Greener Journal of Biomedical and Health Sciences
Vol. 9(1), pp. 65-75, 2026
ISSN: 2672-4529
Copyright ©2026, Creative Commons Attribution 4.0 International.

|
Greener Journal of Biomedical and Health Sciences Vol. 9(1), pp. 65-75, 2026 ISSN: 2672-4529 Copyright ©2026, Creative Commons Attribution 4.0 International. |
|
Effect of Costus lucanusianus (Ukpete) Stem Sap on Hematological Parameters in Wistar Rats
Mandah, Emmanuel Samuel1, Asuzu-Samuel, H.O. 2; Ibubeleye, V.3
1 Department of Biomedical, School of Science Laboratory Technology, University of Port Harcourt, Nigeria.
2 Department of Biomedical, School of Science Laboratory Technology, University of Port Harcourt, Nigeria.
3 Department of Pharmacology, Faculty of Basic Clinical Sciences, College of Health Sciences, University of Port Harcourt, Nigeria.
|
ABSTRACT |
|||
|
This study evaluated the hematological effects of sub-chronic oral administration of Costus lucanusianus stem sap in Wistar rats. Twenty adult rats were randomly assigned into four groups: control, low dose (200 mg/kg), medium dose (400 mg/kg), and high dose (600 mg/kg), and treated daily for 28 days. Hematological parameters including red blood cell (RBC) count, hemoglobin concentration (Hb), packed cell volume (PCV), mean corpuscular indices, white blood cell (WBC) count, and platelet count were assessed using standard laboratory methods. Results showed no statistically significant differences (p > 0.05) between treated and control groups. The control group recorded Hb (11.62 ± 0.47 g/dL), PCV (37.32 ± 1.27%), and RBC (6.17 ± 0.15 ×10⁶/µL), with comparable values observed across all treatment groups. These findings indicate that C. lucanusianus stem sap does not adversely affect hematopoietic function under sub-chronic exposure. The plant appears hematologically safe at the tested doses, although further long-term studies are warranted.
|
|||
|
ARTICLE’S INFO |
|||
|
Article No.: 042426058 Type: Research Full Text: PDF, PHP, HTML, EPUB, MP3 DOI: 10.15580/gjbhs.2026.1.042426058
Accepted: 26/04/2026 Published: 28/04/2026
Keywords: Costus lucanusianus, hematological parameters, Wistar rats, stem sap, sub-chronic toxicity
|
*Corresponding Author
Mandah, Emmanuel Samuel
Email: emmanuel_mandah@uniport.edu.ng, emmanuelsamuelmandah@gmail.com
Tel: +2347031190796 |
Article’s QR code
|
|
|
|
|
|
|
1. INTRODUCTION
Medicinal plants continue to play a central role in primary healthcare delivery for a substantial proportion of the world’s population, particularly in low- and middle-income countries where access to conventional medical services is limited [1,2]. These plants provide affordable and culturally accepted remedies and remain important sources of bioactive compounds for drug discovery and development [3].
The genus Costus has attracted increasing scientific interest due to its diverse pharmacological properties, including antioxidant, anti-inflammatory, and antidiabetic activities [3,4]. However, Costus lucanusianus, although widely utilized in traditional African medicine for the treatment of fever, pain, digestive disorders, and wound healing, remains relatively underexplored in terms of its scientific validation [5,6]
Phytochemical constituents such as flavonoids, tannins, saponins, and alkaloids present in medicinal plants are known to exert both beneficial and adverse biological effects depending on dosage, duration of exposure, and mechanism of action [7,9]. Consequently, there is a need for systematic toxicological evaluation to substantiate the safety of such plants and their traditional applications.
Hematological parameters are widely recognized as sensitive indicators of physiological and pathological status, providing valuable information on the functional integrity of the hematopoietic system, immune response, and overall health condition [10]. In toxicological studies, sub-chronic exposure models, particularly 28-day repeated-dose studies conducted in accordance with established guidelines such as those of the Organization for Economic Co-operation and Development (OECD), are commonly employed to assess potential systemic effects and safety profiles of test substances [11]
Therefore, this study was designed to evaluate the hematological effects of sub-chronic administration of Costus lucanusianus stem sap in Wistar rats.

Plate 1: Fresh stem of Costus lucanusianus collected from Mgbere, Ibaa, Rivers State, Nigeria.
2. MATERIALS AND METHODS
2.1 Experimental Animals
Twenty (20) healthy adult Wistar rats weighing between 90 g and 156 g were used for this study. The animals were obtained from the Animal House of the Faculty of Pharmaceutical Sciences, University of Port Harcourt, Nigeria. Upon arrival, they were acclimatized for a period of two weeks under standard laboratory conditions to minimize stress and stabilize physiological functions.
The animals were housed in clean, well-ventilated cages under controlled environmental conditions, including a temperature of 25 ± 2°C, relative humidity of 60–80%, and a 12–14-hour light-dark cycle. Standard laboratory feed and clean drinking water were provided ad libitum. Animal handling and care were conducted in accordance with internationally accepted guidelines for laboratory animal use [2].
2.2 Experimental Grouping and Treatment Regimen
Following acclimatization, the animals were randomly assigned into four (4) experimental groups of five (n = 5) rats each to ensure even distribution of biological variability and minimize selection bias. The grouping was designed to evaluate the dose-dependent effects of fresh stem sap of Costus lucanusianus on hematological parameters.
Group I (Control): Received distilled water and served as the negative control.
Group II (Low Dose): Received 200 mg/kg body weight of fresh stem sap.
Group III (Medium Dose): Received 400 mg/kg body weight of fresh stem sap.
Group IV (High Dose): Received 600 mg/kg body weight of fresh stem sap.
The stem sap was freshly prepared and preserved under refrigeration prior to administration to maintain its phytochemical stability. Oral administration was carried out using a calibrated gavage to ensure accurate dosing and to simulate the traditional route of administration commonly reported in ethnomedicine.
Treatment was administered once daily for twenty-eight (28) consecutive days, representing a sub-chronic exposure model consistent with standard toxicological evaluation protocols [11,18].
Body weights were measured weekly using a calibrated digital balance, while animals were observed daily for clinical signs of toxicity, including changes in feeding behavior, locomotion, posture, fur condition, and general responsiveness. All observations were documented throughout the study period.
2.3 Preparation of Plant Material (Fresh Stem Sap)
Fresh, mature stems of Costus lucanusianus were collected from a forest reserve in Mgbere, Ibaa community, Emohua Local Government Area of Rivers State, Nigeria. The plant material was authenticated at the Herbarium of the Department of Plant Science and Biotechnology, University of Port Harcourt, where a voucher specimen (Accession No. 002; Specimen No. UPH/P/612) was deposited for future reference.
The stems were thoroughly washed to remove contaminants and cut into smaller sections. Fresh stem sap was obtained through mechanical expression using a mortar and pestle, followed by filtration to remove particulate matter. The filtrate was stored in sterile, airtight containers prior to administration.
2.4 Methods of Data Collection and Laboratory Analysis
At the end of the 28-day treatment period, the animals were fasted overnight and sacrificed under appropriate anesthesia to minimize pain and distress. Blood samples were collected via cardiac puncture using sterile syringes and dispensed into ethylenediaminetetraacetic acid (EDTA) tubes for hematological analysis.
Hematological Analysis
Hematological parameters assessed included hemoglobin concentration (Hb), packed cell volume (PCV), red blood cell count (RBC), white blood cell count (WBC), platelet count, differential leukocyte counts, and erythrocyte indices such as mean corpuscular volume (MCV), mean corpuscular hemoglobin (MCH), and mean corpuscular hemoglobin concentration (MCHC).
Analyses were conducted using an automated hematology analyzer to ensure accuracy and reproducibility, with manual verification methods applied where necessary. These procedures are widely accepted in clinical and experimental hematology [9,10].
Hematological indices serve as essential indicators of physiological status, immune competence, and potential toxicological effects of bioactive compounds [13].
2.5 Acute Toxicity Study (LD₅₀ Determination)
The acute oral toxicity of Costus lucanusianus stem sap was evaluated using Lorke’s method [14], a standard approach for determining the median lethal dose (LD₅₀).
The study was conducted in two phases:
 Phase I (Range-finding): Nine rats were divided into three groups and administered single oral doses of 10, 100, and 1000 mg/kg body weight, respectively.
Phase I (Range-finding): Nine rats were divided into three groups and administered single oral doses of 10, 100, and 1000 mg/kg body weight, respectively.
 Phase II (Dose refinement): Based on Phase I results, three rats were administered higher doses of 1600, 3000, and 5000 mg/kg body weight, respectively.
Phase II (Dose refinement): Based on Phase I results, three rats were administered higher doses of 1600, 3000, and 5000 mg/kg body weight, respectively.
Animals were observed for 24–72 hours for signs of toxicity, including behavioral and physiological changes such as tremors, salivation, altered locomotion, and respiratory distress.
No mortality or severe toxicity was recorded at any dose level. The LD₅₀ was estimated using Lorke’s formula:
LD₅₀ = √(D₀ × D₁₀₀)
where:
(D0) = highest dose producing no mortality,
(D100) = lowest dose producing 100% mortality.
Since no mortality occurred even at 5000 mg/kg, the LD₅₀ was considered to be greater than 5000 mg/kg, indicating low toxicity and a wide margin of safety in accordance with OECD classification criteria [11,14,18].
These findings suggest that the fresh stem sap of Costus lucanusianus possesses a wide margin of safety when administered orally in a single dose. However, it is important to note that acute toxicity studies provide only preliminary safety information, and further sub-chronic and chronic toxicity evaluations are necessary to fully establish the long-term safety profile of the plant.
2.6 Methods of Data Analysis
Data obtained were expressed as Mean ± Standard Deviation (Mean ± SD). Statistical analyses were performed using Microsoft Excel and IBM SPSS Statistics.
One-way Analysis of Variance (ANOVA) was used to compare mean values among groups, followed by appropriate post-hoc tests to identify significant differences. This statistical approach is widely applied in biomedical research for multi-group comparisons [15].
A significance level of p < 0.05 was considered statistically significant.
The statistical analysis therefore enabled the evaluation of the effects of varying doses of Costus lucanusianus stem juice on hematological parameters including hemoglobin concentration (Hb), packed cell volume (PCV), red blood cell count (RBC), white blood cell count (WBC), platelet count (PLT), and other erythrocyte indices.
2.7 Ethical Approval
Ethical approval for this study was obtained from the University of Port Harcourt Research Ethics Committee (UPREC) prior to the commencement of the experiment.
All procedures were conducted in accordance with international guidelines for the care and use of laboratory animals, including those outlined by the World Health Organization and OECD [2,11].
The principles of Replacement, Reduction, and Refinement (3Rs) were strictly adhered to in order to ensure humane treatment of the animals. Animals were monitored daily for signs of distress, and all invasive procedures were performed under appropriate anesthesia to minimize pain.
3. RESULTS
This chapter presents the results obtained from the evaluation of the effects of Costus lucanusianus stem sap on selected physiological and hematological parameters in Wistar albino rats following 28 days of oral administration. The data generated were analyzed using appropriate statistical tools and expressed as Mean ± Standard Deviation (Mean ± SD).
Results are presented using tables and graphical illustrations to enhance clarity, facilitate comparison, and support interpretation. The presentation follows a logical progression, beginning with general physiological observations (body weight changes), followed by hematological assessments.
Overall, sub-chronic administration of Costus lucanusianus stem sap did not produce statistically significant changes (p > 0.05) in the evaluated hematological parameters when compared with the control group, suggesting a lack of overt toxicity.
3.1 Effect of Costus lucanusianus Stem Sap on Body Weight
Body weight is a fundamental indicator in toxicological studies, reflecting general health status, metabolic efficiency, and potential toxic effects of administered substances [11,18].
In the present study, body weights were monitored weekly over a four-week period. The results are summarized in Table 3.1.
Table 3.1: Changes in Body Weight (Mean ± SD)
|
Group |
Week 1 |
Week 2 |
Week 3 |
Week 4 |
|
Control |
97.0 ± 4.36 |
101.0 ± 12.39 |
125.0 ± 1.87 |
126.0 ± 3.16 |
|
Low Dose (200 mg/kg) |
104.6 ± 10.48 |
107.0 ± 12.32 |
122.8 ± 11.53 |
126.6 ± 10.21 |
|
Medium Dose (400 mg/kg) |
120.2 ± 8.82 |
117.6 ± 7.67 |
132.6 ± 12.21 |
135.4 ± 11.82 |
|
High Dose (600 mg/kg) |
120.0 ± 11.58 |
124.2 ± 10.30 |
142.8 ± 8.51 |
146.6 ± 8.32 |
Body weight increased progressively across all experimental groups over the 28-day period as shown in Table 3.1 and Figure 3.1a and Figure 3.1b. No statistically significant differences (p > 0.05) were observed between control and treated groups, indicating absence of systemic toxicity [11].
Body Weight Trend
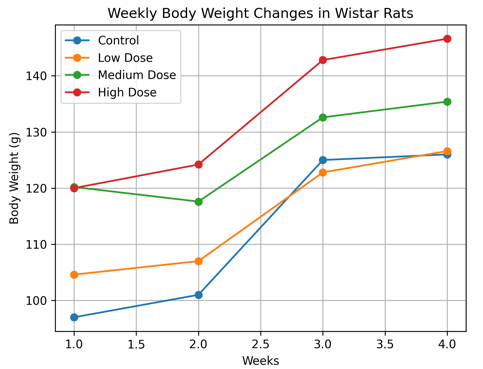
Figure 3.1a. Weekly body weight changes of Wistar rats following 28-day administration of Costus lucanusianus stem sap (Mean ± SD, n = 5).
The observed dose-related increase in body weight, although not statistically significant, may reflect normal physiological growth rather than a toxic effect of the plant extract, rather than toxicity.
Week 4 Comparison
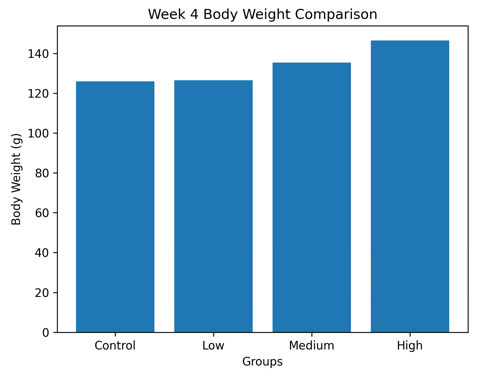
Figure 3.1b: Week 4 Comparison of the body weight changes of Wistar rats following 28-day oral administration of Costus lucanusianus stem sap. Values are expressed as Mean ± SD (n = 5).
The convergence of body weights at Week 4 without evidence of wasting or cachexia further confirms the non-toxic profile of the extract under prolonged exposure conditions.
Discussion of Body Weight Changes
A progressive increase in body weight was observed across all groups throughout the experimental period. The treatment groups, particularly the medium- and high-dose groups, consistently exhibited higher mean body weights compared to the control group.
Despite these increases, statistical analysis revealed no significant difference (p > 0.05), indicating that administration of Costus lucanusianus stem sap did not adversely affect growth or metabolic function.
The steady increase in body weight across all groups suggests normal physiological development and absence of systemic toxicity. Maintenance or increase in body weight is generally considered an indicator of good tolerability in repeated-dose studies [11,18].
The observed trend may also suggest improved feed utilization or mild metabolic stimulation, which could be attributed to bioactive phytochemicals present in the plant. However, since the differences were not statistically significant, these effects cannot be conclusively established.
3.2 Effect of Costus lucanusianus Stem Sap on Hematological Parameters
Hematological parameters are critical indicators of physiological status, immune function, and potential toxicological effects of xenobiotics [9].
3.2.1 Erythrocyte Parameters
Table 3.2: Erythrocyte Parameters (Mean ± SD)
|
Group |
Hb (g/dL) |
PCV (%) |
RBC (×10⁶/µL) |
|
Control |
11.62 ± 0.47 |
37.32 ± 1.27 |
6.17 ± 0.15 |
|
Low Dose |
11.84 ± 0.70 |
37.38 ± 1.41 |
6.15 ± 0.16 |
|
Medium Dose |
10.34 ± 2.83 |
32.92 ± 9.05 |
5.40 ± 1.39 |
|
High Dose |
12.00 ± 1.12 |
38.90 ± 3.36 |
6.38 ± 0.54 |
Hemoglobin (Hb) Mean ± SD Graph
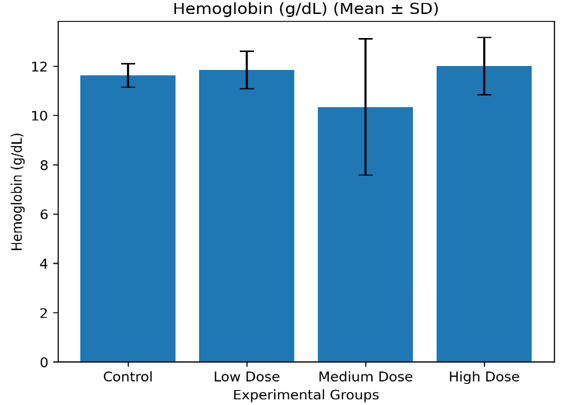
Figure 3.2.1a: Mean hemoglobin concentration (Mean ± SD) of Wistar albino rats administered Costus lucanusianus stem juice at different dose levels for 28 days.
The preservation of erythrocyte indices across all treatment groups suggests that the extract neither impairs bone marrow function nor induces oxidative erythrocyte damage.
Packed Cell Volume (PCV) Mean ± SD Graph
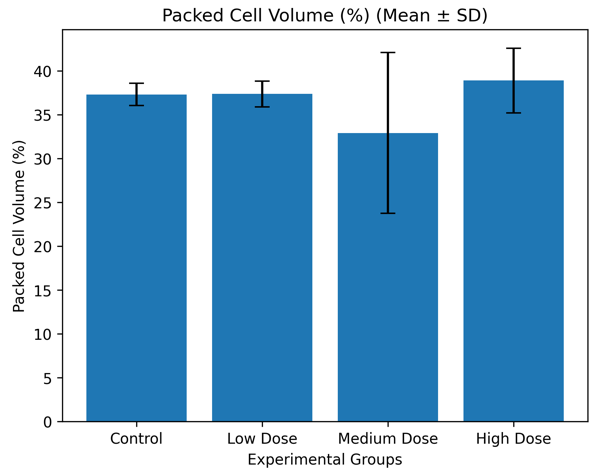
Figure 3.2.1b: Mean packed cell volume (Mean ± SD) of Wistar albino rats following administration of Costus lucanusianus stem juice for 28 days.
Red Blood Cell Count (RBC) Mean ± SD Graph 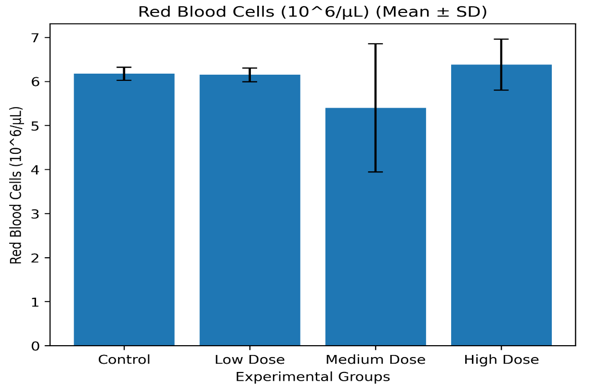
Figure 3.2.1c: Mean red blood cell count (Mean ± SD) of Wistar albino rats administered Costus lucanusianus stem juice at different doses for 28 days.
DISCUSSION
The results indicate that hemoglobin concentration, packed cell volume, and red blood cell count showed only minor variations across the groups, with no statistically significant differences (p > 0.05).
Although slight increases were observed in the low- and high-dose groups, all values were comparable to control. This suggests that the stem sap did not induce anemia, hemolysis, or suppression of erythropoiesis.
The stability of erythrocyte indices may be attributed to the antioxidant properties of phytochemicals such as flavonoids and phenolic compounds, which protect red blood cells from oxidative damage [7,16]
3.2.2 Leukocyte Parameters
Table 3.3: Leukocyte Parameters (Mean ± SD)
|
Group |
WBC (×10³/µL) |
Lymph (%) |
Gran (%) |
|
Control |
13.36 ± 2.80 |
89.28 ± 1.71 |
2.62 ± 0.82 |
|
Low Dose |
14.32 ± 3.37 |
89.96 ± 0.96 |
2.48 ± 0.61 |
|
Medium Dose |
14.96 ± 6.10 |
88.14 ± 3.37 |
3.16 ± 1.30 |
|
High Dose |
15.34 ± 3.44 |
83.40 ± 8.44 |
3.54 ± 2.40 |
White Blood Cell Count (WBC) Mean ± SD Graph
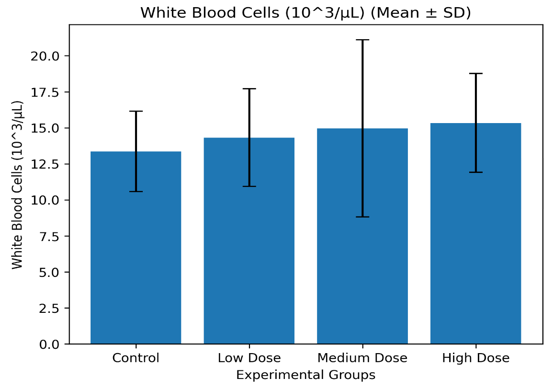
Figure 3.2.2: Mean white blood cell count (Mean ± SD) of Wistar albino rats administered Costus lucanusianus stem juice for 28 days.
Leukocyte parameters showed slight increases in treated groups compared to the control group, although these changes were not statistically significant (p > 0.05).
The mild elevation in white blood cell counts, particularly in the high-dose group, may suggest a possible immunomodulatory effect of the plant extract. This could be linked to bioactive compounds such as alkaloids and saponins, which are known to enhance immune responses [17].
However, since all values remained within normal physiological limits, the extract did not induce pathological leukocytosis or immune suppression.
Similar findings have been reported for plant-derived compounds with mild immunomodulatory properties at moderate doses [8]. Recent experimental studies also demonstrate that short-term exposure to medicinal plant extracts does not significantly alter hematological indices in rats, supporting their relative safety under controlled conditions [19].
3.2.3 Platelet Parameters
Table 3.4: Platelet Parameters (Mean ± SD)
|
Group |
Platelets (×10³/µL) |
MPV (fL) |
PDW (fL) |
|
Control |
500.0 ± 15.3 |
7.48 ± 0.27 |
6.26 ± 0.55 |
|
Low Dose |
577.8 ± 30.4 |
7.54 ± 0.27 |
6.70 ± 0.37 |
|
Medium Dose |
460.6 ± 150.4 |
7.24 ± 0.33 |
6.50 ± 0.30 |
|
High Dose |
529.6 ± 84.4 |
7.82 ± 0.89 |
6.26 ± 0.19 |
Platelet Count (PLT) Mean ± SD Graph
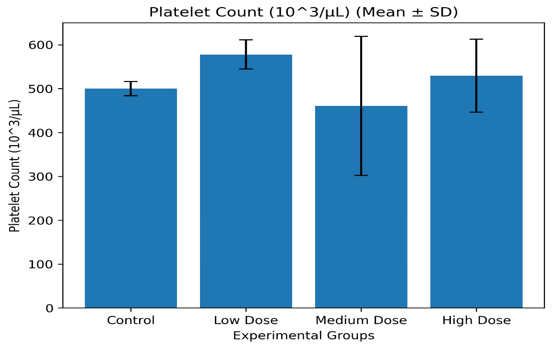
Figure 3.2.3: Mean platelet count (Mean ± SD) of Wistar albino rats administered Costus lucanusianus stem juice for 28 days.
Platelet counts and associated indices exhibited minor variations among the groups, did not differ significantly (p > 0.05).
The absence of consistent dose-dependent changes indicates that Costus lucanusianus stem sap did not adversely affect thrombopoiesis or platelet morphology.
Phytochemicals such as flavonoids are known to influence platelet aggregation and vascular function; however, the present findings suggest that these effects were not pronounced under the conditions of this study [7,16].
3.3 Overall Hematological Interpretation
Table 3.5: Summary of Key Hematological Parameters
|
Group |
Hb |
PCV |
RBC |
WBC |
Platelets |
|
Control |
11.62 |
37.32 |
6.17 |
13.36 |
500.0 |
|
Low Dose |
11.84 |
37.38 |
6.15 |
14.32 |
577.8 |
|
Medium Dose |
10.34 |
32.92 |
5.40 |
14.96 |
460.6 |
|
High Dose |
12.00 |
38.90 |
6.38 |
15.34 |
529.6 |
Statistical analysis (one-way ANOVA) showed no significant differences (p > 0.05) across all parameters.
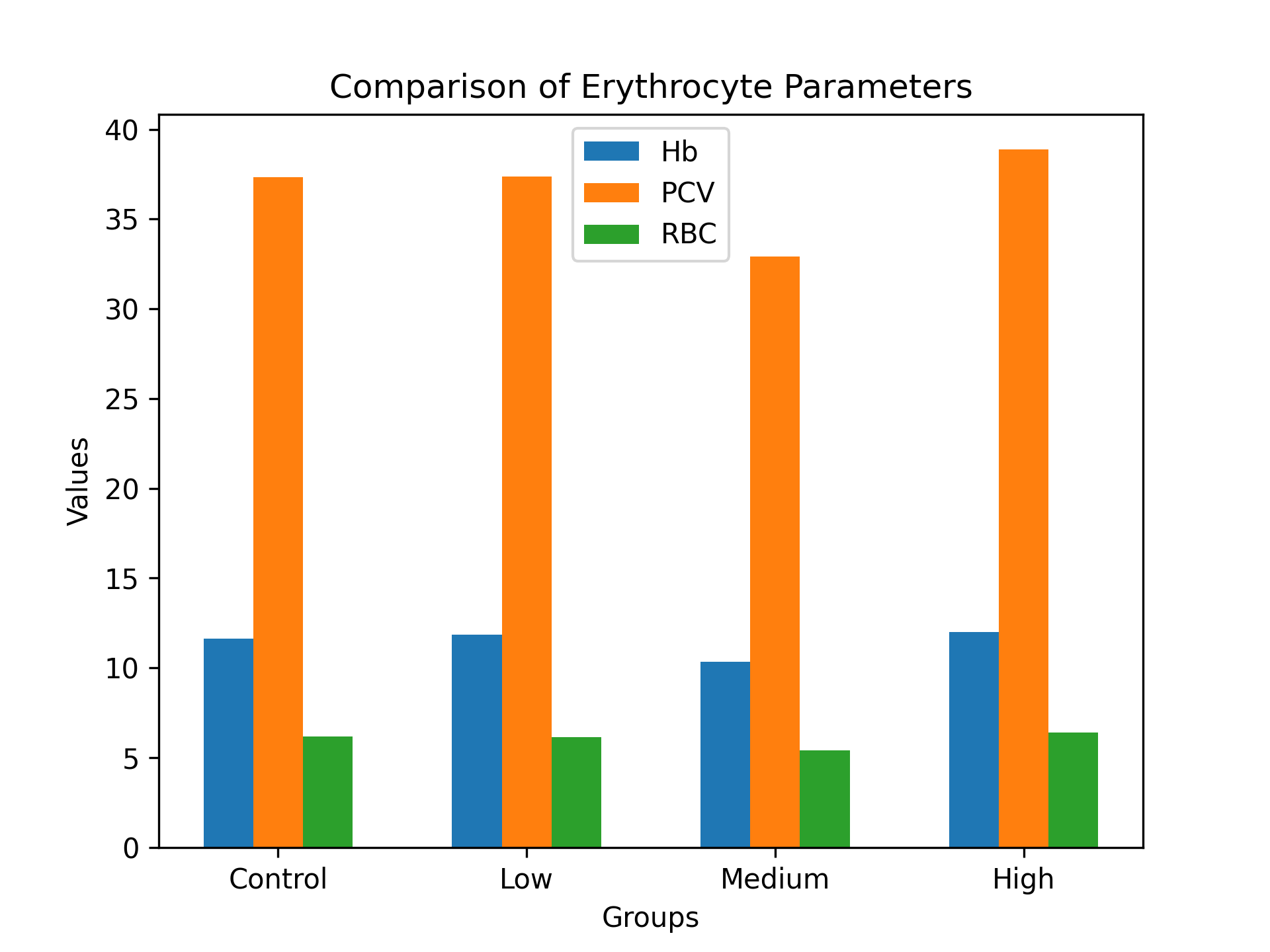
Figure 3.3: Hematological Comparison Summary of Key Hematological Parameters
3.4 Toxicological Implications
The absence of significant alterations in hematological parameters observed in this study suggests that Costus lucanusianus stem sap does not exert deleterious effects on the hematopoietic system under sub-chronic exposure conditions. Hematological indices are widely regarded as sensitive biomarkers of toxicity, particularly for detecting disruptions in erythropoiesis, immune function, and overall physiological homeostasis [9]. The stability of these parameters therefore indicates that the extract did not induce anemia, leukocytosis, leukopenia, or thrombocytopenia, which are commonly associated with toxic insults.
Furthermore, the acute toxicity assessment revealed no mortality or observable signs of toxicity at doses up to 5000 mg/kg body weight, indicating an oral LD₅₀ greater than 5000 mg/kg. According to standard toxicological classification criteria, substances with LD₅₀ values above this threshold are generally considered practically non-toxic [14]. This finding reinforces the safety profile of Costus lucanusianus stem sap and supports its traditional use in ethnomedicine.
The lack of hematological toxicity may be attributed to the presence of bioactive phytochemicals with antioxidant properties, which can mitigate oxidative stress and protect cellular components, including blood cells and bone marrow tissues, from damage [7,16]. Since oxidative stress is a major mechanism underlying toxicity in many plant-derived compounds, the apparent absence of such effects suggests a favorable balance between bioactivity and safety at the administered doses.
In addition, the use of a 28-day repeated-dose model, consistent with established sub-chronic toxicity testing guidelines, provides a reliable assessment of potential cumulative effects [11,18]. The findings from this study therefore indicate that Costus lucanusianus stem sap does not produce cumulative hematotoxic effects within the duration of exposure.
However, it is important to acknowledge that hematological parameters alone may not fully capture all aspects of systemic toxicity. Comprehensive evaluation involving biochemical, oxidative stress, and histopathological assessments would provide a more holistic understanding of the safety profile of the plant. Long-term studies are also recommended to determine potential chronic effects and to establish a definitive therapeutic safety margin.
4. CONCLUSION
The present study demonstrates that sub-chronic oral administration of Costus lucanusianus stem sap does not induce hematological toxicity in Wistar rats at doses up to 600 mg/kg. The absence of significant alterations in erythrocyte, leukocyte, and platelet parameters indicates that the extract does not adversely affect hematopoietic function under the conditions of this study.
The high safety margin observed in the acute toxicity study further supports its low toxicity profile. These findings provide experimental support for the traditional use of the plant. However, extrapolation to human safety requires further long-term and clinical investigations.
Authors’ contributions
This work was collaboration between the three authors. Both author HOA and VI designed the study. Author MES managed the literature searches, performed the statistical analysis and wrote the first draft of the manuscript. While author HOA and VI proof-read the manuscript to make corrections. All the authors read and approved the final manuscript.
REFERENCES
1. Sofowora A. Medicinal Plants and Traditional Medicine in Africa. 3rd ed. Ibadan: Spectrum Books Ltd; 2008.
2. World Health Organization. WHO Traditional Medicine Strategy 2014–2023. Geneva: WHO Press; 2013.
3. Adebayo SA, et al. Antioxidant and pharmacological properties of medicinal plants. J Ethnopharmacol. 2018;214:1–13.
4. Ojo OA, et al. Phytochemical and biological evaluation of medicinal plants. J Complement Integr Med. 2019;16(3):1–10.
5. Ekpe PF, et al. Ethnomedicinal uses of Costus species. Afr J Tradit Complement Altern Med. 2016;13(2):112–120.
6. Nwafor PN, et al. Pharmacological evaluation of herbal extracts. J Herbal Med. 2020;22:100345.
7. Wink M. Modes of action of herbal medicines. Medicines. 2015;2(3):251–286.
8. Wang L, et al. Toxicological evaluation of plant extracts. J Toxicol. 2017;2017:1–12.
9. Feldman BF, Zinkl JG, Jain NC. Schalm’s Veterinary Hematology. 6th ed. Ames: Wiley-Blackwell; 2015.
10. Ochei J, Kolhatkar A. Medical Laboratory Science: Theory and Practice. New Delhi: Tata McGraw-Hill; 2008.
11. OECD. Test No. 407: Repeated Dose 28-Day Oral Toxicity Study in Rodents. Paris: OECD Publishing; 2008.
12. National Research Council. Guide for the Care and Use of Laboratory Animals. 8th ed. Washington DC: National Academies Press; 2011.
13. Dacie JV, Lewis SM. Practical Haematology. 12th ed. London: Churchill Livingstone; 2016.
14. Lorke D. A new approach to practical acute toxicity testing. Toxicol Lett. 1983;14(3–4):307–310.
15. Daniel WW, Cross CL. Biostatistics: A Foundation for Analysis in the Health Sciences. 10th ed. New York: Wiley; 2018.
16. Pietta PG. Flavonoids as antioxidants. J Nat Prod. 2000;63(7):1035–1042.
17. Tona L, et al. Biological evaluation of medicinal plant extracts. Phytomedicine. 2021;85:153541.
18. Hayes AW. Principles and Methods of Toxicology. 6th ed. Boca Raton: CRC Press; 2014.
19. Nagumalli SK, Salley JT, Carstens JD. Assessment of clinical chemistry and hematological parameters in female Sprague-Dawley rats following oral exposure to Echinacea species. J Environ Sci Health C. 2024;42(2):154–171.
|
Cite this Article: Mandah, ES; Asuzu-Samuel, HO, Ibubeleye, V (2026). Effect of Costus lucanusianus (Ukpete) Stem Sap on Hematological Parameters in Wistar Rats. Greener Journal of Biomedical and Health Sciences, 9(1): 65-75, https://doi.org/10.15580/gjbhs.2026.1.042426058. |