INTRODUCTION
Sweet potato
(Ipomoea batatas
(L) Lam) is a member of the plant family Convolvulaceae.
The crop is a dicot with a large starchy, sweet tasting root tuber. The leaves
of the crop are edible and are used as leafy vegetable. Both the tuber and the
leaves are eaten in several cuisines of some parts of tropical Asia and Africa
(Neilsen et al.,
1997). Sweet potato is suspected to have originated from America, from where it
spread throughout the entire tropical world. Tubers of the crop is highly rich in calories (energy), low glycemic
index carbohydrates and sugars, fibre, vitamins A (usually in the form of β-carotene) and C and minerals such as calcium.
It is also reported to contain large amounts of polyphenolics
and anthocyanins related to slowing down effects of
aging, dementia and diabetes in humans (Maranzu,
2019).
Sweet potato in 2016 was ranked 168h most
valuable crop in Hawaii, 3rd most important tuber crop in Nigeria
and classified amongst the 7th most important tuber crop in the
world (Nelson, 2009; Enyiukwu et al.,2014). As such it is seen as one of
the crops with strong potential to contribute to tropical food security (Maranzu, 2019). However, production of this valuable tuber
crop suffers from several constraints especially poor tropical storage
conditions and hygeine, and postharvest microbial
deteriorations (Sokoto and Ibrahim, 2007, Nwanja et al.,
2017; Maranzu, 2019). For these reasons fresh sweet potato
tubers have been reported to store for about three weeks only after harvest if
left untreated (Maranzu, 2019). Loses due to postharvest fungal attacks on
stored tubers ranging 20-100 % have been reported because of inadequate farm
and village-level storage (Arinze, 2005; Anyim, 2010) and this is especially true in sweet potatoes
known to have thin rind where postharvest loss of about 85 % has been
documented (Maranzu, 2019).
Toxic chemical residues have been reported to trail
treatment of stored agro-products with synthetic pesticides. In most instances consumption of such toxic
chemical residues-contaminated products leads to harmful effects such as
teratogenicity, allergies and even death on mammals (Enyiukwu
et al., 2014). Against this backdrop,
many workers have adopted plant-derived compounds as viable and safer
alternatives (Markson, 2010). Extracts of Azadirachta indica and Moringa oleifera
significantly frustrated the initiation and development rot caused by R. stolonifer
in sweet potato in Northern Nigeria. In another evaluation conducted in
Southern Nigeria by Amienyo and Ataga
(2007), these workers demonstrated that extracts of Garcinia kola, Zingiber officinale
and Alchornia
cordifolia inhibited rot development due to B. theobromae
in artificially wounded tubers of the crop. Similarly, Banso
(2009) found extracts of Annona muricata and Monodoro myristica to
sufficiently impede the growth and reproduction of R. stolonifer associated with rot of the
crop.
The
objectives of this study were therefore to evaluate the response of fungal rot
pathogens associated with sweet potato tubers to aqueous extracts of some
medicinal plants (Ocimum gratissimum,
Curcuma longa, Azadirachta indica,
Ficus exasperata and Achornea cordifolia) commonly
found around the University as
possible sources of storage bio-fungicides useable by low-input farmers for
preservation of tubers of the crop in Umudike,
Southeast, Nigeria.
MATERIALS AND METHODS
Source and preparation of plant samples
The experiment was conducted at the Plant Health
Laboratory of the National Root Crops Research Institute, Umudike,
Abia State, Nigeria (Latitude 50 291 N, longitude 70 331
E, and altitude 122 meters above sea level). The plant materials Ocimum gratissimum (leaves), Curcuma longa (rhizomes), Azadirachta indica (leaves), Zingiber officinale (rhizomes) and
Xylopia aethiopica
(pods) collected locally around the University community and authenticated by
the Department of Forestry, College of Natural Resources and Environmental
Management, Michael Okpara University of Agriculture,
Umudike (MOUAU) were used for the study. The plant
materials were washed thoroughly in tap water, rinsed in sterile distilled
water and air-dried on the laboratory bench for 20 days. Thereafter, they were
milled separately into fine powder using a Thomas Wiley machine (Model: A500, USA) to obtain
300 g of each specimen which were stored separately in air-tight bottles. Each
powder was weighed out separately into 30 g and put into different
250 ml conical
flasks, to which
100 ml of
sterile distilled water
was added and
the flask stoppered with foiled
stoppers. These were allowed to stand for 2 h. Thereafter they were strained
separately through 4-folds of sterile cheese cloth into different 200 ml
beakers to obtain the respective aqueous filtrates of 30 % strength of the
extracts (Amadioha, 2003).
Preparation of culture medium (PDA)
About thirty nine point five (39.5) grams of potato
dextrose agar (PDA) (Oxoid™ ThermoScientific
Product, England, UK) was dissolved in 1000 ml of sterile distilled water in
1000 ml conical flask to which 4 drops of lactic acid was added, and then
stirred vigorously before the flask was stoppered with a foiled cotton wool and
autoclaved at 15 pounds per square inch (Psi) (152 cmHg,
120 0C) for 15 minutes.
Isolation and identification of the fungal rot organisms
of sweet potato
The fungal rot organisms associated with sweet potato
were isolated based on standard protocols as adopted by Amadioha
(2004) and Amadioha and Markson
(2007a, b). The colour and colony characteristics of
the isolates were observed under the microscope and recorded. Slides of the
organisms were prepared, fixed, mounted and examined under a low-high power of
the microscope to ascertain the identity of the pathogens with reference to Barnett and Hunter (1998). Records of the
type and number of times each type of organism was observed on the incubated
sweet potato tissues was also taken and used to determine the percentage
occurrence of the mycoflora using the formula adopted
by Maranzu (2019) as:
Pathogenicity test of the organisms
This was
conducted based on the procedure adopted by Amadioha
(2004) and Okigbo and Nmeka
(2005). All the isolates were tested individually for their ability to initiate
rot disease symptom on healthy (uninfected) sweet potato tubers. The healthy
sweet potato tubers were surface sterilized with 70% ethanol and bored on the
surface with a 3 mm cork borer. Disc (3 mm) of the
pure culture isolate was separately inserted into the hole and plugged with the
sweet potato tuber tissue previously removed from the tuber after 1 mm had been
cut to compensate for the thickness of the disc. The inoculated points were sealed with the
sterile gel to prevent contamination. All the inoculated sweet potato tubers
containing different isolates were incubated at room temperature (27–30 0C)
over a period of 8 days and examined daily for rot symptoms such as wetting,
softening, discoloration (darkening) and emission of offensive odour. Those that showed rot symptoms at the end of the
incubation period were each cut longititudinally
through the uninfected portion to expose the rotted regions. The rotted regions
were re-plated on PDA and re-isolated (Amadioha,
2004). The isolate which compared with the original culture introduced. The
isolates that caused rot and bore true morphological and cultural resemblance
of the original culture were regarded as pathogens whereas those isolates that
do not cause any rot were regarded as saprophytes and discarded.
In
vitro Experiment
Preparation of spore suspension
The spores of the pathogenic organisms (R. stolonifer and
B. theobromae) were collected from 10-day old
culture-agar stock in Petri dishes by lifting 60 cm2 pieces into a
beaker containing 200 ml of sterile distilled water. This was sieved through
4-folds of sterile cheese cloth to remove agar and mycelia fragments and the filtrate
centrifuged for 10 minutes (Amadioha, 2004). The
spores suspension was then standardized using a haemocytometer
counting slide to 105spores/ml of sterile distilled water.
Effect of plant extracts on spore germination
of the test pathogens
A disc (3mm) of each of the pathogenic fungi of sweet
potato was placed separately in 3 ml of the 30 % crude aqueous extracts from
the different plant materials or 3 g/L of benomyl as
standard check placed in different test tubes and centrifuged for 10 minutes, and
then filtered through 4-folds of cheese cloth. A drop (0.05 ml) of the
different preparations was placed separately on 3 sterile slides and incubated
for spore germination at 27 0C for 24 h in a humid chamber. The
controls consisted of sterile water or benomyl
instead of the crude plant suspensions. At the lapse of the evaluation time,
further spore germination was stopped by adding one drop of lactophenol
in cotton blue to each preparation on the slides. The effect of the tissue
extracts from the plant materials on the germination of the spores of the test
fungus was determined by randomly examining 100 spores of the pathogen under a
high-power (x400) microscope field. Records of the number of germinated spores
for each treatment/replicate were taken and then used to determine the
percentage inhibition of spore germination of the pathogen compared to the
controls using the formula by Amadioha (2003) as:
Inhibition of spore germination = gc – gt x 100
gc
1
where gc = average
number of germinated spores of
the test fungus with control
gt = average number of
germinated spores of the test fungus with treatment.
Effects of the extracts on radial growth of
the test pathogens
One (1) ml of the 30 % concentration of the crude aqueous
extracts of the plant materials were smeared separately on the surface of
solidified PDA contained in Petri dishes by gentle swirling motion (Amadioha, 2003; 2004). A disc (3mm) of the 10–day old
cultures of the pathogenic fungi was transferred to the centre
of the solidified PDA-extract medium in the Petri dishes which had been marked
underneath with two perpendicular lines intersecting at the center. The dishes
were covered and incubated at 27 0C for 7 days. The controls
consisted of PDA medium impregnated with sterile distilled water or benomyl in the dishes. The radial growth of the pathogen
was measured along the perpendicular lines with a meter rule 7 days after
incubation. This experiment was laid out in a Completely Randomized Design
(CRD) consisting of 7 treatments and 4 replications. The antifungal effects of
the extract was determined as a percentage of mycelial
growth inhibition and calculated by the formula as adopted by Amadioha (2003):
% Growth inhibition = dc – dt x 100
dc 1
where dc = average diameter of fungal colony in
the
control experiment
dt = average diameter of fungal colony with treatment.
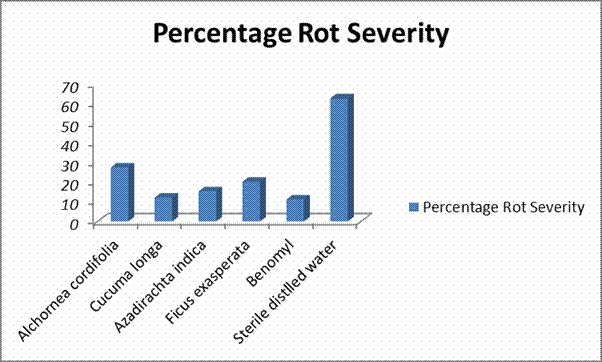
In vivo experiment
This was conducted based on the protocols adopted by Amadioha (2004) and Agu et al. (2016). A total of 24 tubers each
were impregnated with 5 mm diameter disc of R.
stolonifer while another set of 24 of tubers were
inoculated with B. theobromae
and incubated on laboratory bench for 21 days. Rot initiation and development
in the tubers were assessed as percentage area of the treated tubers macerated
by the rot pathogens using the formula by Agu et al. (2016) as:
Rot severity (%) = W-w x 100
W 1
Where W = final weight of the inoculated tubers
w =
weight of rotted regions of the treated tubers
DATA ANALYSIS
The whole experiments was
repeated twice. Data collected from the study were subjected to analysis of
variance (ANOVA) using the general linear model procedure in Genstat Release (Windows/PC Vista, version 12.10) at significant level of 5 %. Means were separated
and compared using Fishers LSD at probability of 0.05.
RESULTS
A total of six fungi were isolated from the rotted tuber
of sweet potato in storage in Umudike. The six fungi
isolated were Rhizopus stolonifer, Botryodiplodia theobromae, Fusarium oxysporium, Penicilium expansum, Aspergilus niger
and Aspergilus flavus (Table
1). Rhizopus stolonifer was the most frequently
isolated organisms (70.01%), followed by Botryodiplodia theobromae
(67.9%). Tests of pathogenicity revealed that these two organisms were
virulently pathogenic causing soft and dry rot respectively on the test tubers,
whereas the other fungal agents only induced slight rot and therefore were
discarded.
Table 1: Frequency of occurrence of fungi associated with
storage rot of Sweet potato
|
Fungal isolate Fequency
(%) Type of rot
|
|
Aspergilius niger
|
40.04
|
Dry rot
|
|
Aspergilius flavus
|
25.92
|
Dry rot
|
|
Botryodiplodia theobromae,
|
67.90
|
Dry rot
|
|
Rhizopus stolonifer
|
70.01
|
Soft rot
|
|
Fusarium oxysporum
|
20.01
|
Dry rot
|
|
Penicilium expansium
|
31.61
|
Dry rot
|
Table 2 indicated the effect of four plant materials and benomyl (a synthetic fungicide) on the inhibition of spore
germination of the fungal rot pathogens (R.
stolinifer and
B. theobromae) of the test sweet potato
tubers. Amongst the botanical treatments,
A. indica
recording 83.08 % demonstrated the highest inhibition of germination of spores
of R. stolonifer
followed by C. longa with 80.02 %; whereas C. longa (74.41 %) and A. indica (69.93 %) showed ultimately and pen ultimately
the highest inhibition of spores germination of B. theobromae.
Extracts of C. longa recorded the
best mean percentage inhibition of spore germination of the pathogens (77.22%).
This was followed by extract of A. indica (76.51%), F.
exasperata (61.93%); and the least effective in inhibition effects was the extract of A. cordifolia which recorded 40.89% reduction in mean spore
germination of the pathogens. Benomyl recording mean
inhibition of (91.37 %) was significantly (P≤0.05) superior to the
inhibition effects from the plant extracts on the pathogens. However, effects
obtained from C. longa and A. indica were
favourably (P≤0.05) comparable to it (Table 2).
DISCUSSION
The results from this study indicated that R. stolonifer
and B. theobromae
doubled as the most frequently isolated and most virulent myco-pathogens
associated with the postharvest microbial spoilage of sweet potato. This
observation is in tandem with the reports of Markson
(2010) and Maranzu (2019) who also observed that B. theobromae
and R. stolonifer
were the most commonly occurring and most virulent fungal rot pathogens involved
in the postharvest deterioration of white yam (Dioscorea rotundata)
and sweet potato (Ipomea batatas L.)
in storage in Southeast and Northwest Nigeria respectively. This finding is also in conformity with the
findings of Tijjani et al. (2013) who also found R.
stolonifer as the most frequently occurring and
virulent pathogen on mechanically wounded sweet potato tubers in Northern
Nigeria. However, findings in this study are not in agreement with the
submissions of Eze and Amah (2011) who reported that R. stolonifer
was the least virulent pathogen in an evaluation of postharvest rots of cocoyam
corms in Southeast Nigeria. Differences
in the inherent genetic potentials of the fungal biotypes (Nwaneri,
2017) or differences in structure and composition of the starch granules (Ballogou et al.,
2013) or other biochemical dissimilarities in the different varieties of tubers used in the studies may be the reason for the
divergent observations.
The test fungi (R. stolonifer and B. theobromae)
of sweet potato in this study were strongly sensitive to the plant extracts
incorporated in the study medium resulting in the inhibition of their growth
and reproduction (Tables 2 and 3).. Many workers have reported that plant-derived
pesticides act by impeding the germination of spores and retarding mycelia
growth or biomass accumulation of pathogenic fungi (Amadioha,
2003; 2004). Ethanol extracts of Afromonium meleguata and Zingiber officinale as well as aqueous extracts of Garcinia kola massively inhibited spore
germination and strongly retarded radial growth of B. theobromae in culture. Studies
conducted by Enyiukwu and Awurum
(2011; 2012) showed that the spore germination and mycelia elongation of Colletotrichum destructivum
responsible for anthracnose disease of southern pea were significantly
inhibited in culture medium impregnated with aqueous extracts of Piper guineense seeds.
Therefore findings in this study where the spore germination and radial growth
of R. stolonifer
and B. theobromae
were frustrated by the different plant extracts agree with the observation of
the workers as stated above.
Several extracts of plant origin has been reported to
effectively inhibit mycelial elongation and biomass accumulation
of pathogenic rot fungi as such foster protection of treated tubers for many
months post-harvesting (Enyiukwu et al., 2014). Seed tissue extracts obtained from Piper nigrum, A. meleguata, Garcinia kola, and
leaves of Dennetia
tripetala, Cassia alata and
Ocimunm basilicum
potently protected cassava and cocoyam tubers against deteriorations due to
attacks by B. acerina,
B. theobromae, R. orryzae, and
Sclerotium rolfsii in vivo
(Amadioha and Markson,
2007a, b; Nwachukwu and Osuji,
2008; Ugwuoke et
al., 2008; Anukworji et al., 2012). In like manner yam and sweet potato tubers have been
reported to be protected in vivo from
postharvest spoilage due to B. theobromae and R.
stolonifer by extracts of Annona muricata, Monodora
myristica, M. oleifera,
and A. cordifolia
(Banso, 2009; Amienyo and Ataga, 2007; and Tijani et al., 2013). Findings in this study in
which the test plant extracts demonstrated significant inhibition of the
attacks of the test pathogens on the tubers leading to their protection are in
agreement with the reports of the earlier workers as mentioned above.
Therefore based on the results of this study aqueous
extracts obtained from C. longa, A. indica, and F. exasperata in this order) could be used to protect
sweet potato tubers in storage and enhance their shelf-life. In addition, we
recommend that these plant materials especially C. longa be subjected to phytochemical profiling to isolate and
characterized their active ingredients so as to hasten development of
bio-pesticides from them.
REFERENCES
Agu, K. C., Awah N., Benjamin, O.
C., ... Umeadugwu, N.
(2016). Mycological determination and pathogenicity studies
of postharvest cassava. Food Science Technol. 4(2): 23-30.
Amadioha, A. C. (2003). Evaluation of some plant leaf extracts against
Colletotrichum lindemuthianum in
cowpea. Acta Phytopathologica et Entomologica Hungarica 38: 259 – 265.
Amadioha, A. C. (2004). Control of black rot of potato caused by Rhizoctonia bataticola
using some plant leaf extracts. Archives
of Pytopatholology and Plant Protection 37:
111-117.
Amadioha, A. C. Markson, A. A. (2007a).
Postharvest control of cassava tuber rot caused by Botrydiplodia acerina using extracts of plant origin. Arch Phytopathol,
40(5):359-366.
Amadioha, A. C and Markson, A. A.
(2007b). Control of storage rot
of cassava tuber caused by Rhizopus oryzae using some plant extracts. Arch Phytopathol, 40(6):381-388.
Amienyo, C. A. and Ataga, A. E.
(2007). Use of indigenous plant extracts for the
protection of mechanically injured sweet potato (Ipomea batatas (L.) tubers. Scientific
Research and Essays 2(5): 167-170.
Anukworji CA, Putheti RR, Okibgo RN (2012). Isolation of fungi
causing rot of cocoyam (Colocasia esculenta
(L.) Schott) and control with plant extracts: (Allium sativum L., Garcinia kola, Azadiachta indica
and Carica papaya L.). Global Adv Res J Agric
Sci. 1(2):33-47.
Arinze, A. E. (2005). Plant pathology and
postharvest food losses. An inaugural lecture series
(University of Port Harcourt) No. 43 pp. 29-72.
Ballogou, V.Y.,
Soumanou, M. M. Fatiou, T. and Houndouigan, J. D. (2013).
Structure and nutritional composition of fonio (Digitaria exilis)
grains: A review. Int. Res. J. Biological Sci. 2(1): 73-79.
Banso, A, (2009). Effects of extracts of Monodora myristica and
Zingiber officinale on
the growth of fungi in sweet potato. Afr. J. Microbiol.
Res. 3(9):487-490.
Barnett, H. L. and Hunter, B. B. (1998). Illustrative
genera of imperfect fungi (4th Ed.) American American Phytopathological Society, Minnesota, USA.
Enyiukwu, D. N and Awurum, A. N.
(2011). Effects of phytochemicals
from Carica papaya roots and seeds and Piper guineense
seeds on the germination of spores of Colletotrichum destructivum. Cont. J. Biol. Sci. 4(20):55-59.
Enyiukwu, D. N. and Awurum, A. N. (2012). Comparative fungitoxicity
of benomyl and extracts of Carica
papaya roots and seeds and Piper guineense seeds on Colletotrichum destrucivum
O’Gara. Continental J. Biol Sci. 6(1):38-42.
Enyiukwu, D. N. and Awurum, A. N. and Nwaneri,
J. A. (2014). Efficacy of plant-derived
pesticides in the control of myco-induced postharvest
and storage rots of tubers and agricultural products: A review. Net J. Agric. Sci. 2(2): 30-36.
Eze, C. S. and Amah, G. I. (2011). Comparative assessment of pathogenicity of storage rot causing
fungi of cocoyam (Colocasia esculentus L)
Schott and their hosy-pathogen relationship. Nig. J. Biotecnol.
22: 23-27.
Maranzu, J. O. (2019). Antagonistic activities
of some mycobiota against postharvest fungal rot of sweet
potato in storage. A Dissertation presented to the Department of
Plant Health Management, Michael OKpara University of
Agriculture, Umudike, December,
2019.
Markson, A. A., Omosun, G., Umana, E. J., Madonagu, R. E., Amadioha, A. C. and Udo, S. E. (2014).
Differential response of Solanum tuberosum L. and Ipomea batatas L. to three pathogens. International Journal of Natural Sciences 2(1): 40-51.
Markson, A. A. (2010). Fungicidal potential of extracts of some
plants for the control of postharvest tuber rot of yam (Dioscorea rotundata) In Crross
River State Nigeria. A Dissertation submitted to the Department of Plant Health
Management, Michael Okpara University of Agrriculture, Umudike, Febuary, 2010.
Nwaneri JA (2017). Potentials of some plant
extracts in the control of postharvest fungal rots of Hausa potato (Solenostemon rotundifolius Poir)
J. K Morton in Nigeria. A thesis submitted to the
Department of Plant Health Management, Michael OKpara
University of Agriculture, Umudike
June, 2017.
Nelson S (2009). Rhizopus soft rot of sweet potato. Department of Plant and
Environmental Protection Sciences College of Tropical Agriculture and Human
Resources, University of Hawaiii at Manoa (Cooperative Extension Servive)
CTA-HR Publ. www.ctahr.hawaii.edu/freepubs Accessed June 6,
2019.
Nielsen, S.S., Ohler,
T.A. and Mitchell, M.C. (1997): Cowpea leaves for human consumption:
production, utilization and nutrient composition. In: Advances in Cowpea
Research, Singh, B. B. (Eds), Ibadan, IITA, pp.
126-132.
Nwachukwu, E. O. and Osuji, J. O.
(2008). Evaluation of plant extracts for antifungal
activity against Sclerotium rolfsii
causing cocoyam cormel rot in storage. Res. J. Agric. Biol. Sci. 4(6):787-793.
Nwanja, Y. P., Golden, E. E. and Gugu,
F. M. (2017). Assessment of rot and
vine yields of sweet potato (Ipomea batatas L.) landraces as influenced by plant population
density in Jos Plateau
State, Nigeria. Int. J.
Agric. Res. 12: 88-92.
Okigbo, R. N. and Nmeka, I. A. (2005).
Control of yam tuber rot with leaf extracts of Xylopia
aethiopica and
zingiber officinale. Afr J Biotechnol,
4(8):804-807.
Sokoto, M. B. and Ibrahim, Z. (2007). Analysis of sweet potato consumption
in Sokoto metropolis of Sokoto
State, Nigeria. J. Biol. Sci.
7: 445-447.
Tijjani, A., Adebitan, S. A., Gurama, A. U., Aliyu, M., Dawakiji, A. Y., Haruna, S. G.,
Mohammed, N. A. (2013). Efficacy of some botanicals for control of wet rot
disease of mechanically injured sweet potato caused by Rhizopus
stolonifer in Bauchi
State. Int J Sci Res Pub,
3(6):1-10.
Ugwuoke, K. I., Onyeke, C. C. and Tsopmbeng, N. G. R. (2008). The efficacy of botanical protectants
in the storage of cocoyam (Colocasia esculenta (L.) Shott). Agro-Sci J Trop Agric
Food Environ Ext. 7(2):93-98.