Cowpea (Vigna unguiculata
(L.)
Walp.), is both a delicacy
and livelihood crop for many households in Sub Saharan Africa where it
contributes to food and nutrition security (Adeigbe
et al., 2011; Alemu et al., 2016; Silver et al., 2018). This is associated with its good
protein quality with a high nutritional value, nitrogen fixing ability,
tolerance to drought and heat, quick growth, and rapid ground cover (Magashi et al., 2014; Lado et al., 2016). In harsh
environments, the crop yields comparably higher than other food legumes (Shiringan & Shimelis, 2011).
Therefore, cowpea provides multiple benefits to smallholder farmers in terms of
food, income, and livestock feed, and improving and maintaining soil fertility
(Adeigbe et al.,
2011; Olasupo et al., 2016; Thio
et al., 2016; Lado
et al., 2016).
Despite the importance of the crop, it is yet
affected by a number of biotic and abiotic constraints that lead to low yields
of both grain and fodder (Animasaun et al., 2015;
Horn et al., 2015). Among the most important biotic factors that limit the productivity
of cowpea in Tanzania, yellow witchweed (Alectra vogelii), is the principal parasitic weed. The
weed attacks cowpea especially in semi-arid areas with low or acute nutrient
deficiency in the Sub-Saharan Africa region (Kutama
et al., 2014; Mbwando
et al., 2016; Njekete et al., 2017). The weed needs
to be controlled before it flowers and produce seeds. If uncontrolled the weed
will produce seeds which will results into soil contamination with the Alectra seeds.
Once the soil is contaminated with the seeds, soil seed bank will increase and
weed seeds will spread to new areas which will results into poor land quality
and consequently food insecurity (Atera et al., 2013;
Mbega et al., 2016).
A. vogelii
causes tremendous damage to the host plants before it emerges from the soil
affecting its vigour and performance as it emerges (Kwaga, 2014).
Several control measures against Alectra vogellii have been
developed which include application of fertilizers, herbicide and cultural
practices among others. However, these measures are not economically feasible
and sometimes not successful in controlling the weed (Kwaga,
2014). Therefore, the development and deployment of resistant cowpea varieties
remain the most effective method to combat the problem presented by the weed
and enhance food security among small holder farmers (Omoigui
et al., 2012; Abdou, 2017). Despite of the efforts to popularize cowpea
crop, the incidence and spread of A. vogelii appears to be on the increase due to
aggressiveness of the weed and appearance of new and highly infective A. vogellii
strains (Kabambe et al. 2013). Therefore, the aggressiveness and evolution
of new virulent strains of A. vogelii, call for studying the response of the improved
cowpea varieties to Alectra
strains in selected areas. The purpose
of this study was to determine the effect of Alectra
strains infestation on performance of improved cowpea genotypes.
MATERIALS AND METHODS
Study area
A screen house pot experiment to determine
the response of improved cowpea varieties against yellow witchweed
was conducted at Ilonga Agricultural Research Institute
(ARI Ilonga), in Morogoro,
Tanzania, (06° 42’S, 37°02’ E, Altitude 506 meters above sea level)
Alectra vogellii
germination test
Petri dish
method was used during germination test. This method allows more detailed
studies on induction of germination. Preconditioning of Alectra seeds was performed under
sterile conditions in a Laminar Air flow cabinet before the seeds become
responsive to germination stimulants. The active seed germination stimulant of
the parasitic weeds of Orabanchaceae
called GR24 was used to test for germination of Alectra seeds. Five gram (5g) of Alectra seeds
were surface sterilized in 5 % (v/v) sodium hypochlorite solution for 30
minutes in a test tube, with gentle agitation. Subsequently the seeds were
rinsed thoroughly with 100 ml of sterile distilled water, then spread on a
glass fiber filter paper (Whatman GFA), put into
sterile Petridishes and wet with 2.5 ml of sterile
distilled water. The Petridishes were then sealed
with parafilm and wrapped with aluminium
foil to prevent water losses and exclude light. There after the Petridishes were placed in an incubator for 14 days at 30 0C
for conditioning. The period of conditioning allows the seed to germinate
because seeds imbibe water before exposure to germination stimulant. After
conditioning, the Alectra
seeds were treated with a sterile germination stimulant (GR 24) to induce
germination. Equal volume of 2.5 ml of the GR24 stimulant was added to each
Petri dish of 9.0 cm having the pre-conditioned seeds. When the radicle protruded through the seed
coat, the seeds were considered to have good germination.
About 500 Alectra seeds of each strain were
thoroughly mixed separately with 250 ml of sterilized sieved sand to form the
inoculum stock. The inoculum stock was used to inoculate the top 5 cm of each
experimental pot which contained a mixture of soil and sand (3:1 v/v). After
inoculation, the Alectra seeds were pre-conditioned for seven
days before sowing the cowpea seeds to enhance Alectra seed germination. After
the soil was inoculated with Alectra seeds, the pots were watered daily to field capacity
for seven days consecutively to precondition the seeds to break their dormancy
and ensure optimum germination.
Experimental design
The experiment was designed as a spilt plot
experiment laid in a randomized complete block design (RCBD) with three
replications. Alectra strains (4 strains namely strain 1, 2, 3 and 4) were used as main
plot (factor A) and the cowpea genotypes were used as sub plot (factor B). Five
cowpea genotypes were used as treatments in this experiment, namely; Vuli AR1, Vuli ARI 2, Mkanakaufiti, and with local checks B 301 (resistant) and Vuli- 1 (susceptible). The genotype Vuli-1 is the locally
adapted and a widely grown cowpea cultivar. Three (3) seeds of cowpea were sown
per pot in 3 replications and at uniform depth in holes 5 cm deep. The
seedlings were thinned out and two were maintained per pot at two (2) weeks
after germination. The soil was kept moist by watering regularly every two days
or when necessary.
Data collected include; plant height, number
of leaves per plant, number of branches per plant, number of nodes per plant,
leaf area index (LAI), days to 50% flowering of cowpea plants, days to 95%
physiological maturity, days to Alectra emergence, and number of Alectra per plant. The cowpea
plants were harvested at physiological maturity, when more than 95% of the pods
were dry and brown.
Yield in a crop is governed by yield
components (Oladejo et al., 2011). Yield variables
measured include the number of pods per plant, pods length, number of seeds per
pod, 100 seeds weight, and seed yield. The number of pods per plant was
obtained by randomly selecting five plants within the sub-plot and counting the
pods on them. The average number of pods per plant was then determined. Also,
10 pods from each sub plot per replication were selected and their lengths
measured with a meter ruler. For the average number of seeds per pod, twenty
pods from each sub plot per replication were shelled and the seeds counted and their
averages calculated. Four lots of 100 seeds from the shelled pods of each sub-
plot were counted and weighed. The average was then taken as the weight of 100
seeds. After harvesting pods from each sub- plot, were shelled, the seeds were
weighed and the average was calculated to determine the final yield and
expressed in grams per plant (g plant-1). These data were then analysed using GenStat Discovery
16th Edition. Means were separated using Turkey Honest Significance
Difference test at 5% level of significance.
RESULTS AND DISCUSSION
There were significant differences (P = 0.001), among
cowpea genotypes in response to Alectra strains (Table 1). Results
showed that, four resistant genotypes (B301, Mkanakaufiti,
Vuli AR1 and Vuli AR2) were
also infested by A. vogelii. Cowpea genotypes
supported Alectra emergence whereas
Vuli-1 supported the earliest emergence of the weed, followed by Vuli AR1, and the latest was B301. The effect of cowpea
genotypes on Alectra emergence was first
observed in strain 4 of A. vogelii, followed
by strain 2 and latest in strain 1. The difference in days to emergence
was observed among the strains of A. vogelii and among cowpea genotypes. Such differences
were also reported by Alonge et al. (2001) that
cowpea varieties have genotypic differences in their response to A. vogelii. These observed responses of cowpea genotypes
to A. vogelii strains indicate that the genes controlling these parasites are
non-allelic and independent of one another (Omoigui et al., 2012; Mbwambo et al., 2016; Ugbaa et al., 2020).
There was significant (P=0.001) effect of Alectra
vogelii emergence on cowpea genotypes (Table 1).
Genotype Vuli 1 was the earliest, supported the A. vogelii emergence 33.2 days after planting (DAP) and the genotype B302 was the latest (42.8
DAP). Cowpea genotypes differ in days to emergence due to the differences in
the thickness of the seed coat and tissue layers among the genotypes (Onyishi et al., 2013). Also, the difference in days to A. vogelii
emergence is the result of the ability of cowpea genotypes to stimulate the germination of Alectra seeds and
subsequently allowing for the emergence of shoots. The
days to Alectra emergence in B 301 coincided with its days to flower onset (Table 1 and 2). Thus, B301 is having the attributes of low
production of stimulants for the germination of Alectra
seeds as well as attachment and prevention of haustoria
formation and subsequent development of the seedling of the parasite. Previous studies on cowpea, soybean, and
groundnuts reported the emergences of A. vogelii at 55 DAP, 75 DAP and 109 DAP, respectively (Kabambe et al.,
2008) which contrast with 37.5 DAP found in this study. This would suggest
that, the A. vogelii
strains used in this study were very aggressive, or cowpea genotypes were able
to allow the emergence of A. vogelii strains
due to their ability to produce high levels of stimulants.
There were
significant differences (P = 0.001), on the effect of genotypes
on number of Alectra shoots at 35 DAP, 49 DAP
and 63 DAP. At 35 DAP, Vuli-1 had the
highest number of Alectra shoots per plant
(SPP), followed by Mkanakaufiti and the lowest SPP
was recorded in B301 (Table 2). At 49 DAP, Vuli-1 had highest number of Alectra shoots per plant, followed by Vuli AR1 and Vuli AR2 which had
the same number of Alectra shoots per plant
and the lowest was recorded in B301. At 63 DAP, Vuli-1
had higher number of Alectra shoots per plant,
followed by Mkanakaufiti and lowest in B301. The
resistance in genotype B 301 has been reported to be controlled by a
single major gene, which may not be durable (Gnanamanickam
et al., 1999) because resistance conferred by a single major gene (vertical
resistance), frequently fails to provide long term control to parasitic weeds.
If such varieties are grown over a broad area they potentially lead to serious
breakdown of resistance. Therefore, breeding for vertical gene resistance
requires pyramiding of more than one gene from diverse resistance sources into
a single genotype as vertical resistance is associated with a common phenomenon
of the resistance breakdown (Gnanamanickam et al.,
1999). This would provide a better option so as to delay breakdown, broaden the
resistance genetic base and provide much needed durable resistance.
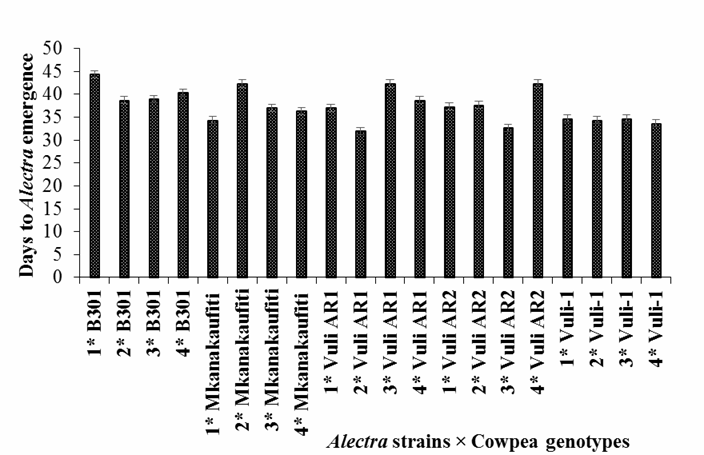
Interaction effect on cowpea genotypes and A. vogelii
showed that, days to Alectra
emergence were different among cowpea genotypes and Alectra strains (Figure 1).
However, Alectra emerged earlier in Vuli-1 with strains
2, 3 and 4, followed by the same genotype Vuli-1 with strain 1 and Vuli AR1 with strain 4, all with the same number of days to
Alectra emergence, whereas Alectra emerged latest in B301.
Interaction effect of cowpea
genotypes and strains of Alectra vogelii on
number of Alectra
shoots per plant is presented in Figure 2.
The genotypes Vuli AR1 with strain 1 and B301
with train 3 had the lowest number of Alectra shoots. At 35 DAP, more Alectra shoots per plant were
observed in Vuli-1 with strain 4, followed by Vuli
AR1 with strain 4 and lowest in three different genotypes which are B 301 with
strain 1 and 3, Vuli AR1 with strain 1 and 3 and Vuli AR2 with strain 2. At 49 DAP, Vuli-1 with strain 1, 2,
3 and 4, recorded more Alectra
shoots per plant followed by Mkanakaufiti with strain
3. At 63 DAP, Vuli-1 had the highest number of Alectra shoots per plant in all
strains, followed by Mkanakaufiti and the lowest was
in B 301.
The plant height
of cowpea was significantly affected by the different Alectra
strains (Table 2). The tallest plant was observed in strain 3, followed by
strain 4 and shortest plant was observed in strain 1. With respect to genotypic effects, tallest
plants were observed in Vuli AR1, followed by Mkanakaufiti and the shortest plants were in Vuli 1.
Genotypes did not differ significantly in
number of leaves per plant (Table 3). However, Vuli
AR 1 had numerically higher number of leaves per plant compared with the other
genotypes. This characteristic is important for the genotype if its leaves are
used as vegetables and also can be used as livestock feed during the dry season
of the semi-arid areas when fodders are scarce. The genotype Vuli AR1 also recorded the highest mean leaf area index
(LAI) whilist the lowest leaf area index was recorded
in genotype Vuli- 1. Varietal differences among the
cowpea genotypes or differences in anatomical, morphological and physiological
features affect the leaf area resulting to differences on leaf area index of
the genotypes (Onyishi et al., 2013).
The results also indicated that there were
significant differences (P<0.05) in days to 50% flowering among the
genotypes whereas the earliest days to 50% flowering plants were recorded in
genotype B 301, followed by Vuli AR1 while the latest
were observed in Mkanakaufiti. The genotype Mkanakaufiti was also recorded as a late flowering on set genotype. The delayed onset of flowering in
cowpea genotypes due to Alectra infestation,
can lead to reduction in number of flowers, number of pods, weight of pods and
seeds (Alonge et al. 2001). The
number of seeds per pod was affected by of A.vogelii, and Mkanakaufiti genotype produced the least number of seeds
per pod. The genotype B 301 attained 50% flowering earlier and the latest was Mkanakaufiti (Table 2). The mean value to 50% flowering in
B 301 was 46.58 DAP. The days to 50% flowering as reported by Ishayaku andSingh (2003), on two
cultivars of cowpea were 31 and 38 days. Thus, different host genes might
result to differential maturity periods. The attribute of flowering is
controlled by a single dominant gene in cowpea (Ishayaku
& Singh 2003). The difference in the genotypes on time to flowering varies
depending on the environmental factors like temperature, altitude, soil
conditions, and photoperiod during the period for growth and development. Time
to 50% flowering determines the maturing period of genotypes (Table 2). Thus, the days to 50% flowering provides an
opportunity for selection of earliness on different cowpea genotypes. Earliness
is an important trait as it facilitates mechanism of Alectra resistance through escape
from Alectra
and may enable selection for planting in Alectra infested areas. The earlier the genotype flowers, the earlier the
physiological maturity is reached. But, the
earliness character (days to flowering, pod filling and days to physiological
maturity enables B 301 to flower, pod fill and mature early and therefore
escape the effect of A. vogelii. The genotype B301 gave higher seed
yield and this was attributed by its resistance to A. vogelii. Seed yield is the major
universal breeding objective of the cowpea crop (Oladejo
et al. 2011), being representing the final product from physiological and
developmental process, which occur from time of sowing
to maturity.
All four strains showed no significant
differences (P >0.05), on seed yield and yield components (Table 3), however,
significant differences between genotypes were observed for 100 seed weight,
seed per pod, pod length and pod weight. The direct effect of A. vogelii is to reduce
leaf area and photosynthetic activity which in turn reduces number of pods,
number of seeds per pod, pod weight and seed yield in cowpea (Alonge et al.,
2001; Zitta et al., 2014).
Increasing major
components of seed yield such as pods per plant, pod length, weight of pods,
seeds per pod and 100 seeds weight, allows improving cowpea yield potential (Makanur et al., 2013). Similarly, Alonge
et al. (2001) found that Alectra reduced
number of pods per plant, pod weight, number of seeds per pod and seed yield in
cowpea. The effect of genotypes showed that, the genotype Vuli ARI produced the longest pods per plant, followed by
B301 and the shortest pods was recorded in Vuli AR2.
The pod length is a genotypic characteristic. This implies that if the genotype
has longer pod length, the seeds within the pods become widely spaced, compared
to the genotypes with short pods. The character for long pod length is
important in crop improvement because the longer pods more space is provided
for seeds (Onyishi et al., 2013).
There were significant differences among
genotypes on number of seeds per pod and in 100-seed weight. The genotype B 301
produced the highest number of seeds per pod, followed by Vuli
1 and a fewer seeds per pod were produced by Mkanakaufiti.
The genotype Vuli AR2 recorded the highest 100 seed
weight thus high seed size, whereas the genotype B301 has very small seeds (Hela et al., 2013),
and consequently the reason for its lowest 100- seeds weight.
The effect of
genotypes showed that, the highest mean values for 100 seeds weight
were produced by Vuli AR 2, followed by Vuli AR 1, and the lowest was B 301. There was no significant difference in seed
yield per plant between genotypes. Seed yield is an important trait in plants
because it is the final aggregate product of many interwoven physiological,
biochemical and development traits controlled by different arrays of genes. In
order to achieve high seed yield, understanding traits of yield components is
paramount (Oladejo et al., 2011). The genotype Vuli-1
was able to produce high seed yield per plant under A. vogelii infestation, so it can be
considered as being tolerant to Alectra.
Among the types of resistance, tolerance is considered as a type of horizontal
resistance which is polygenic in contrast to vertical resistance which is
monogenic (Kwaga et al., 2010). Normally, the
horizontal resistance has co-existence between the host and the parasite and it
is more sustainable than vertical resistance which breaks down faster with time
(Kwaga et al., 2010).
Despite high parasitism of A. vogelii, the tolerant genotypes produce high yield, which
implies they are efficient in the production of, assimilates to give high
yields and in turn support the parasites (Kwaga et
al., 2010). The genotype B 301 proved to be the best genotype for resistance
against A. vogelii.
It should be used as donor parent to provide the desirable traits to a
recipient. Consequently, in order to achieve higher yields, the use of
resistant varieties and controlling the weed with other management strategies
should be practiced.
CONCLUSIONS
Strains
of A. vogelii
did not differ significantly on all the studied variables. Significant
genotypic effects were evident for the entire variables studied except for pods
per plant and yield per plant. Significant interaction between genotypes and
strains was evident for leaf area index and number of shoots at 63 days after
planting. A. vogelii
had significant effect on pod length, number of seeds per pod, 100 seed weight
however there was no significant effect on total yield per plant. There was variation in number of days to emergence of A. vogelii among cowpea
genotypes. Genotype B 301 a resistant
genotype showed that, strain 3 reduces its number of pods per plant. The study
showed that, each strain responds differently to each cowpea genotype. Apart
from the effects of A. vogelii, the differences in performance are even due to
cowpea inherent genetic differences. Venture requires to
develop a genotype and extensively testing it across a wider geographic
area using many populations of Alectra. This will ensure stability and durability of the
variety without easy breakdown once it is moved to another area with more
virulent strains. The combination of different resistance mechanisms into a
single cultivar will provide durable outcome of the resistance in the field.
This can be achieved by pyramiding resistant genes in cowpea using existing
molecular markers.
ACKNOWLEDGEMENT
This work was part of MSc Dissertation by the
first author at Sokoine University of Agriculture
(SUA) in Morogoro, Tanzania. Financial support to one
of us FMR by McKnight Foundation through TARI-Ilonga
to carry out this study is highly appreciated.
CONFLICT OF INTEREST
Authors have no any contending effect on that
work
REFERENCES
Abdou,
I. R., Adamou, M., Karimou, I., Mahamane,
A. S., Adam, T., & Moumouni, S. (2017).
Evaluation of cowpea lines on natural infested field of Striga gesnerioides in Sahel Sudan of Southern
Niger. Agricultural Science Research Journal, 7(2), 35-41.
Adeigbe,
O. O., Adewale, B. D., & Fawole,
I. O. (2011). Genetic variability, stability and
relationship among some Cowpea, Vigna unguiculata (L.) Walp breeding lines. Plant Breed. Crop Science, 3(9), 203-208.
Alonge,
S. O., Lagoke, S. T. O. & Ajakaiye,
C. O. (2001). Cowpea reactions to Alectra
vogelii: effect on growth. Crop Protection,
20(4), 291-296.
Animasaun, D. A., Oyedeji, S., Azeez, Y. K.,
Mustapha, O. T. & Azeez, M. A. (2015). Genetic Variability Study among Ten Cultivars of Cowpea (Vigna unguiculata
L. Walp) Using Morpho-agronomic
Traits and Nutritional Composition. Journal of Agricultural Science, 10 (2), 119 – 130.
Atera,
A. U., Ishi, T., Onyango,
J. C., Kazuyuki, I. & Azuma, T. (2013). Striga infestation in Kenya:
Status, Distribution and Management Options. Journal of Sustainable
Agriculture, 2 (2), 01-09.
Ddungu,
S. P., Ekere, W., Bisikwa,
J., Kawooya, R., Okello, K.
D. & Biruma, M. (2015). Marketing and market integration of cowpea (Vigna Unguiculata L.Walp)
in Uganda. Journal of Development and Agricultural Economics, 7 (1): 1-11.
Gnanamanickam, S. S., Priyadarisini, V. B., Narayanan, N. N., Vasudeven,
P. & Kavitha, S. (1999). An
overview of bacterial blight in rice. Current Science, 77(11),
1435-1444.
Hella, J. P., Chilongo, T., Mbwaga,
A. M., Bokosi, J., Kabambe,
V., Riches, C. & Massawe, C. L. (2013). Participatory market-led cowpea breeding Sub Saharan Africa: Evidence
pathway from Malawi and Tanzania. Merit
Research Journal of Agricultural Science and Soil Sciences, 1(2),
011-018.
Horn, L., Shimelis, H., &
Laing, M. (2015). Participatory appraisal of production
constraints, preferred traits and farming system of cowpea in the northern
Namibia: implications for breeding. Legume Research, 38(5), 691-700.
Ishayaku, M. F. & Singh, B. B. (2003). Genetics of juvenile phase
of cowpea (Vigna unguiculata
(L) Walp.). Journal of
Food, Agriculture and Environment, 1,133-136.
Kabambe,
V., Katanga, L., Kapewa, T. & Ngwira,
A. R. (2008). Screening legumes for integrated
management of witchweeds (Alectra vogelii and Striga asiatica) in Malawi. African
Journal of Agricultural Research, 3(10), 706-713.
Kabambe,
V. H., Tembo, Y. L. B. & Kazira,
E. (2013). Awareness of the Parasitic Weed Alectra vogelii (Benth.) Amongst Extension Officers
in Three Districts in Malawi. American
Journal of Experimental Agriculture, 3(2), 432-442.
Karanja,
J., Nguluu, S. & Gatheru,
M. (2012). Farm yard manure reduces the virulence of Alectra vogelii (Benth) on cowpea (Vigna unguiculata). African
Journal of Plant Science, 6(3), 130-136.
Kutama, A. S., Hayatu, M. & Umar, S. (2014). Effect
of Alectra vogelii (Benth) Infestation on the Growth of some Genotypes of
Cowpea (Vigna unguiculata [L] Walp). Standard Research Journal of Agricultural Sciences, 2(3), 033- 039.
Kwaga, Y.M. (2014). Poultry Droppings
ameliorate infection of Alectra vogelii (Benth) in Groundnut (Arachis hypogaea L.) in Mubi, in Semi –
Arid Ecology of Nigeria. The
International Journal of Engineering and Science, 3(2), 74 – 78.
Kwaga,
Y. M., Olussoji, O., Tanimu,
B., Shebayan, J. & Lagoke,
S. T. O. (2010). Genotype Effect on the reaction of groundnut to Alectra
vogelii (Benth.)
infestation in a Sub-humid Environment. Journal of American
Science, 6(10), 01-08.
Lado, A., Hussaini, M. A.
& Kamara, A. Y. (2016). Molecular Characterization of Imazaquin
Tolerant and Sensitive Cowpea Genotypes. International
Journal of Genetics and Genomics, 4(2), 5-10.
Magashi,
A. I., Fulani, M. S. & Ibrahim, M. (2014). Evaluation
of Cowpea Genotypes (Vigna Unguiculata (L.)
Walp) for
some Yield and Root Parameters and their Usage in Breeding Programme
for Drought Tolerance. International Journal of Advances in Agricultural and Environmental
Engineering, 1(1), 2349-1531.
Makanur, B., Deshpande, V. K. & Vyakaranahal,
B. S. (2013). Characterization of cowpea genotypes based on quantitative
descriptors. An
international quarterly journal of life sciences, 8(4), 1183-1188.
Mbega,
E. R., Massawe, C. R. & Mbwaga,
A. M. (2016). Alectra vogelii, a
Threat to Bambara Groundnut Production in Singida and
Dodoma Regions, Tanzania. SCIENCEDOMAIN
international, 7(5), 1-8.
Mbwando,
A., Lungu, D. M., Tryphone,
G. M. & Tembo, L. (2016). Nature of resistance of cowpea Alectra vogelii infestation. African Crop Science Journal, 24(4), 389
– 395.
Njekete,
C., Midzi, J., Ncube, B.
& Madanzi, T. (2017). Response of Alectra vogelii Benth to Different Crop Root Exudates. International Journal of Plant and Soil
Science, 15(4), 1-12.
Okonya,
J. S. & Maass, B. L. (2014). Protein
and iron composition of cowpea leaves: an evaluation of six cowpea varieties
grown in eastern Africa. AJFAND. 14 (15), 01-12.
Oladejo AS, Akinwale RO, Obisesan IO. 2011. Interrelationships between grain yield
and other physiological traits of cowpea cultivars. African
Crop Science Journal, 19(3), 189-200.
Olasupo, F. O., Ilori,
C. O., Forster, B. P. & Bado, S. (2016). Mutagenic
Effects of Gamma Radiation on Eight Accessions of Cowpea (Vigna unguiculata [L.] Walp.). African Journal
of Plant Sciences, 7, 339-351.
Omoigui, L. O., Kamara, A. Y., Ishiyaku,
M. F. & Boukar, O. (2012). Comparative responses of cowpea breeding lines to Striga and Alectra
in the dry savanna of northeast Nigeria. African Journal of Agricultural Research, 7(5), 747-754.
Onyishi, G. C., Harriman, J. C., Ngwuta,
A. A., Okporie, E. O. & Chukwu,
S. C. (2013).
Efficacy of Some Cowpea Genotypes against Major Insect Pests in
Southeastern Agro-Ecology of Nigeria. Middle-East Journal of Scientific Research, 15(1), 114-121.
Silva, A. C., da Costa Santos, D., Junior, D. L. T., da
Silva, P. B., dos Santos, R. C. & Siviero, A.
(2018). Cowpea: A strategic legume species for food security and health. In Legume Seed Nutraceutical Research.
IntechOpen publication
Shiringan,
R. P. & Shimelis, H. A. (2011). Yield
response and stability among cowpea genotypes at three planting dates and test
environments. African Journal of Agricultural Research, 6(14), 3259-3263.
Thio,
I. G., Zida, E. P., Sawadogo,
M. & Sérémé, P. (2016). Current
status of Colletotrichum capsici
strains, causal agents of brown blotch disease of cowpea in Burkina Faso. African
Journal of Biotechnology, 15(5), 96-104
Ugbaa, M., Omoigui, L. & Bello, L. (2020).
Phenotypic screening of cowpea (Vigna unguiculata)
genotypes in
response to parasitic weed Alectra vogelii. Asian Journal of Agriculture, 4(1):
14-17
Zitta,
C., Magani, E. I. & Ahom,
R. I. (2014). Screening of cowpea genotypes for their reactions to Alectra
vogelii (Benth.) in the
Southern Guinea Savanna of Nigeria. Journal of Natural Sciences Research,
4(15), 2224-3186.