Greener Journal of Agricultural Sciences
ISSN: 2276-7770
Vol. 16(1), pp. 37-42, 2026
Copyright ©2026, Creative Commons Attribution 4.0 International.

|
Greener Journal of Agricultural Sciences ISSN: 2276-7770 Vol. 16(1), pp. 37-42, 2026 Copyright ©2026, Creative Commons Attribution 4.0 International. |
|
Effect of Supplemental Lighting and Elicitors on Antioxidant-Related Metabolites of Artemisia annua
*Agada O.O1, Onoja E.A2, and Ashiru Tukur1
1 School of Agriculture, Federal Polytechnic, Kaura Namoda; 2School of Science and Technology, Federal Polytechnic, Kaura Namoda
|
ABSTRACT |
|||
|
The use of Artemisinin in the treatment of Malaria has shown great promise. However, the low level of the metabolite produced by the plant remains a major drawback. This work examined the use of supplemental lighting and elicitors as a strategy to enhance artemisinin biosynthesis. The work was conducted at the School of Agricultural Technology, Federal Polytechnic, Kaura Namoda. The experimental design was a split plot design with supplemental lighting, with two levels, normal lighting (12 hours) and supplemental lighting, using artificial grow light to extend lighting to 16 hours, in the main plot and three levels of elicitors, (Moringa leaf extract, Ascorbic acid, and no elicitor) in the sub-plot, with each treatment replicated three times. The work was set up using 8*10-inch grow bags. At harvest, whole plant samples were sun-dried, ground, and analyzed for artemisinin content, total phenol, total flavonoid, and ferric reducing antioxidant power (FRAP). The results showed that the artemisinin level was Below Detection Limit (BDL), supplemental lighting with ascorbic acid elicitor consistently showed higher values for FRAP (86.84 %) and flavonoid (7.7 %), while supplemental lighting without elicitor showed higher results for phenolic content (0.68 mg/g).
|
|||
|
ARTICLE’S INFO |
|||
|
Article No.: 030226031 Type: Research Full Text: PDF, PHP, HTML, EPUB, MP3 DOI: 10.15580/gjas.2026.1.030226031
Accepted: 04/03/2026 Published: 23/03/2026
Keywords: Artemisinin, elicitors, metabolites
|
*Corresponding Author
Agada O.O
E-mail: steveagada004@gmail.com |
Article’s QR code
|
|
|
|
|
|
|
INTRODUCTION
With about 27 % malaria burden and an estimated 68 million reported cases, resulting in 194,000 deaths in 2021, Nigeria ranks as the country with the highest burden of malaria globally, making it a major public health concern (Shekarau et al. 2024). In response, the Nigerian government, through the National Malaria Elimination Program (NMEP), envisioned a strategic plan, with a five-year (2021-2025) accomplishment time frame, to reduce malaria morbidity to below 10 % parasite prevalence and less than 5 % malaria-attributable mortality. It is posited that malaria eradication would represent a major public health achievement, which would not only reduce human suffering and save lives but also boost economies. WHO has put the cost of eradicating malaria globally at $34 billion, but noted that the benefit-to-cost ratio is estimated to be about 8:1. This represents an unassailable attraction to pursuing its eradication.
Drugs represent an important member of the cocktail of tools being deployed in the fight against malaria. Among the first-line drugs recommended for use are artemisinin (ARS) and its derivatives (Poulson & Thai, 2015). Artemisinin is an endoperoxide-bridged sesquiterpene lactone synthesized in glandular trichomes of Artemisia annua L. The need for artemisinin use becomes critical because it represents one of the best replacement drugs for Chloroquine, which has been rendered ineffective by increased resistance.
A major problem with the use of artemisinin, however, is the low level of the substance produced by the plant (Namuli et al., 2018). It has been shown that under natural and unassisted growth, the plant produces between 0.01 % - 1 % on a dry weight basis (Zhang et al., 2023)which is considered too low for commercial extraction. Poulson & Thai, (2015) have reported that, whereas synthetic production is possible, the resulting product is not equally as valuable since there is evidence that other compounds found in A. annua work synergistically with artemisinin for enhanced efficacy against malaria (Ferreira et al., 2010) and cancer (Ivanescu et al., 2015), making it pertinent to find means of enhancing in-plant production.
Researchers have deployed a variety of approaches aimed at increasing the productivity of A. annua. These include transgenic, breeding, controlled lighting, bio-stimulants, and a gamut of other elicitors. (Widiyastuti & Subositi, 2019) reported positive effects of manipulating photoperiod on artemisinin yield when fluorescent lights (32 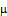 E · s ‒1 per m2) were used to extend the photoperiod to 16 hours. A similar result was observed by (Sankhuan et al., 2022) When A. annua was grown under light-emitting diode (LED). Zhang et al. (2023), however, found no significant effect of supplemental light treatment on the secondary metabolite productivity of A. annua. (Liu et al., 2023) asserted that the enhancement of artemisinin by phytohormones such as Jasmonic acid was modulated by light and, as such, made a case for the integration of light and phytohormones for effective regulation of its biosynthesis.
E · s ‒1 per m2) were used to extend the photoperiod to 16 hours. A similar result was observed by (Sankhuan et al., 2022) When A. annua was grown under light-emitting diode (LED). Zhang et al. (2023), however, found no significant effect of supplemental light treatment on the secondary metabolite productivity of A. annua. (Liu et al., 2023) asserted that the enhancement of artemisinin by phytohormones such as Jasmonic acid was modulated by light and, as such, made a case for the integration of light and phytohormones for effective regulation of its biosynthesis.
General objective
This research is aimed at evaluating the effect of supplemental lighting and phytohormone elicitors on the biosynthesis of artemisinin and other secondary metabolites.
Specific objectives
1. To determine the effect of supplemental lighting, ascorbic acid, and moringa leaf extract application on artemisinin productivity
2. To ascertain the effect of supplemental lighting, ascorbic acid, and moringa leaf extract application on total phenol, flavonoid, and ferric reducing antioxidant power content of Artemisia annua
3. To assess the modulating effect of supplemental lighting on foliar-applied ascorbic acid and moringa leaf extract
MATERIALS AND METHODS
The research was set up at the School of Agriculture, Federal Polytechnic, Kaura Namoda with average night and day time temperature within the experimental period ranging between 23 to 26 ⁰C and 37 to 42 ⁰C respectively. Seedlings for the work were purchased from the Institute of Agricultural Research, ABU Zaria, Nigeria. The seedlings were transferred into 8*10-inch grow bags and allowed to stabilize for two weeks before treatment application. Treatments consisted of supplemental lighting (S) and normal lighting (N), elicitor 1 (moringa leaf extract), elicitor 2 (ascorbic acid), and no elicitor. For the supplemental lighting group, plants were subjected to 4 hours of artificial lighting at night, using full-spectrum LED grow light. The elicitors were sprayed once, at 6 weeks after transplanting, using one molar solution of ascorbic acid and 2.2 % moringa leaf extract. Water was used as the control. The experiment was set up in a split plot design, with supplemental lighting in the main plot and elicitors in the sub plot and replicated three times. At harvest whole plants were harvested and sun dried. These were then ground using a mortar and sieved using a 2 mm sieve.
Determination of artemisinin
For artemisinin determination, the sample was extracted using Ultrasound Assisted Extraction (UAE) method. Briefly, 0.75 g of the sample was extracted with 40 mL of pet ether using a sonicator for 30 minutes. Then the n-hexane phase was filtered through a glass membrane filter (0.45µm) and washed with 80 mL of 2% NaOH solution in a separating funnel to remove NaOH-soluble impurities. After phase separation, the alkali solution present in the lower layer was drained, leaving the upper solution. This was then washed with an equal volume of distilled water, five times to neutralize it. 3 % activated charcoal was then added to remove colored pigments. The resulting solution was distilled to obtain the final extract. The extract was then dried, dissolved in 1 ml of methanol, and stored at 40⁰C.
For artemisinin determination, 1mL of the extract (or standard artemisinin) was transferred into an amber-colored, air-tight bottle, and 4 mL of 0.2% NaOH added. This was maintained at 50 °C for 30 minutes in a preheated water bath. This was allowed to cool to room temperature. Thereafter, 5mL of 0.08 mol/L acetic acid was added to make up the final volume to 10mL for HPLC quantification.
Determination of Total Phenolic Content
Total phenolic content was determined by the Folin-Ciocalteu method as described by (Ainsworth, E. A., & Gillespie, 2007).
Determination of Total flavonoid content
Total flavonoid quantification followed the modified aluminum chloride method of (Yamin et al., 2021).
Determination of ferric reducing antioxidant power
The ferric-reducing antioxidant power of the crude methanolic extract of A. annua will be determined using the potassium ferrocyanide-ferric chloride method. Two millimetres of the extract (10mg/g) will be added to 2.5 mL of potassium ferrocyanide and then incubated at 50 °C for 20 min. About 2.5 mL of trichloroacetic acid will be added to the mixture and then centrifuged at 650x g for 10 min. To the supernatant (2.5 mL), distilled water (2.5 mL), and 0.5 mL ferric chloride will be added (Oyaizu, 1986). The absorbance will then be read at 700 nm. Higher absorbance indicates greater reducing capacity, which is calculated as follows:
Reducing power =
where, AM is absorbance of reaction mixture, AC is absorbance of control mixture (Distilled water)
Statistical Analysis results were presented as means ± of standard deviations of triplicate measurements. Data were analyzed with ANOVA procedure using SPSS version 20. Treatment means were compared using DUNCAN LSD at p≤0.05.
RESULTS
Artemisinin content
Results of analysis indicated that the artemisinin content of the samples were generally below detection limit (BDL) (see sequence summary report attached as appendix 1).
Ferric Reducing Antioxidant Power (FRAP) Assay
The Ferric Reducing Antioxidant Power (FRAP) assay results is presented in Figure 1. It demonstrated a concentration-dependent increase in antioxidant activity across all samples evaluated (0.2–1.0 mg/mL) (Fig. 1). At the lowest concentration (0.2 mg/mL), antioxidant activity ranged from 16.82% (NE1) to 54.69% (SE2). NE2 (54.30%) and SE2 (54.69%) exhibited comparatively higher reducing power at this concentration, while NE0 (20.26%) and NE1 (16.82%) showed relatively lower activity. At 0.4 mg/mL, %FRAP values increased across all samples, with SE2 showing a marked rise to 77.34%, followed by SE1 (59.13%) and NE2 (56.30%). NE1 remained comparatively lower (27.70%), although still demonstrating improvement relative to 0.2 mg/mL. Further increases were observed at 0.6 mg/mL and 0.8 mg/mL. At 0.8 mg/mL, NE0 (85.62%) and SE2 (81.67%) displayed notably high antioxidant activity, while SE1 (75.90%) and NE2 (78.12%) also exhibited strong reducing capacity. NE1 showed moderate activity (44.36%) at this concentration. At the highest concentration tested (1.0 mg/mL), all samples exhibited their maximum reducing power. NE0 recorded the highest %FRAP value (96.66%), followed by SE2 (86.84%), NE2 (83.89%), SE0 (84.73%), and SE1 (81.39%). NE1 showed the lowest activity among the samples at this concentration (50.49%), though still demonstrating a clear dose-dependent increase. Among the samples, NE0 and SE2 consistently exhibited superior reducing power, particularly at higher concentrations, suggesting a stronger antioxidant potential. In contrast, NE1 showed comparatively lower reducing capacity across all concentrations tested. These findings confirm that the extracts possess appreciable antioxidant properties, with variation in potency depending on sample type and concentration..
Fig. 1. Ferric Reducing Anti-Oxidant Power (FRAP) of Artemisia annua whole plant sample as affected by supplemental lighting and elicitors.
Note: NE1: normal lighting with moringa leaf extracts; NE2: normal lighting with ascorbic acid; NE0: normal lighting without elicitor; SE1: supplemental lighting with moringa leaf extracts; SE2: supplemental lighting with ascorbic acid; SE0: supplemental lighting without elicitor. Bars are standard error of means.
Phenolic Content
Results of analysis of total phenolic content of Artemisia annua dried sample is presented in Table 1. The result indicated that the mean phenolic content varied significantly (p<0.05) across groups, with SE0 recording the highest mean value (0.6833 ± 0.00416), followed by SE1 (0.5703 ± 0.00252), NE0 (0.5417 ± 0.00321), NE2 (0.5057 ± 0.00551), SE2 (0.4747 ± 0.00153), and NE1 (0.3357 ± 0.02136). Overall, SE0 demonstrated the highest phenolic concentration, while NE1 exhibited the lowest.
Table 1: Phenolic content of Artemisia annua whole plant samples as affected by supplemental lighting and elicitors.
|
Group |
Phenolic Content (mg/g) |
|
NE1 |
0.336 ± 0.021ᵃ |
|
SE2 |
0.475 ± 0.002ᵇ |
|
NE2 |
0.506 ± 0.006ᶜ |
|
NE0 |
0.542 ± 0.003ᵈ |
|
SE1 |
0.570 ± 0.003ᵉ |
|
SE0 |
0.683 ± 0.004ᶠ |
Values are expressed as mean ± standard deviation (n = 3). Means with different superscript letters within the column differ significantly at p < 0.05 (Duncan/LSD post hoc test).
Note: NE1: normal lighting with moringa leaf extracts; NE2: normal lighting with ascorbic acid; NE0: normal lighting without elicitor; SE1: supplemental lighting with moringa leaf extracts; SE2: supplemental lighting with ascorbic acid; SE0: supplemental lighting without elicitor
Flavonoid Content
Presented in Table 2 is the result of analysis of the effect of supplemental lighting and elicitors on the total flavonoid content of the whole plant sample of Artemisia annua. The results showed significant (p<0.05) variation in flavonoid content among the groups. SE2 had the highest mean flavonoid content (7.7483 ± 0.00764), followed by NE0 (5.8950 ± 0.01500), SE0 (5.5950 ± 0.03122), SE1 (4.7367 ± 0.01258), NE2 (3.8600 ± 0.01000), and NE1 (3.4383 ± 0.02082). SE2 consistently exhibited the highest flavonoid concentration, while NE1 recorded the lowest.
Table 2: Flavonoid content of Artemisia annua whole plant samples as affected by supplemental lighting and elicitors.
|
Group |
Flavonoid Content (%) |
|
NE1 |
3.438 ± 0.021ᵃ |
|
NE2 |
3.860 ± 0.010ᵇ |
|
SE1 |
4.737 ± 0.013ᶜ |
|
SE0 |
5.595 ± 0.031ᵈ |
|
NE0 |
5.895 ± 0.015ᵉ |
|
SE2 |
7.748 ± 0.008ᶠ |
Values are expressed as mean ± standard deviation (n = 3). Means with different superscript letters within the column differ significantly at p < 0.05 (Duncan/LSD post hoc test).
Note: NE1: normal lighting with moringa leaf extracts; NE2: normal lighting with ascorbic acid; NE0: normal lighting without elicitor; SE1: supplemental lighting with moringa leaf extracts; SE2: supplemental lighting with ascorbic acid; SE0: supplemental lighting without elicitor
DISCUSSIONS
The results of analysis indicated no significant effect of treatment on artemisinin content. Across all treatments, the artemisinin content was below detection limit (BDL). This result contrasted with that of Zhang et al (2023), who found that supplemental lighting enhanced artemisinin biosynthesis and (Jeyasri et al., 2023) who reported the positive effect of elicitors on the biosynthesis of bioactive compounds. Liu et al. (2023) demonstrated that supplemental lighting complemented with elicitors enhanced artemisinin content in A. annua. The observation of BDL in this study may, however, not be attributable to a lack of response to the treatment, since the control was similarly affected; rather, this effect may be due to environmental impact. Artemisinin productivity is highly susceptible to environmental variability. (Thi Tuong Thu et al., 2011) had reported that growing temperatures above 26.2 ⁰C significantly reduced plant growth and artemisinin content of two clones of Artemisia annua. It is likely that the unusually high temperatures (over 40 ⁰C) that characterized the growing season may have hampered growth and overall artemisinin biosynthesis.
Ferric reducing antioxidant power, Phenolic and flavonoid content were significantly enhanced by treatment with supplemental lighting. This accords with the findings of Sankhuan et al. (2022) and (Rai et al., 2024), who reported that exposure to various light treatments induced significant changes in a wide range of attributes of A. annua, including phenolic and flavonoid contents. However, it was observed that coupling supplemental lighting with elicitors was beneficial only in the enhancement of FRAP and flavonoid content, as phenolic content indicated no advantage in combining the treatments.
CONCLUSIONS
Considering the possibility of environmental limitations to growth and metabolite productivity, any trial involving A. annua in the dry savannah is probably best conducted in the colder harmattan season. The effect of supplemental lighting on metabolite productivity is however encouraging and should be further evaluated.
Funding
This work was funded by the Nigerian Government, through the Tertiary Education Trust Fund, Institutional-Based Research funding program.
Conflict of Interest
The authors declare no conflict of interest.
REFERENCES
Ainsworth, E. A., & Gillespie, K. M. (2007). Estimation of total phenolic content and other oxidation substances in plant tissues using Folin-ciocalteu reagent. Nature Protocol, 2(4), 875–877.
Ferreira, J. F. S., Luthria, D. L., Sasaki, T., & Heyerick, A. (2010). Flavonoids from Artemisia annua L. As antioxidants and their potential synergism with artemisinin against malaria and cancer. In Molecules (Vol. 15, Number 5, pp. 3135–3170). https://doi.org/10.3390/molecules15053135
Ivanescu, B., Miron, A., & Corciova, A. (2015). Sesquiterpene Lactones from Artemisia Genus: Biological Activities and Methods of Analysis. In Journal of Analytical Methods in Chemistry (Vol. 2015). Hindawi Publishing Corporation. https://doi.org/10.1155/2015/247685
Jeyasri, R., Muthuramalingam, P., Karthick, K., Shin, H., Choi, S. H., & Ramesh, M. (2023). Methyl jasmonate and salicylic acid as powerful elicitors for enhancing the production of secondary metabolites in medicinal plants: an updated review. In Plant Cell, Tissue and Organ Culture (Vol. 153, Number 3, pp. 447–458). Springer Science and Business Media B.V. https://doi.org/10.1007/s11240-023-02485-8
Liu, H., Li, L., Fu, X., Li, Y., Chen, T., Qin, W., Yan, X., Wu, Z., Xie, L., Kayani, S. llyas, Hassani, D., Sun, X., & Tang, K. (2023). AaMYB108 is the core factor integrating light and jasmonic acid signaling to regulate artemisinin biosynthesis in Artemisia annua. New Phytologist, 237(6), 2224–2237. https://doi.org/10.1111/nph.18702
Namuli, A., Bazira, J., Casim, T. U., & Engeu, P. O. (2018). A review of various efforts to increase artemisinin and other antimalarial compounds in Artemisia Annua L plant . Cogent Biology, 4(1), 1513312. https://doi.org/10.1080/23312025.2018.1513312
Oyaizu, M. (1986). Studies on Products of Browning Reactions: Antioxidative Activities of Product of Browning Reaction Prepared from Glucosamine. The Japanese Journal of Nutrition and Dietetics, 44, 307–315.
Poulson, M. E., & Thai, T. (2015). Effect of high light intensity on photoinhibition, oxyradicals and artemisinin content in Artemisia annua L. Photosynthetica, 53(3), 403–409. https://doi.org/10.1007/s11099-015-0130-5
Rai, N., Ansari, N., Apoora, Kumari, S., Singh, S., Saha, P., Bisen, M. S., & Pandeh-Rai, S. (2024). Red and Blue light-mediated Physiological and Metabolic insights Artemisia annua L. Industrial Crop and Products, 222(4).
Sankhuan, D., Niramolyanun, G., Kangwanrangsan, N., Nakano, M., & Supaibulwatana, K. (2022). Variation in terpenoids in leaves of Artemisia annua grown under different LED spectra resulting in diverse antimalarial activities against Plasmodium falciparum. BMC Plant Biology, 22(1). https://doi.org/10.1186/s12870-022-03528-6
Thi Tuong Thu, B., Van Minh, T., Peng Lim, B., & Lai Keng, C. (2011). Effects of Environmental Factors on Growth and Artemisinin Content of Artemisia annua L. In Tropical Life Sciences Research (Vol. 22, Number 2).
Widiyastuti, Y., & Subositi, D. (2019). Photoperiod effect on the growth and artemisinin content of Artemisia Annua grown in tropical region. AIP Conference Proceedings, 2099. https://doi.org/10.1063/1.5098432
Yamin, Ruslin, Mistriyani, Sabarudin, Ihsan, S., Armadany, F. I., Sahumena, M. H., & Fatimah, W. O. N. (2021). Determination of total phenolic and flavonoid contents of Jackfruit peel and in vitro antiradical test. Food Research, 5(1), 84–90. https://doi.org/10.26656/fr.2017.5(1).350
Zhang, N., Yang, H., Han, T., Kim, H. S., & Marcelis, L. F. M. (2023). Towards greenhouse cultivation of Artemisia annua: The application of LEDs in regulating plant growth and secondary metabolism. Frontiers in Plant Science, 13. https://doi.org/10.3389/fpls.2022.1099713
|
Cite this Article: Agada, OO, Onoja, EA, Ashiru, T (2026). Effect of Supplemental Lighting and Elicitors on Antioxidant-Related Metabolites of Artemisia annua. Greener Journal of Agricultural Sciences, 16(1): 37-42, https://doi.org/10.15580/gjas.2026.1.030226031.
|