrichum, Cladosporium, yeast and salmonella (Jeddi et al., 2014). Leafy vegetables therefore should not be packed so
tightly in storage especially when damp to avoid pathological decays. However,
produce do not keep well more than 4-6 days with good attention without cool
storage. Vegetables for salad may be preserved with brine solution or vinegar.
Generally leafy vegetables should be unwrapped and spread on thatch roof or table tops every night to allow dew to
fall and keep them fresh and humid (Personal communication). Association of T. occidentalis
with R. stolonifer,
Botrydiplodia theobromae, A. niger and Erwinia carotovora ssp
carotovora
leading to its fruit rot has been reported with virulence highest in R. stolonifer
and least in A. niger.
(Nwufo and Emebiri,
1990). These workers attributed 95 % incidence of the rots to the fungal
agents and 5% to the bacterial agent. These agents are likely responsible for
the decay of its leaves in damp conditions. Seed protein, carbohydrate and
lipid contents reduction of 48-61.0 %, 63-79.0 % and 60.3-71.5 % by these
fungal agents has been documented (Nwufo and Emebiri, 1990).
Ptericarpus osun popularly called Uha in eastern Nigeria is a well appreciated potherb. It
suffers dehydration and wilting when exposed in the air for a long time after
harvesting. The leafy vegetable should be kept dry and wrapped with printing
papers or old newspapers before storing in a refrigerator where available in
which form it can keep well for about 7-14 days. Alternatively, the fresh
leaves of the plant should be stored in open bamboo baskets overlaid with old
newspapers under tree shades. Damp leaves of the crop are easily attacked by
decay causing microbes, therefore avoid storing the produce when damp (Personal
communication).
Biter leaf (Vernonia cordata, V. amygdalina) is another widely consumed
potherb especially in eastern Nigeria. fresh leaves
are washed and squeezes in warm water and the resulting minimally processed
leaves are soaked in fresh water stored with the water changed each morning. In
othe instances, the leaves are first minimally by sun
or air dried and then wrapped with old newspaper and stored in cool dry places
such as under trees or thatched roofs (Personal communication).
Plantains and bananas are well grown and eaten in Nigeria being common in
western and eastern parts of the country. In agronomic terms, they may be
regarded as fruits or vegetables depending on variety or use to which they are
put. Plantains and bananas should be harvested at three-fourth maturity and stored
in cool dry places to allow for gradual bunch by bunch ripening. During
shipment, bunches should never be packed tightly, however, they should be
packed with dried banana leaves sandwiched between bunches. Field infection of
bananas with anthracnose (Colletotrichum spp.)
makes the produce unsightly and should be controlled by sprays of benomyl or neem kernel oil at
3-monthly intervals (Enyiukwu et al., 2014a). Bunches should never be stored in ethylene laden
environments as this could facilitate ripening and encourage attacks by rot
inciting microbes. If possible store plantains and banana along with plenty of lime or lemon fruits
to forestall unwarranted microbial decays (Personal communication, 2017).
Dacryodes edulis (Afrcan Pear, Butter fruit, Safou
pear)
African pear commonly called Ube in Igbo is a highly
cherished fruit in southern Nigeria. It is seasonal and is commonly on sale
between March and June. Mature fruit turns from pink to blue-black in colour when ripe; and is usually eaten boiled or roasted
with roasted corn. Harvesting is done using go-to-hell or hooks made from tree
forks either early in the morning or late in the evening. The fruit is reported
to be highly perishable storing less than 1 week post-harvesting in the warm
humid tropics especially when harvested in wet weather (NAP, 2008). Rot in the
crop is reported to be quite huge, being triggered or worsened by wounds
incurred due to poor pre- and postharvest handling, poor storage and shipment
practices or conditions (Hai, 2019). In fact losses,
spoilages and wastages amounting up to 50 % have been document in the African (safou) tree fruits
(www.worldwidefruits.com/dacryodes-edulis-safou-tree.html, 2019). Fungi and
bacteria namely B. theobromae, R. stolonifer, A. niger and Erwinia spp. were
implicated in the rots of the crop with the first two organisms actively
virulent and destructive accounting for
80 % of the rots in most cases (NAP 2008; Hai, 2019)
.
Management
Harvest African pear fruits only in dry weather
(www.worldwidefruits.com/dacryodes-edulis-safou-tree.html, 2019). Like other
perishable agro-produce, avoid wounding the fruits during harvesting. Ensure
you store them in local bamboo baskets without lids or jute bags, in cool airy
and moist-free places (Hai, 2019). Never store the
fruits in hermitic containers and do not store them densely. Dampness on fruits
or moisture migration into store-houses should be strongly avoided as these
trigger unwarranted softening and mould deterioration
of the fruits (NAP, 2008). Never stack baskets one upon another so as not to
pressure the fruits to rots and decays. Introduce few fruits of lime fruit into
the storage baskets and cover the tops of baskets with dry moist-free leaves
and twigs of bitter leaf (Vernonia cordata L.) before storing (Personal communication,
2018).
Canarium shweinfurthi (African elemi, canarium) is
widely distributed in the rain forest-guinea savana
belt from Senegal, Ghana, Nigeia,
Cameroon etc. The fruit is commonly called Ube
osa, Ube nwanunu or Gbaruruo in
Igbo speaking region of Nigeria.. Mature fruit of the
crop is blue-black in colour in much the same way as
African Pear, however it is smaller and harder than the African pear and unlike
it, requires longer soaking in hot water (2-3 days) to soften (Okpala, 2016). It is rich in many kinds of fatty acids such
as palmitic, stearic. oleic
and linoleic acids. Resins from the
plant provide local candles for natives while the char is used in making ink.
Various parts of the plant find applications in a plethora of ethnobotanical uses (Okpala,
2016). The fruit is harvested by picking also, stored damp-free under tree
shades away from scorchimg sunlight and shipped in
local bamboo baskets padded with dried plantain leaves or jute bags (Personal
communication, 2019).
Chrysophyllum albidum (African star apple) commonly called Udara in igbo is another well cherished fruit making debuts
about the onset of farming seasons. The fruit which is somewhat heart shaped is
golden yellow in colour, and contains about 4-5 seeds.
Harvesting is done by picking of mature fruits from the tree. Post-harvest
losses in the crop are huge too due to poor handling and post-harvest microbial
deteriorations. Though cool storage at 10oC makes the fruit keep
well for a long time without spoilage (Nwufo et al., 2002); but in village level
farmsteads the fruits are stored in un-lided bamboo
baskets and kept under cool tree shades (Personal communication, 2018).
Persea americana (Avocado pear, Alligator pear)
This fruit is commonly called Ube bekee in Igbo speaking areas of
southern Nigeria. They vary in shape
from round to pear; and colours from green in most
cases to dark brown (FM, 2015; Gunmars, 2018).
Because of its high nutrient density, avocadoes are termed superfoods.
One hundred grams of the fruit is reported to contain protein (2 g), fat (15
g), calories (160 g) and about 20 vitamins and minerals including vitamins k
(26 %). C (17 %), E (10 %), B5 (14 %), B6
(13 %), folic acid (20 %) and K (14 %).
Mature fruits are harvested by picking just like the African pear; like
many other fruits it does not store for a long time before rotting, being
attacked by Botryotinia
(Botrytis) spp., Colletotrichum spp. and Aspergillus spp.
(Gunnars, 2018).
In most cases these organisms affect the fruits from the field while
expressing symptoms in the store. Anthracnose caused by Colletotrichum spp. attack a variety of crops and
agro-produce in the field and store (Awurum and Enyiukwu, 2013). C. gloeosporioides in particular causes unsightly necrotic
lesions of the avocado fruit which leads to pulp blackening and rot to the
stone of the fruit. This organism attacks mango and papaya leading to serious
fruit blemishes. Alongside other rot organisms, up to 70 % postharvest losses
or wastages have been attributed to this fungus in mangoes (Madrid, 2011). Also
stem rot caused by B. theobromae
is another serious spoilage disease which affects avocado fruits as small pale
brown necrotic lesions which rot the fruit pulp being exacerbated by injury and
damp conditions (FRM, 2015).
Management
Do not harvest avocadoes during the rains and avoid moist
soils sticking on the fruits. Picked fruits should be kept under tree shades
away from direct sunlight. Avoid bruises on fruits during harvesting and down
the value chain lines as these pose portals of entry to rot inciting microbes.
Ensure that store houses are well ventilated and leak-proof; and store only
damp-free fruits in local baskets overlaid with dried banana leaves to cushion
shocks during shipment and reduce the effects of rot causing agents (Kebede and Bailey, 2019). Generally, post-harvest
management of fruits are similar. Natives follow the
same manners to handle, store and ship fruits with little modifications to the
practice here and there.
5.2 Cereals
5.2.1 Maize
storage rot
Mature maize should be harvested from the field when the
moisture content (MC) is less and equal to (≤) 20-25% (Tan, 2015). They
should be stored immediately in cribs or over fire places in tropical
traditional homesteads (Personal communication, 2018). The produce stores well
when its moisture content is less than 12 % and the surrounding air is warm
enough to discourage fungal propagules from growing
on the crop (www.knowledgebank.irri.org/sep-by-step-..., Tan, 2015). Storage rots reduces the market grade
and the feeding value of the maize grains. Insects infestation of maize cobs in
field, transit and storage not only lowers its nutritional profile; it
contributes immensely to making grains loose more moisture and when stored in
enclosed store houses lead to development of hotspots, undue sprouting and
increased damage to seed viability. Ultimately all these predispose the stored
grains to fungal colonization (www.knowledgebank.irri.org/sep-by-step-..., Tan, 2015).
Moist store environment in tropical settings contributes
to making produce go mouldy some of these fungal contaminants such as Fusarium graminaerum, Aspergillus flavus, and Penicillium oxalicum produce noxious toxins. Consumption of maize
grains or feeds contaminated with various toxins from these microbes have been
reportedly linked to chronic or severe impairment of the metabolic processes of
the liver, skin, kidney and the CNS (Enyiukwu et al., 2014b; Enyiukwu
et al., 2018). Threshed kernels store
longer with every 1 % fall in seed moisture content and 5 % drop in ambient
temperature storage life of kernels is doubled. However, though there is
greater danger of breaking kernels during threshing when moisture content has
reached 3 %; at this moisture level storage moulds
are completely and successfully kept at bay.
Management of maize storage rots:
Ears should be harvested in dry state and stored in maize
cribs. The cribs should be rectangular in shape and positioned perpendicular to
prevailing wind direction to give maximum air circulation and drying. Depending
on the prevailing regulations, the walls should be dusted regularly with
insecticides such as essential oils such as neem
kernel and tea tree oils or extracts of black pepper (Asawalam
and Emosairue, 2006; Awurum
et al., 2014). Ensure that the crib
has adequate roof overhang and well protected from rodents. Don’t keep ears
beyond 3-6 months in cribs. When the moisture content of kernels falls to 12 %,
cobs should be shelled and grains stored in hermetic/airtight containers such
as tins, screw drums and polythene bags. Ensure also that the grains are cooled
before storing in the air tight containers (Lane, 2016).
5.3 Oil
seeds
5.3.1 Groundnuts
Groundnut should be harvested at 18-20 % moisture content
from the fields. They should be dried with the thrash and husk to about 15 %
moisture content which is good for threshing and dehusking
(www.knowledgebank.irri.org/step-by-step-production/prot..., 2019). Dehusked nuts
should be dried to less than 7-8 % moisture content to discourage fungi from
growing on the nuts. Nuts should be spread out on platforms which can be
carried inside at night or during wet weather. Very high temperatures stimulate
a degradative lipid process (lipolysis) leading to
release of free fatty acids (FFAs) in oils from groundnuts. Poorly dried
produce attract the growth of some mycotoxigenic moulds such as species of Fusarium, Aspergillus, Penicillium,
and Claviceps
purpurea (Enyiukwu et al.,
2014a, 2014b). Following harvest, aflatoxins and other hazardous fungi-derived toxins
contamination of the nuts would be prevented by rapid drying of the produce at
30oC in the sun for 6-7 days to moisture content of 12 %. At this point though the nuts will not be
entirely fungi-free, but the inocula density will be
tolerable.
Relative humidity (RH) of 70 % (or less) is essential for
good nut storage. Above this, there may be a stimulation of successive mould growth on the nuts and RH of 85 % activates aflatoxigenic moulds to grow and
contaminate the nuts. Other factors which can encourage fungal growth and
production of aflatoxins during storage of nuts
include:
o Presence and activity of insects,
o Rodent activity,
o Moisture migration through the store,
o Badly maintained store houses
Mycotoxins occur on a wide range of agro-produce especially oil
produce such as groundnuts and have been implicated in mycotoxicosis
in humans and domestic animals when ingested. The effects could be chronic
leading to deleterious effects on the CNS, cardiovascular system, hepatic
organs, liver, and the alimentary canal or acute exposures culminating
ultimately to death if not well treated. Effects such as immune-suppression,
carcinogenicity, mutagenicity and teratogenicity have also been linked with
these toxic microbial metabolites (Enyiukwu et al., 2018).
Aspergillus flavus is ubiquitous in the hot humid tropics and can colonize
groundnuts at various stages of development in the field. Moisture and
temperature are important weather parameters which determine the incidence of
the disease on oil seeds. Insects could exacerbate occurrence of mycotoxins contamination of nuts and oil seeds. Some important moulds
and their mycotoxins which commonly contaminate nuts
and oil seeds are presented below in Table 1.
Table 1: Some important moulds
and their mycotoxins on oil seeds
|
Moulds
|
Toxic metabolite(s) on oil produce
|
|
Aspergillus flavus
|
Aflatoxins
B1 and B2; Cyclopiazonic
acid
|
|
A. parasiticus
|
Aflatoxins
B1, B2, G1 and G2
|
|
Fusarium sporotrichoides
|
T-2
toxin
|
|
F. verticilloides
|
Fumonisins
B1 and B2
|
|
F. graminaerium
|
Deoxynivalenol, Zearalenone
|
|
Penicillium verrucosum
|
Ochratoxin
A
|
|
A. ochraceous
|
Ochratoxin
A
|
Source: Enyiukwu et al. (2014a; 2018)
Management of groundnut rots
Avoid application of nitrogen-rich fertilizers late in
the growth stages of groundnuts and other oil seeds. Harvested oil nuts and
seeds are best air dried in artificial chambers as sun drying takes about 5-7
days at 30oC and 75 % RH, which long period could permit mycotoxigenic rot microbed to
grow even to threshold levels. Stored
nuts should be maintained at 8 % moisture content in well cleaned
environments (Enyiukwu et al., 2014b). Packing stores in large agro-concerns should be
fumigated with phostoxin or methyl bromide. In
low-input agriculture, open village level stores should be well ventilated and
possibly installed with fans if cost and power permits. Stored nuts should be
processed for oil as quick as possible and the resultant cakes adequately
sundried before use in feeds.
5.4 TUBERS
5.4.1 Sweet
Potato
Sweet potato should only be held for a short period in
storage prior to marketing. A wide range of storage is used in the tropics including
storage in caves and trenches (Maranzu, 2019). Like
other perishable high moisture produce, ensure they sustain no or minimal
bruises during harvesting, transportation and marketing. Where tubers wounds are minimal or
non-existent, it is necessary to cure the tubers immediately after harvesting
at 30oC and relative humidity of 85-90 % for 5-7 days. Avoid
harvesting just before or after rains or irrigation as this could predispose
the produce to microbial spoilage. Ideally, the produce should be stored at
13-16oC and relative humidly of about 90 % (Enyiukwu
et al., 2014a; Maranzu,
2019). Generally, the red skinned varieties store better than the white skinned
ones (Personal communication, 2018). At present vitamin A rich orange fleshed
novel varieties which can be eaten raw or cooked to help fight vitamin A
deficiency (VAD) diseases in humans have been developed (Inyang,
2010). In traditional tropical farmsteads, storage of sweet potatoes are mainly
done in caves, pits or trenches lined with straw, bamboo and covered with
thatch or asbestos roof for protection against dew and rains. Depending on
agro-ecological zone, it can also be stored in clamps or huts whose walls are
lined with dry old newspapers, off-cuts and floors littered with dry saw dust or
grasses (Personal communication, 2018). The saw dust must be dry to keep tubers
cool and dry and discourage microbial rot development on the produce. When
storing large volumess of produce, ventilation is
necessary to circulate air through the mass of produce and keep them dry and
maintained at optimum storage temperature of about 25oC (Personal
communication, 2018).
5.4.2 Cassava
Traditionally in tropical farms of sub-Saharan Africa,
cassava is left in ground and harvested only when needed while in larger
ago-concerns they may be harvested for immediate use or processed into other
longer storing products such as garri (Personal
communication, 2018). On-plant in-ground
storage may be desirable, but it decreases other useful uses to which the land
could be put into. In this kind of method, susceptibility of the tubers to
pathogenic attacks and losses increase; while palatability and extractable
starch content declines when harvesting is greatly delayed (Personal
communication). Re-burial of small quantities of cassava tubers can preserve
them for a few days in moist soil. They may be alternatively coated with mud to
keep them fresh for a few days. Clamp storage has proved very successful and
should be used to store fresh tubers for 6-8 weeks. Clamps are made by heaping
cassava on a straw mat and coating it with a layer of mud, before covering it
with soil. An air vent is usually created in the clamp. Periodically the clamp
is watered and covered with palm fronds or dried grasses (Personal
communication). Cassava tubers keep well in clamps which
provides them with curing temperature and humidity (Maranzu,
2019).
5.4.3 Yam
For ages yams have been stored by traditional means in
barns. Though during dry season can be
left in the soil without appreciable loss of quality, however it is better to
harvest them into barns away from rodents and pig attacks. In some small scale
production systems they may be stored in room whose floors are matted with dry
straw or stovers.
Typically West African barns have the advantage of maximum air
circulation (Enyiukwu et al., 2014b). Yams for barn storage are first cured for 4 days at
29-30oC and 85-90 % RH at which time satisfactory healing occurs
around deep cuts. Tubers which do not heal such bruised areas are cut off. Yam barns
should be created under forest shades at the back of the farmers’ houses
(Personal communication). In general, cured yams should be stored at 16oC
and 70 % RH for 3-4 months. Removal of shoots from sprouted tubers helps to
prolong the storage life of yams and other tubers (Markson,
2010; Maranzu, 2019).
5.4.4 Irish
and Hausa potatoes
Irish potatoes are commonly grown in southern Kaduna and
around the Jos plateau areas of Nigeria. Care should be taken during
harvesting, transporting and marketing of potatoes to minimize abrasions which
could predispose them to microbial attacks. Harvested tubers should be cured at
8-20oC, 90% relative humidity for 5-8 days. Ensure that moisture does not accumulate on
the surface of the tubers. Potato is sensitive to oxygen tension; they should
therefore be stored in well ventilated stores which should be dark and dry (Enyiukwu et al.,
2014a; Tsedaley et
al., 2014). In many traditional agro-systems, potato is stored in clamps or
partial underground pits with air ducts under the tubers to allow the produce ultilize cool night air. Hausa potato (Solenostemon rotundifolius Poir)
J. K. Morton which are small-sized tubers and a commonly consumed tuber crop in
the middle belt and northen regions of Nigeria are
handled in much the same way. However, their storage is many in eaten vessel or
in underground pits located under tree shades (Enyiukwu
et al., 2014a; Nwaneri,
2017).
Management of tuber crops
Attacks by microorganisms are probably the most serious
cause of postharvest losses in root and other tuber crops in the country. Most postharvest bacteria and fungi are wound
requiring parasites launching their attacks from sites of injury. Hence
physical injury during harvesting, handling and transportation of tubers serves
to predispose the produce to microbial attacks. Quantitative losses have been
attributed to pathogenic breakdown of tuber tissues by several microbes. The
pattern of attack is frequently as an initial infection either from harvest or
commonly through a postharvest wound by a few specific pathogens, which is
later followed by massive attacks from a broad spectrum of organisms which grow
on dead or moribund tissues of the tubers. Qualitative losses on tubers also
results from blemishes incurred from nematodes and other parasites which
renders the tubers unsightly, less attractive and so reduces the market value.
Some pathogens which attack tubers include Macrophomina phaseolina (charcoal rot), gray mould rot (Botrytis
sp.) on sweet potato, Botrydiplodia theobromae on Hausa potato, Sclerotium
root rot (Sclerotium rolfsii), Phytophtora root rot (Phytophtora sp.) and bacterial blight (Xanthomonas manihotis) on cassava; watery rot (Erwinia sp), dry rot (Corynebacterium sp.) and Rhizopus sp., Aspergillus sp., Penicillium
sp. and Fusarium
sp etc on yams. Some of
these pathogens and their low-key botanical controls on tubers have been
broadly reviewed by Enyiukwu et al. (2014a). Ensure you cure bruised tubers by sun drying them.
Store tubers in barns shaded by trees or in stores whose floors are adequately cartoned or littered with saw dust. Ensure there are no
leaks from the roof and that a proper roof-overhang of stores is maintained
(Personal communication, 2018)
6.0 CONCLUSION
The place of fruits and vegetables in human diets cannot
be over emphasized. They represent
classes of produce associated with providing the body with wide ranging amino
acids, calories, vitamins and minerals in addition to anthocyanins
(constituents responsible for plant colours) which
helps to strengthen heart health and fight certain diseases and inflammatory conditions including cancer.
However, high doses of post-harvest
losses averaging 10- 50 % in fruits and vegetables have been reported to occur
due to pathological influences especially from wound-requiring fungi and
bacteria. Stemming post-harvest food wastages through avoidance of undue wounds
on produce during harvesting, curing, proper handling and transit or ensuring
prevention of insect and water migration into the store houses could contribute
to elongating shelf-lives of produce and food security in the continent.
REFERENCES
Afolabi IS Akpokene
ON Fashola
DV Famakin TC
(2012). Comparative evaluation of the nutritional benefits of some
underutilized plant leaves. J. Nat. Prod.
Plant Res. 2(2): 261-266.
Ajibade,
S. R. and Amusa, A. N. (2001). Effects
of some fungal diseases on some cowpea lines in the humid environment of South
Western Nigeria. Journal of Sustainable
Agriculture and Environment 2(3): 246-256
Ajuru M Nmom
F (2017). Review of the economic uses of species of curcubitaceae
and their sustainability in Nigeria. Am. J. Plant Biol. 2(1): 17-24. Doi:10.11645/j.ajbb.20170201.14.
Akang EN Oremosu AA Dosumu OO Noronha
CC and Okanlawon
AO (2010). The effect
of fluted pumpkin (Telferia occidentalis) seed oil (FPSO) on testis and sperm
parameters. Agric. Biol. J. N. Am.
1(4): 697-703.
Amadioha AC
(2012). Reducing food losses through sustainable methods of
plant disease management: An imperative for actualization of food security in
Nigeria. The 13th Inaugral Lecture
Presentation, Michael Okpara Universisty
of Agriculture, June 20, 2012.
Amadioha, A. C. (2004). Control of
black rot of potato caused by Rhizoctonia bataticola using some plant leaf extracts. Archives of Pytopatholology
and Plant Protection 37: 111-117
Amadioha, A. C. (1994a). Synergism between oxalic acid and polygalacturonase
in the depolymerization of potato tuber tissue.
World Journal of Micrbiology
and Biotechnology 9: 599-600.
Amadioha, A. C. (1994b). Pectolytic enzymes of Rhizopus arrhizus produced in culture and
infected potato tubers. Fitopatologica Brasilera 19:
139-143.
Amadioha AC Markson
AA (2007). Postharvest control of cassava tuber rot by Botrydiplodia acerina using extracts of plant origin. Phytopathol.
Plant Protec. 40(5): 357-366. DOI: 10.1080/03235400600624079.
Amadioha AC Enyiukwu,
DN (2019a). Alterations of
biochemical composition of leaf and stem of cowpea (Vigna unguiculata
L. Walp.) by Colletotrichum destructivum
O’Gara in Nigeria. Journal Experimental
Agric. Int. 32(2): 001-007.
Amadioha AC Enyiukwu
DN (2019b). Biochemical composition
of seed and husk of cowpea (Vigna unguiculata L. Walp.)
infected by Colletotrichum destructivum
O’Gara in storage. Annual research and
reviews in Biology 31(1): 001-007.
Asawalam EF Emosairue
SO (2006). Comparative efficacy of Piper guineense (Schumm.
Thonn.)
and primophos-methyl as
poisons against Sitophilis zeamais (Motsch). E-Journal
Environ Agric. Food Chem. 5 (5): 1536-1545.
Awurum AN Enyiukwu
DN
Ucheagwu PO (2014). Effectiveness of duration of storage and phytochemicals in the
management of seedborne mycoflora
of stored cowpea (Vigna
unguiculata
L. Walp) seeds. Greener J. Agric. For. Hort. 022-026.
Awurum AN Enyiukwu
DN Odoemena VK (2016). Influence of plant-gleaned compounds on the
initiation and development of fungal diseases of onion (Allium cepa L.) in the field. J. Biol. Agric. Healthcare 6(9): 71-80.
Awurum AN Enyiukwu
DN (2013). Evaluation of the seed-dressing potentials of
phytochemicals from Carica papaya and Piper guineense on the germination of
cowpea (Vigna unguiculata
L. Walp.) seeds and incidence of the seed-borne
fungi. Continental J. Agric. Sci.
7(1): 29-37.
Begum MM Sariah
M Puteh
AB Zainal-Adidin
MA (2008). Pathogenicity of Colletotrichum truncatum and its influence on soybean seed quality. Int.
J. Agric. Biol. 10(4): 393-398.
Begum JMA Venudevan
B Jayanthi M
(2013). Storage fungi in groundnut and the associated seed
quality deterioration – A review. Plant Pathol. J. 12(3):127-134.
Enyiukwu, D. N. (2019). Nature’s Pharmacy Vol. 1: A Focus on Carica Papaya L. Lagos Nigeria, GPS Publications Pp. 70
Enyiukwu DN Awurum
AN Nwaneri JA
(2014b). Mycotoxins
in stored agricultutal products: Implications to food
safety and health and prospects of plant-derived pesticides as novel approach
to their management. Greener
J. Microbiol. Antimicrobials 2(3): 032-048.
Enyiukwu DN Awurum
AN Nwaneri JA
(2014c). Potentials of Hausa potato
(Solenostemon
rotundifolius
Poir) J. K Morton and management of its tuber rot in
Nigeria. Greener J. Agric. Forestry
Horticulture 2(2): 027-037.
Enyiukwu DN Awurum
AN Nwaneri, JA
(2014a). Efficacy of plant-derived
pesticides in the control of myco-induced postharvest
and storage rots of tubers and agricultural products: A review. Net J. Agric. Sci. 2(2): 30-36.
Enyiukwu, DN Ononuju
CC Maranzu
JO (2018). Mycotoxins in foods and indoor air:
their attendant diseases and modes of injury on biological and human systems. Greener J. Epidemiology Public Health
6(1): 034-051.
Ephytia (2019). Identify, knowing and controlling
non-parasitic diseases – Black heart of potato.
www.ephytia.inra.fr/en/G/21136/Potato-Black-heart Accessed June 2, 2019.
FFTC (Food and Fertilizer Technology Cernter
for the Asian and Pacific Region) Anonymous (2019). Postharvest losses in fruits and
vegetables in Asia.
www.fftc.agnet.org/library.php?func=view&id=20110630151214.
FRM (Fruitrop Magazine Online)
Anonymous (2015). Avocado: Packing and postharvest management. Citrad Press, Pp 98-102. www.fruitrop.com/en/articles-by-subject/packing-post-harvest... Accessed June
15, 2019.
Garcha S (2018). Control of food spoilage moulds
using Lactobacillus bacteriocins.
Journal Pure Appl. Microbiol. 12: 1365-1373. Doi:
10.22207/JPAM.12.3.39.
Greenlife (Greenlife Crop Protection
Africa) Anonymous (2019). Bacterial soft rot of carrot. www.greenlife.co.ke/bacterial-soft-rot-of-carrot/ Accessed June, 14, 2019.
Gummars K
(2018). Twelve proven health benefits of avocadoes. www.healthline.com/12-proven-benefits-of-avocadoes#section1. Accessed June 14, 2019.
Hai HD (2019). Safou tree (Dacryodes edulis L.) In: worldwide fruits (Hai
Ho Dinh ed).
Long-An Vietnam. www.worldwidefruits.com/dacryodes-edulis--safou-tree.html Accessed June 12, 2019.
Hailu G Derberi
B (2015). Extent, causes and reduction strategies of postharvest losses of
fresh fruits and vegetables: A review. J.
Biol. Agric. Healthcare 5(5):
Imperial College London Anonymous (2012). Tackle fungal forces to save crops, forests and
endangered animals, says scientists. www.imperial.ac.uk/news/108986/tackle-fungal-forces-save-crops-forests/ Accessed May 21, 2017.
Inyang P
(2010). Effect of varietal vine pruning on vine production and yield of three
orange-fleshed sweet potato (Ipomea batatas (L.) Lam) varieties in southeast
Nigeria. A MSc
Results Presentation submitted to the Department of Agronomy, Michael OKpara University of Agriculture, Umudike
June, 2016.
Jeddi MZ Yunesian
M Khanki GR
(2014). Microbial evaluation of fresh and minimally- processed vegetables and
bagged sprouts from chain supermarkets. J. Health Pop. Nutri. 32(3): 391-399 (PMCID
4221445).
Kasso M Bekele
A (2018). Postharvest losss and
quality deterioration of horticultural cropsin Dire Dawa region Ethiopia. J. Saudi Society Agric. Sci.
17(1): 88-96. Doi: doi.org/10.1016/j.jssas.2016.01.005. www.sciencedirect.com/science/article/pii/S16580....
Kebede MK Baley
A (2019). Fungi associated with postharvest avocado fruit rot at Jimma Town Southern Ethio[pia. www.researchgate.net/publication/3332207545_2019_fungi-associated...
Accessed June 14, 2019.
Knowledge
Bank Anonymous (2019). Grain moisture content and grain
quality. In: step by step production, post production, drying and drying
basics . www.knowledgrbank.irri.org/step-by-step-production/postharvest/drying/drying-basics/grain
Accessed June 15, 2019.
Kumar, D Kalita,
P (2017). Reducing postharvest losses during storage of grain
crops to streghten food security in developing
countries. Foods 6(1): 8 Doi:
103390/foods6010008.
Lane B
(2016). Assessment of hermetic storage of maize under different
environmental conditions. Doi:
10.1603/ICE.2016.105521 Int. Congrress of Entomology, 2016. www.researchgate.net/publications/305804559_Assesment_of_hermetic...
Accessed June 24, 2019.
Madrid M (2011). Reducing postharvest losses and improving fruit qualitywordlwide: The one billion dollar untapped business
opportunity. www.fruitprofit.com/new/reducing-postharvest... Accessed June 4, 2019.
Magsasaka J
(2016). Cassava production: Harvest and harvest activities.
www.questrontech.wixsite.com/juanmagsasaka/single+post/2016/2016/1/7/Cassava...
Accessed June 4, 2019.
Mahovic M J. Bartz (2019). Disease management: Postharvest diseases of
tomatoes. UF/IFAS Plant Pathology Department, Fifield
Hall,Gainesville, Pp 142-156. www..ifas.ufl.edu/…
Mansfield J Genin
S Magori
S Citosky
V Sriariyanum
M (2012). Top 10
bacteria pathogens in molecular plant pathology. Molecular Plant Pathol. 13 (6): www.onlinewiley.com/doi/full/101111/j.1364-
Maranzu JO
(2019). Antagonistic activities of some mycobiota
against postharvest fungal rot of sweet potato in storage. A
Dissertation presented to the Department of Plant Health Management, Michael OKpara University of Agriculture, Umudike,
April, 2019.
Markson AA Omosun
G Umana
EJ Madunagu
BE Amadioha
AC Udo SE
(2014). Differential response of Solanum tuberosum L. and Ipomea batatas L. to three rot pathogens. Int. Res. J. Natural Sci. 2(1): 40-50.
Meyer, V., Andersen, M. R., Brakhage, A. A., Braus, G. H., caddick, M., Cains, T., de Vries, R. P., Haamannn, T., Hansen, K., Herz-Fowler,
C., Mortensen, U. H., Penalva, M. A., Ram, A. F.J.
and Head, R, M. (2016).
Current challenges of research on filamentous fungi in relation to human
welfare and a sustainable bio-economy – Scientific white paper. Fungal Biology and Biotechnology 3: 6
Mgbeahuruike, EE Yrjonen, T Vourela,
H Holm, Y (2017). Bioactive compounds from medicinal
plants: focus on PIper
species. South Affrican
J. Botany112: 54-59.
Mgbeahuruike, EE
Holm, Y Vourela, H Amandikwa, C Fyrquist, P (2018). An ethnobothanical
survey and antifungal activity of Piper guineense used for the treatment of fungal infections
in west African traditional medicine. J. Ethnopharcology
229: 157-166.
Mohammed, A., Chimbekujwo, I.
B., Bristone, B. (2013). Identification and control
of fungi associated with the postharvest rot of Solenostemon rotundifolius (Poir)
J. K. Morton in Adamawa State. J. Agric.
Healthcare 3(5): 136-140.
Muanya C (2016). Boost your libido this valentine with Mucuna and fluted pumpkin seeds. www.guardian.ng/features/boost-your-libido... Accessed June 8, 2019.
NAP
(National Academies Press) Anonymous (2008). Fruits – Butter fruit (Safou, Bush mango) In: Lost crops of Africa Volume III. Washington DC, NAP Press. Pp
61-75.
Nelson S
(2009). Rhizopus soft rot of sweet potato.
Department of Plant and Environmental Protection Sciences College of Tropical
Agriculture and Human Resources, University of Hawaiii
at Manoa (Cooperative Extension Servive)
CTA-HR Publ. www.ctahr.hawaii.edu/freepubs Accessed June 6,
2019.
Nischwitz, C Dhiman,
C Drost,
D (2013). Botrrytis
neck rot of onion. Utah Pest Factsheet, Utah State
Extension and Utah Plant Pest Diagnostic Laboratory, PLP 020, November, 2013.
Njoku DN
(2009). Cassava post-harvest physiological deterioration In: Food security and
nutritional quality GORE LAB
in collaboration with NRCRI, Umuahia. http://blogs.cornell.edu/gorelab/2017/02/ Accessed June 3, 2019.
Nwaneri JA
(2017). Potentials of some plant extracts in the control of
postharvest fungal rots of Hausa potato (Solenostemon rotundifolius Poir) J. K Morton in
Nigeria. A thesis submitted to the Department of Plant
Health Management, Michael OKpara University of
Agriculture, Umudike June,
2017.
Nwufo MI Emebiri
LC (1990). Pod rot of fluted pumpkin (Telferia occidentalis) in Imo State, Nigeria.
Int. Biodeterioration 26(1): 63-68.
Nwufo MI Obiefuna
JC Akobundu
ENT (2002). Effects of packaging materials and storage
temperature on quality and postharvest life of star apple (Chrysophyllum albidum). IJAR 2: 131-137.
Odiaka NI Amande
AP Maga TJ
(2013). Morpho-markers as sex determinants in fluted pumkin
(Telferia occidentalis)
Plants. Acta Hortic. 1007: 241-246. Doi: 10.17660/actahortic2013.1007.24.
Okigbo RN
(2003). Fungi associated with peels of postharvest yams in storage. Global J. Pure Appl. Sci. 9: 19-23.
Okigbo RN
(2004). A review of biological control methods for
postharvest yams (Dioscorea spp.) in South Eastern Nigeria. KMTL Sci.
J. 4: 207- 215.
Okpala B
(2016). Benefit of Canarium shweinfurthi
(African elemii). http://globalfoodbook.com/benefit-of-canarium-schweinfurthi-african-elemi-ube-osa. Accessed June 14, 2019.
Olayemi FF Adegbola
JA Bamishaiye
EI Awagu EF (2012). Assessment of postharvest losses
of some selected crops in eight local government areas of Rivers State,
Nigeria. Asian J. Rural Dev. 2:
13-23. Doi: 10.3923/ajrd.2012.13.23.
Pandy PK and Pandy KK (2012). Emerging strategies to control fungal
diseases in vegetables. Applied Mycol. Biotechnol. 2: 197-217.
Plumer, B. (2012). How the US manages to waste $165 billion in food each
year. In Economic policy Washington Post.
www.washingtonpst.com/news/work/wp/2012/03/22/how...
PNW (Pacific North West) handbook Anonymous (2019). Plant disease management. In:
PNW Pest Management Handbooks.
pnw.handbooks.org/plantdisease/host-disease/carrot-carota-soft-rot Accessed June 12,
2019.
Sadiku NA Sadiku
IBS (2011). Indigenous efforts by African farmers ensuring sustainability in
agricultural productivity in the face of changing climate: A proceeding of
Environmental Management Conference, FUNAAB, Abeokuta, Nigeria; September
12-15, 2011.
Salau IA Shehu
K (2015). Overview of the fungal diseases of vegetables in Sokoto State, Nigeria. Global Adv. Res. J. Agric.
Sci. 4(1): 001-005
Shephard GS (2008). Determination of mycotoxins
in human foods. Chem. Soc. Rev. 37(11).
2468-2477. Doi: 10.1039/b713084h.
TAMU (Texas Agricultural and Techanical
University) Anonymous (2019). Onion (Allium cepa L.) Texas Plant Disease
Handbook. http://plantdiseasehandbook.tamu.edu/food-crops/vegetable-crops/onion/
Accessed June 6, 2019.
Tan S
(2016). Storage of fresh fruits and vegetables.
Department of Primary Industries and Regional Development, Government of
Western Australia. www.agric.wa.gov.au/fruits/storage-fesh-fruits...Accessed
June 18, 2018.
Tsedaley, B.
(2014): early blight (Alternaria spp.) of potato disease and its
management options. Journal Boil. Agric.
Healthcare 4(27): 191-198.
TSGC (Tri-State Grain conditioning) Anonymous (2019). Is grain moisture content throwing a wet blanket on your
profits? http://tsgcinc.com/news/grain-moisture-content-affects-pofits
Wani, AH Taskeen-Un-Nisa (2011). Management of black mold rot
of onion. Mycopathol. 9(1): 43-49.
Worldwide Fruits Anonymous (2019). Dacryodes edulis (Safou
tree). www.worldwidefruits.com/dacryodes-edulis-safou-tree.html.
accesssed June 15, 2019
APPENDIX 1:
PERSONAL COMMUNICATIONS (2018, 2019) INTERVIEWS
A total of 8 farmers (growers) and 12 marketers/sellers
(all adults) of various kinds of fruits and vegetables were randomly
interviewed at the farm-gates and two major markets (Ndoru
and Ariam) markets respectively in Ikwuano Local Government Area of Abia
State on what things they do and how they them to prolong the shelf-lives of
their agricultural produce. The brief
interviews were carried out in the wet season (April-September) and dry season
(November-December) of 2018; with a totalof 10
respondent per cropping season. The views of or practices by these growers and
marketers of agricultural produce were captured in the interview as structured
below.
My Name is ……………….. am a Researcher at t……………………………….. I am conducting a research on the simple ways natives of this LGA preserve
their fruits and vegetables at home before transiting to the markets, as well
as during and after sales at the weekly markets. Could you answer these few
questions intended to capture some data/information for use in the above
research topic.
1.
What is your name?
2.
What is the name of your community/village? And your LGA?
3.
How old are you (Sir /Madam)? (What is your age?)
4.
Are you a farmer, grocer or both?
5.
Do you preserve your harvested fruits/vegetables? Yes or No
6.
If yes, where? On the farm, home or market, all of the
above?
7.
Do you think that bruises add to accelerated
spoilages/decays? Yes; No.
8.
How do you
minimize the spoilages? Storing under tree shades, wrapping with old
newspapers, admixing with lime or lemon, spraying with botanical oils.
9.
How do you
ship your fruits and vegetables – with local baskets or rubber crates?
10.
Either ways
do you overlay with plant leaves (such as igo, dry banana or Tectonia spp.) or old newspapers? Yes or No
11. If yes, how often? (That is do you use them: Regularly,
Sometimes and Occasionally)
12. How do you store your left-overs after sales each day? In
Fridges or others
13. If others, how? In
the open at night with cover or without cover?
14. How long can your produce last in these traditional
storage forms? 1-3; 4-7; above 1 week
15. What are your challenges and t what stage do you have the
most challenge along the chain harvesting, handling, transit of marketing, all
of the above?
16.
Thank you very much.
|
Cite this Article: Enyiukwu DN; Bassey IN; Nwaogu GA; Chukwu LA; Maranzu JO (2020). Postharvest Spoilage and Management of
Fruits and Vegetables: A Perspective on Small-Holder Agricultural Systems of
the Tropics. Greener Trends in Plant
Pathology and Entomology 3(1): 1-17.
|





