By Ozele, KC; Ozele,
N (2022).
|
Greener
Journal of Medical Sciences
Vol. 12(1),
pp. 06-24, 2022
ISSN:
2276-7797
Copyright
©2022, the copyright of this article is retained by the author(s)
https://gjournals.org/GJMS
|

|
Anaemia in Pregnancy and
Malaria Parasitaemia in Women at Delivery after 2
Doses of Sulfadoxine-Pyrimethamine Combination in
Jos.
Ozele, Kingsley Chukwuka
(MBBS, FMCOG); Ozele Nonyelim
(BSC, FMLSCN)
Consultant Special Grade 1 (Obstetrics
and Gynaecology) and Head Medicine and Health
Services Department, National Institute for Policy and Strategic Studies Kuru Jos Plateau State, (NVRI) Vom.1
Chief Medical Laboratory Scientist
Biochemistry Division National Vertinary Research
Institute (NVRI) Vom.2
|
ARTICLE INFO
|
ABSTRACT
|
|
Article
No.: 020122010
Type: Research
Full
Text: PDF, HTML, EPUB, PHP
|
Background: Malaria
infection in pregnancy especially by Plasmodium falciparium
leads to parasite sequestration in the maternal placental vascular space with
consequent maternal anaemia and delivery of infant
with low birth weight.
Objective: The objective of the
study is to determine if significant differences exist in the occurrence of
anaemia, malaria parasitaemia, placenta parasitisation and low birth weight in women at delivery after administering 2 doses of sulfadoxine-pyrimethamine combination at JUTH when
compared to those women that had none or one dose of sulfadoxine-pyrimethamine.
Design: Hospital based
descriptive cross sectional study.
Methodology: Consenting 171 parturients in control Group who booked for ANC at JUTH
were recruited during the last four weeks of pregnancy at the antenatal
clinic and a pre –structured questionnaire administered. Consenting 167 parturients in the study Group were recruited as they
present in labour ward. Blood samples were collected as the women present in
labour at the labour suite. Placental blood was collected after delivery of
the placenta
Result: Statistically significant higher prevalence of
maternal malaria parasitaemia in the
none or once SP study group compared to twice SP control group. x2=
32.937, L.R= 33.660, P Value=0.000
The prevalence of anaemia
before delivery was higher in the none or once SP
study group compared to twice SP control group. But this was not
statistically significant. X2=0.823, L.R= 0.826, P Value=0.364(Fisher
exact test)
Statistically significant higher prevalence of
placental parasitisation in the
none or once SP study group compared to twice SP control group. X2=31.335,
L.R= 34.725, P Value=0.000
Statistically significant higher prevalence of
low birth weight (LBW) in the none or once SP study
group compared to twice SP control group. X2=5.197, L.R= 7.120, P
Value=0.029 (Fisher exact test)
Conclusion: 2 doses of SP
is more effective than none or once SP in preventing malaria parasitaemia, placental parasitisation
and reducing the prevalence of low birth weight in pregnancy.
|
|
Accepted: 01/02/2022
Published: 17/02/2022
|
|
*Corresponding
Author
Dr
Ozele KC MBBS FMCOG
E-mail: kingsleyozele9@ gmail.com
|
|
Keywords: Anaemia, pregnany, malaria, parasataemia, sulphadozine-pyrimetamine.
|
|
|
|
INTRODUCTION
1.1
Malaria Burden and Prevention in Pregnancy.
The female Anopheles species of the
mosquito is the vector that is responsible for the transmission of the
plasmodium parasite from human to human through its bite. There are four main
types of parasites that cause malaria but Plasmodium falciparium
is the most common and causes the most severe infection1.
According to
statistics by the World Health Organization (WHO), malaria is the largest
parasitic disease killer globally, killing 2 persons every minute1.
Globally, malaria causes 1-3 million deaths per year and 90% of such deaths
occur in sub-Saharan Africa. Children and women are the most affected.1 In the past, several chemoprophylaxis
like chloroquine, proquanil,
pyrimethamine and mefloquine
had been used in pregnancy but problems of rising levels of plasmodium falciparium resistance to most of these drugs has become an
issue.
The WHO, in a policy
for prevention and control of malaria recommends that the prevention of malaria during pregnancy
in areas of stable transmission should emphasize a preventive package of
Intermittent Preventive Therapy (IPT), insecticide treated bed nets (ITNs)
and ensure effective case management of malaria illness and anaemia1.
Intermittent Preventive Therapy is a public health intervention aimed at
preventing malaria episodes in Infants (IPTI), Children (IPTC),
School Children (IPTSC) and pregnant women (IPTP). The
intervention builds on two tested malaria control strategies that is to clear existing parasites and prevents new infections.
Intermittent preventive therapy in pregnant women involves the administration
of a single curative dose of an efficacious antimalarial drug at predefined
intervals (at least twice during pregnancy) regardless of whether or not the
woman is manifesting with symptoms of malaria, with the intent to protect a
pregnant woman against malaria. The drug is administered under supervision
during antenatal care (ANC) visit. Sulfadoxine –pyrimethamine is the combination currently recommended by
the WHO because of its safety and efficacy. 2, 3 It involves the use
of sulfadoxine -pyrimethamine
antimalarials at treatment doses given at predefined
intervals to clear a presumed burden of parasites.
IPT of malaria during
pregnancy is based on the assumption that every pregnant woman living in areas
of high malaria transmission has malaria parasites in her blood or placenta,
whether or not she has symptoms of malaria and could affect her baby.4
Malaria
infection contributes to as much as 15% of maternal anaemia,
14% of low birth weight infants, 30% of preventable low birth weight, 70% of
intrauterine growth restriction, 30% of premature delivery and 8% of infant
mortality.5, 6 The administration of SP twice in pregnancy has been
proven to reduce maternal anaemia by 39%, placental
malaria by 56% and low birth weight by 43%7
Two doses of SP are
required to achieve optimal benefit in most women. A third dose causes no
additional risk and is advocated for patients with lower natural acquired
immunity to malaria like those with HIV and sickle cell disease in pregnancy.7,8,
9
Nigeria
has adopted the use of IPT with SP since 20056. Malaria infection during pregnancy
contributes significantly to anaemia in pregnancy and
low birth weight. Antenatal anaemia has
shown positive correlation with low birth weight and high infant mortality rate8,9. The use of effective antimalarial drugs during
pregnancy has been found to lower the frequency of low birth weight and infant
mortality rate.9, 10 The use of IPT is a
promising approach in malaria control because it has shown potential to provide
some of the benefits of sustained prophylaxis for women.11
1.2 Statement of the Problem
Malaria infestation in pregnancy especially
by Plasmodium falciparium leads to parasite
sequestration in the maternal placental vascular space, with consequent
maternal anaemia and infant low birth weight (LBW)
due to both prematurity and intrauterine growth restriction (IUGR). 5, 6,
12 LBW is known to be the most important risk factor for infant
mortality.13 The WHO has
recommended the use of intermittent preventive therapy with at least 2 doses of
SP and other additional prevention measures during pregnancy. But some pregnant
women register for antenatal care at gestational ages when it is impossible for
them to have 2 doses of SP before 36 weeks of gestation. It also pertinent to
note that while some have their antenatal care where SP is not given routinely,
some others react to sulphur-based drugs and hence
may not have had the routine 2 doses of SP during pregnancy. The effect of this
observation in relation to the occurrence of anaemia
in pregnancy as well as malaria parasitaemia in women
at delivery in JUTH create an established medical question, for which this
study provides an answer.
1.3
Justification of the Study
In areas of endemic transmission, malaria in
pregnancy is associated with severe maternal anaemia
and low birth weight babies. The WHO has recommended the use of intermittent
preventive therapy with at least 2 doses of SP and other additional preventive
measures during pregnancy.3 Studies in Kenya, Malawi and Mozambique
have shown that IPT with at 2 curative doses of SP is highly effective in
reducing maternal anaemia and placental malaria
infection at delivery and also the number of low birth weight babies.14
Some pregnant women do not get the recommended 2 doses of SP
because of late booking for ANC. Some also book at centres that have not yet
adopted this WHO recommendation and as a result are not given 2 doses of SP. Others
react to sulphur-based drugs and hence may not have
had the routine 2 doses of SP during pregnancy. But some of these women come in and deliver
in our facility. This study is aimed at assessing if the risk of anaemia and
malaria parasitaemia on this group of mothers and
risk of low birth weights and placental malaria parasitaemia
for their babies are significantly higher than in those parturients
who had 2 doses of SP.
Prevention of malaria during pregnancy is one of the
major interventions to reduce maternal and infant morbidity and mortality with
the aim of contributing to achieving the fourth (two-thirds reduction in child
mortality rate), fifth (three-fourth reduction in maternal mortality rate) and
sixth (Combat HIV/AIDS, malaria and other diseases) millennium development
goals (MDG’s).3
The findings
from this study will be useful in assessing the effectiveness of the
intermittent preventive therapy and aid further actions. The study will help in health planning.
It is for this reason that this dissertation: Anaemia in pregnancy and malaria parasitaemia
in women at delivery after 2 doses of sulfadoxine-pyrimethamine
combination at JUTH is being conducted.
1.4 AIM
GENERAL: The
aim of the study is to determine if significant differences exist in the
occurrence of anaemia, malaria parasitaemia, birth
weights and placental parasitaemia in women at
delivery after 2 doses of sulfadoxine-pyrimethamine combination at JUTH when compared
to those women that had none or one dose of sulfadoxine-pyrimethamine
combination.
Objectives:
This study serves the
objective of identifying the prevalence of malaria parasitaemia
and specie types involved in the study group
Also, it identifies
the prevalence of anaemia before delivery at JUTH in the study group
In addition to the
forgoing, it further identifies the prevalence of low birth weight and that of
placental parasitisation in the study group.
Finally, it measures
the above four indices in control group and find out whether there are
statistically significant difference in the two groups.
1.5 Working Hypothesis
The prevalence of malaria parasitaemia,
placental parasitisation, anaemia and LBW are higher
in parturients who did not receive the recommended
doses of SP compared to those that received 2 doses of SP.
2. LITERATURE REVIEW
2.1 Prevalence
Each year, there are approximately 350–500 million cases of malaria,1
killing between one and three million people, the majority of whom are young
children in sub-Saharan Africa.2 Ninety percent
of malaria-related deaths occur in sub-Saharan Africa2. Malaria in
pregnancy is also a major public health problem in endemic tropical and
subtropical countries and a major cause of foetal and maternal morbidity.15
Anaemia in pregnancy is an important public health problem worldwide. It is the
most prominent haematological manifestation of malaria infection. This results
from destruction of red blood cells (both parasitized and unparasitized),
reduced feeding during malaria episodes, suppression of haemopoiesis,
intense sequestration of infected erythrocytes in the placenta and other
contributory factors.8 It is worse with Plasmodium falciparium which invades erythrocytes of all ages.16
Other causes of anaemia are poor nutrition, deficiencies of iron and other
micro nutrients, hookworm infestation, and schistosomiasis
disease including HIV infection and haemoglobinopathies
are additional factors.17 WHO estimates that more than half of
pregnant women in the World have a haemoglobin level indicative of anaemia
(< 11.0gldl), the prevalence may however be as high as 56 or 61% in
developing countries.18
A prevalence of 59.9% of malaria parasitaemia
and anaemia of 62.4% was found in a cross sectional study involving 272
pregnant women in a community study in Ebonyi State.19
However these patients were not followed up until delivery so as to assess the
impact of asymptomatic maternal malaria parasitaemia
on foeto-maternal outcome. Nonetheless, this
prevalence rate is comparable to others in Libreville Gabon (57%),20 and in Enugu Nigeria (58.4%).21
In a study at Obafemi Awolowo University Teaching Hospital Ile Ife, malaria parasitaemia of 21.1% was noted. Thirty-six (36%) of the
women had anaemia.22 Those with positive smear were treated with
600mg of chloroquine and after re-testing 2 weeks
later, were found to be malaria parasite negative.22 This study
followed up the patients after an intervention but the sample size was small
and now widespread resistance to chloroquine is
reported.
Also in a comparative experimental study in south
western Nigeria involving 294 participants, who were randomised into sulfadoxine-pyrimethamine SP and pyrimethamine
group, the result showed that at 34 weeks of gestation 35.3% and 6.6% of
malaria parasitaemia were found in the pyrimethamine and SP group respectively, which was
statistically significant.23 There were also more participants with
haemoglobin concentration less than 8g/dl at 34 weeks in the pyrimethamine group compared to the SP group. Participants
were given pyrimethamine and sulfadoxine-
pyrimethamine in the 2nd and early 3rd
trimesters23. Findings from this study in the southwest have shown
that 2 doses of SP for the prevention of malaria in pregnancy is associated
with a significant lower incidence of malaria parasitaemia,
maternal anaemia in pregnancy compared to weekly 25mg of pyrimethamine.23
But birth weights and placental malaria parasitaemia
were not assessed and compared at delivery.
In a multicentre study conducted for peripartum malaria in Nigeria, 21.6% of prevalence of
malaria parasitaemia (maternal and /or placental) was
found.24 Reduction in maternal haematocrit and higher proportion of
LBW babies were also found in those with malaria parasitaemia.
A cross sectional study in Maiduguri North Eastern
Nigeria using 437 women at delivery found a prevalence of 33.9% placental
malaria, while cord parasitaemia and maternal parasitaemia were 16.2% and 30.7% respectively.25
Jos University Teaching Hospital (JUTH), is located in Jos, the Plateau State capital. Jos Plateau
lies between latitude 70 and 110 North and Longitude 700
and 2500 east. This region is on a height of 1,200m above sea level.26
IPT has been adopted in JUTH. There is however no local study from this area.
2.2 Pathophysiology
A mosquito
infests a person by taking a blood meal. First, sporozoites
enter the bloodstream, and migrate to the liver. They infect liver cells
(hepatocytes), where they multiply into merozoites,
rupture the liver cells, and escape back into the bloodstream. Then, the merozoites infect red blood cells, where they develop into
ring forms, then trophozoites (a feeding stage), then
schizonts (a reproduction stage), and then back into merozoites. Sexual forms called gametocytes are also
produced which, if taken up by a mosquito, will infect the insect and continue
the life cycle 27, 31.
Malaria in humans develops via two phases: an exoerythrocytic
and an erythrocytic phase31. The exoerythrocytic phase involves infection of the hepatic
system, whereas the erythrocytic phase involves
infection of the erythrocytes, or red blood cells. When an infected mosquito
pierces a person's skin to take a blood meal, sporozoites
in the mosquito's saliva enter the bloodstream and migrate to the liver. Within
30 minutes of being introduced into the human host, the sporozoites
infect hepatocytes, multiplying asexually and asymptomatically for a period of
6–15 days. Once in the liver, these organisms differentiate to yield thousands
of merozoites, which, following rupture of their host
cells, escape into the blood and infect red blood cells, thus beginning the erythrocytic stage of the life cycle.27 The
parasite escapes from the liver undetected by wrapping itself in the cell
membrane of the infected host liver cell.28
Within the red blood cells, the parasites multiply further, again
asexually, periodically breaking out of their hosts to invade fresh red blood
cells. Several such amplification cycles occur. Thus, classical descriptions of
waves of fever arise from simultaneous waves of merozoites
escaping and infecting red blood cells.
Some P. vivax and P. ovale sporozoites do not
immediately develop into exoerythrocytic-phase merozoites, but instead produce hypnozoites
that remain dormant for periods ranging from several months (6–12 months is
typical) to as long as three years. After a period of dormancy, they reactivate
and produce merozoites. Hypnozoites
are responsible for long incubation and late relapses in these two species of
malaria.29
The parasite is relatively protected from attack by the body's immune
system because for most of its human life cycle it resides within the liver and
blood cells and is relatively invisible to immune surveillance. However,
circulating infected blood cells are destroyed in the spleen. To avoid this
fate, the P. falciparum parasite displays adhesive proteins on the
surface of the infected blood cells, causing the blood cells to stick to the
walls of small blood vessels, thereby sequestering the parasite from passage
through the general circulation and the spleen.30 This
stickiness is the main factor giving
rise to hemorrhagic complications of malaria. High
endothelial venules can be blocked by the attachment
of masses of these infected red blood cells. The blockage of these vessels
causes symptoms such as in placental and cerebral malaria. In cerebral malaria
the sequestrated red blood cells can breach the blood brain barrier possibly
leading to coma.31
Although the red blood cell surface adhesive proteins (called PfEMP1,
for Plasmodium falciparium erythrocyte
membrane protein 1) are exposed to the immune system, they do not serve as good
immune targets because of their extreme diversity; there are at least 60
variations of the protein within a single parasite and effectively limitless
versions within parasite populations.31,32 The parasite switches
between a broad repertoire of PfEMP1 surface proteins, thus staying one step
ahead of the pursuing immune system.
Some merozoites turn into male and female
gametocytes. If a mosquito pierces the skin of an infected person, it
potentially picks up gametocytes within the blood. Fertilization and sexual
recombination of the parasite occurs in the mosquito's gut, thereby defining
the mosquito as the definitive host of the disease. New sporozoites
develop and travel to the mosquito's salivary gland, completing the cycle.
Pregnant women are especially attractive to the mosquitoes,32
and malaria in pregnant women is an important cause of stillbirths, infant
mortality and low birth weight,33 particularly in P. falciparum
infection, but also in other species infection, such as P. vivax.33
2.3 Placental Malaria
Currently, susceptibility to Plasmodium parasitaemia
has been linked to the level of antibodies to placental sequestrated
parasites.34 Indeed these parasites preferentially adhere to
chondroitin sulphate-A receptors (CSA) expressed by the syncytiotrophoblasts in the placenta.35
Women in their first and second pregnancies are more susceptible as anti adhesion antibodies against CSA binding parasites
develop after successive pregnancies.36 The presence of parasites
in peripheral blood without symptoms is common in hyper-endemic
areas, and is associated with chronic anaemia and placental
sequestration .37
2.4 Malaria and Anaemia in
Pregnant Women
Malaria infection caused by Plasmodium falciparum is
a major cause of fever and anaemia in pregnant women resident in hyper
endemic areas of Africa. Basically, this is as a result of reduced immunity
to malaria in pregnancy38, making the pregnant women prone to severe
malaria attack and subsequently anaemia. It has been shown that severe anaemia
was more than twice as common in women with peripheral parasitaemia as in those without parasitaemia.39
Incidentally malaria infection is more rampant among the primigravidae
and secundigravidae than the multigravidae.21
The preferential susceptibility of these sets of pregnant women may be related
to some evidence that immunosuppression associated with pregnancy, occurs
more in the first than subsequent pregnancies.40, 41, 42. Previously, the depression of cell mediated
immune response to Plasmodium falciparum antigens has been implicated in
this phenomenon.41 Age has also been implicated as epidemiological
studies have shown that malaria in pregnancy is more prevalent in younger than
older age groups,10,37,43,44. Malaria infection during pregnancy contributes
significantly to anaemia in pregnancy and low birth weight babies.45
Antenatal anaemia has shown positive correlation with low birth weight
(LBW) and high infant mortality rate (IMR). 11, 28
2.5 Consequences of Malaria in
Pregnancy
In Africa, malaria is highly endemic and is the
leading cause of morbidity and mortality. It contributes 4—19% to low birth
weight, 3—15% to maternal anaemia, and 3—8% to infant deaths, while maternal
anaemia contributes 7—18% to low birth weight.27-29
2.6 Diagnosis
2.6.1 Microscopy
Staining
thick and thin blood films on a glass slide to visualize the malaria parasite
microscopically is the method of diagnosis of malaria. The patient’s finger is
cleaned with alcohol, allowed to dry and then the side of the fingertip is
pricked with a sharp sterile lancet or needle and two drops of blood are placed
on a glass slide. To prepare a thick blood film, a blood spot is stirred in a
circular motion with the corner of the slide, taking care not make the
preparation too thick, and allowed to dry without fixative. As they are unfixed
the red cells lyse when a water based stain is applied. A thin blood film is
prepared by immediately placing the smooth edge of a spreader slide in the drop
of blood, adjusting the angle between slide and spreader to 45°, and then
smearing the blood with a swift and steady sweep along the surface. The film is
then allowed to air dry and is fixed with methanol. Blood smears are
stained using Giemsa stain and parasites are counted
against 100 leukocytes and expressed as number of parasites/ml of blood
assuming a standard leukocyte count of 8000/ml of blood. A blood smear is
negative when minimum of 100 high power fields is examined with no parasites
seen. It is positive when parasites are seen
Parasite density is calculated as mean geometric mean parasite density
(GMPD/ml). Larger
volume of blood is examined in the thick film, so it is more sensitive than the
thin film (down to around 40 parasites per μL or
1 parasite per 200 white blood cells) although examination of the thick film
requires more expertise to read46.
The
simple, direct microscopic observation of blood specimens to observe the
malaria parasite is still the gold standard for malaria diagnosis47.
2.6.2
Molecular methods
The
polymerase chain reaction (PCR) allows the specific amplification of a selected
region of the malarial genome.48 This technique is highly specific
and sensitive (1-5 parasite/mL of blood) and permits genotyping.49, 50
Furthermore, PCR using single nucleotide polymorphism (SNP) analysis allows the
detection of drug resistant parasites and mixed infections. 51, 52, 53
However, PCR is expensive and requires a sophisticated laboratory manned with
well-trained staff. This is not available in our facility
2.6.3
Rapid methods
Detection
in patient samples of malaria parasite antigens such as histidine rich protein II (HRP-II) or plasmodium lactate
dehydrogenase (pLDH) can be performed by
rapid, point-of-care tests based on immunochromatographic
methods. There are many commercially-available rapid tests including Para Sight F54,55 and Paracheck, Binax NOW P.f./ P.v. and OptiMAL (Flow Inc.,
USA).56,57,58
The advantages of these tests are
that they are quick to perform and have high sensitivity.57, 59 The
disadvantages of the rapid format are the relatively high cost, the inability
of some tests to distinguish malaria species, and manufacturing variation.58
Those based on HRP II detection may give positive results in the convalescent
phase of the illness due to the persistence of HRP II in the blood after
parasite clearance.60
2.6.4
Quantitative Buffy Coat method
Quantitative
buffy coat (QBC; Becton Dickinson, USA) is a method for identifying the malarial
parasite in the peripheral blood. It involves staining of the centrifuged and
compressed red cell layer with acridine orange and
its examination under an ultraviolet (UV) light source60. Blood is
collected (from a finger prick) in a haematocrit tube
containing acridine orange and anticoagulant. The haematocrit tube is centrifuged at 12,000 g for 5 min and
immediately examined using a microscope equipped with a UV light source.
The
parasite nucleic acid fluorescence bright green and the cytoplasm appears
yellow-orange60. This test has sensitivity similar to the
conventional thick blood film microscopic methods. It is reliable and
user-friendly and should be used together with thick blood film microscopic
screening. However, QBC requires specialized instrumentation, has a higher cost
than microscopic methods and is poor at species determination and parasite
quantification60.
2.6.5
Serological methods
Serological
tests for the diagnosis of malaria infection rely on the detection of
antibodies against asexual blood stages of the malaria parasite. The first
serological test used for the detection of malaria antibodies was the
immunofluorescence assay, often abbreviated to IFA.61 This method
uses specific antigen or crude antigen prepared on a slide, coated and kept at
–300C until use, and quantifies both IgG
and IgM antibodies in patient serum samples. Titres >1:20 are classified as positive, and those below
1:20 classified as of doubtful significance. High titres
(>1:200) represent strong evidence of a recent infection. Serological tests provide retrospective
confirmation of malaria infection or a history of infection, and are useful in
epidemiology surveys and the screening of blood collected for blood banks.
Nevertheless, the utility of serological methods for the diagnosis of acute
malaria infection is limited owing to the delay in antibodies development, lack
of species confirmation and the need for fluorescence (UV) microscope.62
2.6.6 Diagnosis Of
Placental Malaria Parasitaemia
There are two main methods of
diagnosing placental malaria parasitisation. These
are microscopy or histology.
2.6.6A Microscopy:
Placental blood is collected within an hour after child-birth by incising the
cleaned maternal surface (basal plate) of the placenta, and drawing 5ml of
blood welling from the incision using a sterile syringe and needle. The
placental blood is then processed as described above.
2.6.6B Histology:
Immediately following delivery, the placenta is obtained and a large biopsy
specimen of placental tissue (2 by 2 by 1 cm) is fixed in 10% neutral buffered
formalin for histopathological studies. Fixed
placental biopsies are transferred to the histopathology laboratory where they
were processed, embedded in paraffin wax and sectioned onto slides by standard
techniques. Sections will later be stained with haematoxylin-
eosin stain for detection of active and past infections. One thousand intervillous cells (IVS) are counted to determine the level
of parasitaemia in placenta tissue sections. Past
infection is defined as the presence of malaria pigment in fibrin or monocyte
/macrophage without malaria parasites 63. Active infection is when
malaria parasite is seen with or without pigments. It is negative when neither
is seen. Sections may also be observed under polarised
light to assess the presence of malaria
pigment64
Placental histology is considered
the gold standard of malaria
diagnosis in pregnancy for epidemiological or biological study purposes,
because it can show signs of active, active chronic or past infections.65,
66 However, it is expensive and it is not done routinely at the Jos
University Teaching Hospital.
2.7 Prevention
Methods used to prevent the spread of disease, or to protect individuals
in areas where malaria is endemic include prophylactic drugs, mosquito
eradication, and the prevention of mosquito bites10.
In the past decade, strategies have been developed to
more effectively control the adverse effects of malaria during pregnancy. The
African Summit on Roll Back Malaria (RBM) in April 2000 adopted the Abuja
Declaration in which regional leaders committed to ensuring that sixty percent of pregnant women in malaria-endemic communities
accessed effective prevention and treatment of malaria by 2005. The following
approaches were to be used:
1.
Supporting and promoting access to correct, affordable and appropriate treatment
within twenty-four hours of the onset of symptoms.
2.
Supporting and promoting access to a suitable combination of personal
and community protective measures such as Insecticide Treated Nets (ITNs).
3.
Supporting and promoting the use of malaria preventive measures such as
chemoprophylaxis or intermittent preventive treatment for pregnant women (IPTp).
The WHO recommended that the policy for the prevention
of malaria during pregnancy in areas of stable transmission should emphasize a
preventive package of Intermittent Preventive Treatment (IPT) and Insecticide
Treated bed Nets (ITN’s) and ensure effective case management of malaria
illness and anaemia. IPT is the use of anti-malarial drugs given in treatment
doses at predefined intervals to clear a presumed burden of parasites. IPT of
malaria during pregnancy (IPTp) is based on the
assumption that every pregnant woman living in areas of high malaria
transmission has malaria parasites in her blood or placenta, whether or not she
has symptoms of malaria.
The IPT package means that all pregnant women in areas
of stable malaria transmission should receive at least two doses of IPT after
quickening, that is, after 16 weeks gestation.
The most effective drug for IPT currently is Sulphadoxine Pyrimethamine (SP)
because of its safety for use during pregnancy, effectiveness in
reproductive-age women, and the feasibility for use in programs as it can be
delivered as a single- dose treatment under direct observation (DOT) by the
health worker.
3. SUBJECTS, MATERIALS AND METHOD
3.1 Study Area.
This is a
hospital-based study conducted at the Maternity Unit of the Department of
Obstetrics and Gynaecology, Jos University Teaching Hospital (JUTH).
JUTH is a tertiary health institution located at
its permanent site at Lamingo Jos, the Plateau State
capital. It is one of the three teaching hospitals in the North-central zone of
Nigeria,
although there are Federal Medical Centres (FMCs) in
the remaining states within the geo-political zone.
Plateau State lies
between latitude 70 and 110 North and Longitude 700
and 2500 east. The capital city is a pear shape upland known as Jos
Plateau. This upland stretches for approximately 104km from north to south, and
80km from east to west, covering an area of about 8,600sqkm26.
This region has a
height of 1,200m above sea level.26 Plateau State has over 30
different ethnic groups.26 The 1991 Nigerian census puts the
population of Plateau State at 2,959,588 with 1,031,662 being females.26
The Departmental
protocol for malaria chemoprophylaxis is the intermittent preventive treatment
using sulfadoxine-pyrimethamine combination. 2 doses
are given. The first is after quickening and the second at least 4weeks after the
second but before the last 4 weeks of pregnancy. A third dose is
usually given to some patients like those with HIV, and sickle cell disease in pregnancy
that is if the HIV positive pregnant woman is not on cotrimoxazole7.
3.2 Study Population
The study population are
women presenting in labour at the Jos University Teaching Hospital, North
Central Nigeria.
3.3 Study Design
The study is a prospective,
descriptive cross sectional hospital based study conducted over a six-month
period (June- December, 2011)
Study Group: Delivery
at 37-42 weeks in
1.
Women who received 1 dose of SP because of late
booking
2.
Women who did not receive any dose of SP because they
were unbooked
3.
Women who did not receive any dose of SP because they
booked very late in pregnancy
4.
Women who did not receive any dose of SP because they
booked for antenatal care elsewhere where they were not given SP.
Control Group: Shall
include women delivering at 37-42weeks who received 2 doses of SP in the index
pregnancy.
Consenting parturients in
Control Group who booked for ANC with JUTH were recruited during the last four
weeks of pregnancy at the antenatal clinic and a pre–structured questionnaire
administered. Consenting parturients in the Study group
were recruited as they present in labour ward. Blood sample was collected as
the women present in labour at the labour suite. Placental blood was collected
within 1 minute of delivery of the placenta
3.3.2 Exclusion Criteria
The following categories of parturients
were excluded from the study:
1. Sickle cell Disease patients
2. Multiple pregnancies
3.
Women with severe preeclampsia
4.
Women with Intrauterine foetal death
5.
Women with chronic anaemia from other causes like poor nutrition, if
readily ascertained from history and or physical examination
6.
Women who are none compliant on haematinics
7.
Preterm and post term deliveries
8.
Women who decline to participate in the study.
3.4 Ethical Consideration
The proposal for this study was presented to the
Research and Ethical Committee of Jos University Teaching Hospital for approval.
Informed consent was obtained from the subjects before enlistment into the
study.
3.5 Sample Size
The sample size estimation
was calculated using the formula for studying proportions with population
>10,000 i.e. N=Z2pq/d2 in Araoye67:
N=z2 pq/d2
N= desired sample
size
Z=standard normal
deviate 1.96 which correspond to 95% confidence interval.
P= prevalence
expressed as 100% i.e 12%22
q=complimentary
proportion 1-p
d=degree of accuracy
desired=0.05
N=(1.96)2
x 0.12 x 0.88
(0.05)2
161.4532
A total of 167 pregnant
women were recruited in the Study (none or once SP) group and 171 in the Control
(twice SP) group.
3.6 Data Collection
Collection of Blood Samples
3mls of blood samples were collected aseptically by
venepuncture using 5 ml sterile disposable hypodermic syringes and needles
before delivery in labour ward and dispensed into prelabelled
EDTA specimen bottles and transferred to the medical microbiology laboratory of
the hospital.
Maternal blood samples were also collected into
heparinised microhaematocrit tubes, sealed and
spurned at 12000 revolutions per minute for 5minutes using a haematocrit
centrifuge(Hawskey and Sons Lancing UK) at the
haematology laboratory of the JUTH. The packed cell volume was read off as percentages
using a Hawskey haematocrit tube reader.
Thick blood smears were prepared and examined at
medical microbiology laboratory. Blood smears were stained using Giemsa stain and parasites were counted against 100 leukocytes and expressed
as number of parasites/ml of blood assuming a standard leukocyte count of 8000/ml
of blood. A blood smear was negative when minimum of 100 high power fields was
examined with no parasites seen.
Parasite density was calculated as mean geometric mean
parasite density (GMPD/ml).
Placental blood was collected within an hour after child-birth,
by incising the cleaned maternal surface (basal plate) of the placenta and
drawing 5ml of blood welling from the incision using a sterile syringe and
needle. Thick blood film was prepared, stained with Giemsa
stain and also analyzed according to the procedure for microscopic diagnosis
for malaria parasitaemia.
Quality control was ensured by re-examination of a
randomly selected 10% sample of all slides by another Scientist to confirm the
accuracy of the results.
Sample collection was done by the researcher with
assistance from some Resident Doctors and House Officers in the Department. The
test was carried out by 2 experienced Laboratory Scientists. One is a
Haematologist and the other a Microbiologist. Samples were sent to the
laboratory as they were collected and the researcher supervised each and every
analysis.
3.7 Statistical Method for Data Analysis
The comparisons were
made using means or chi square test. Chi square was used to determine
significance of association between categorical data. Continuous independent variables
like birth weight were tested using analysis of variance (ANOVA). P value of < 0.05 was considered statistically
significant in all statistical comparisons .All analyses were conducted using
the SPSS version 15 software.
3.8 Limitation to the Study
Apart from malaria in pregnancy, there
are other risk factors for anaemia that may not be easy to control in this
study. These include difficulty in
assessing nutritional status of participants, difficulty in assessing precisely
the socio-economic status of participants and also difficulty in assessing
contributions by other factors like hookworm etc.
Also inability to test for placental
malaria parasitaemia using histological methods,
which is the gold standard, is another limitation. Histology was not used for
diagnosis in this case because of the huge cost implication
3.9 Benefits of the Study
To the Patient
1.
Opportunity to have their pack cell volume and malaria
parasite test done at no cost to the participant.
To Humanity
Efficacy or otherwise
of current policy on Intermittent Preventive Therapy would have been proven and
this would affect further policy decisions.
RESULTS
Table 1: Mean age, weight, height
and booking PCV.
|
Characteristics
|
None or 1 SP
|
Twice SP
|
P- value
|
|
Mean age (years)
|
28.41±5.5
|
29.83±5.8
|
0.022 *
|
|
Mean weight (Kg)
|
75.83±46.87
|
77.71±43.17
|
0.700
|
|
Mean height (cm)
|
160.31±6.00
|
159.69±7.40
|
0.403
|
|
Booking PCV (%)
|
33.84±2.82
|
34.26±2.76
|
0.204
|
*Statistically significant.
Table 2:
Social Class
|
Participant’s level of education
Primary
Secondary
Tertiary
None
Total
|
None or once SP
13
87
63
4
167
|
Twice SP
13
63
92
3
171
|
|
Participant’s occupation
Civil/public
servant
Business
woman
House
wives
Hair
dresser
Students/
NYSC
Missionary
Total
|
None or once SP
39
26
61
17
21
3
167
|
Twice SP
59
37
40
14
20
1
171
|
|
Husband’s level of education
Primary
Secondary
Tertiary
None
Total
|
None or once SP
8
83
76
0
167
|
Twice SP
10
43
116
2
171
|
|
Husband’s occupation
Artisan
Business
man
Civil
/ public servant
Student
/ NYSC
Farmer
Missionary
Unemployed
Total
|
None or once SP
17
50
86
9
0
2
2
166
|
Twice SP
19
54
90
3
4
1
0
171
|
|
Social classification(Olusanya et al 68)
Social
class 1
Social
class 11
Social
class 111
Social
class 1V
Social
class V
Total
|
None or once SP
2
77
66
21
1
167
|
Twice SP
6
75
69
19
2
171
|
No statistically significant association
between the study group and social classification. X2=3.790, P
value=0.876.
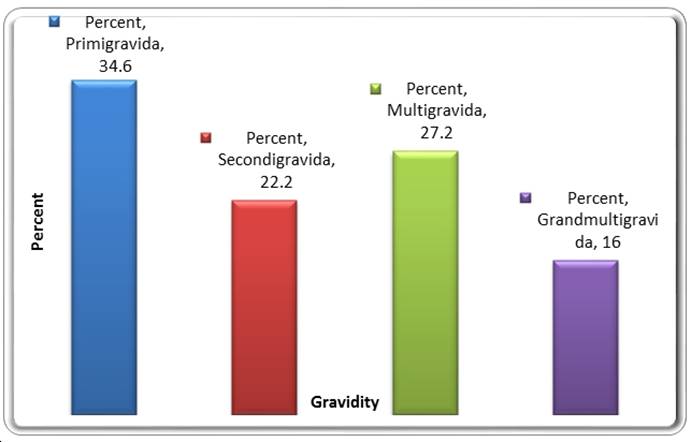
Figure 1. Distribution of General Subjects by Gravidity
Primigravidae=1st
pregnancy, secundigravidae=2nd pregnancy, multigravidae=3rd and 4th pregnancies and grandmultigravidae=5th and above
X2=9.407, L.R= 9.571, P value=0.024
Table 3:
Malaria treatment in index pregnancy and parasite density
|
Treatment
|
-
|
+
|
++
|
+++
|
Total
|
|
Yes
|
30
(34.9%)
|
52
(60.5%)
|
2
(2.3%)
|
2
(2.3%)
|
86
(100.0%)
|
|
No
|
108
(42.9%)
|
130
(51.6%)
|
14
(5.6%)
|
0
(0.0%)
|
252
(100.0%)
|
|
Total
|
138
(40.8%)
|
182
(53.8%)
|
16
(4.7%)
|
2
(0.6%)
|
338
(100.0%)
|
X2=9.211, P value=0.027
Table 4:
Use of insecticide treated nets and maternal peripheral parasitaemia.
|
Use of ITNS
|
Positive MP
|
Negative MP
|
Malaria pigments
|
Total
|
|
Yes
|
95
(56.5%)
|
49
(29.2%)
|
24
(14.3%)
|
168
(100.0%)
|
|
No
|
106
(63.9%)
|
38
(22.9%)
|
22
(13.3%)
|
166
(100.0%)
|
|
Total
|
201
(60.2%)
|
87
(26.0%)
|
46
(13.8%)
|
334
(100.0%)
|
No
association between use of ITNS and placental malaria parasitisation; X2 =5.555 P value= 0.135
Table 5: Maternal
peripheral malaria parasitaemia
|
Study Groups
|
Positive MP
|
Negative MP
|
Pigments
|
Total
|
|
None or once SP
|
126
(75.4%)
|
25
(15.0%)
|
16
(9.6%)
|
167
(100.0%)
|
|
Twice SP
|
75 (44.9%)
|
62
(37.1%)
|
30
(18.0%)
|
167
(100.0%)
|
|
Total
|
201
(60.2%)
|
87
(26.0%)
|
46
(13.8%)
|
334
(100.0%)
|
X2=32.937 P Value= 0.000.
Table 6:
Species types involved
|
Study Groups
|
P. falciparium
|
P. malariae
|
P. Ovale
|
P. vivax
|
Total
|
|
None or once SP
|
74
(59.2%)
|
47
(37.6%)
|
2(1.6%)
|
2(1.6%)
|
125(100.0%)
|
|
Twice SP
|
53
(71.6%)
|
19
(25.7%)
|
0(0.0%)
|
2(2.7%)
|
74(100.0%)
|
|
Total
|
127 (63.8%)
|
66(33.2%)
|
2(1.0%)
|
4(2.0%)
|
199(100.0%)
|
Table 7: prevalence of anaemia
(Using PCV<30% and PCV<33%)
|
Study Groups
|
PCV<33% (anaemic)
|
PCV
<33 (not anaemic)
|
PCV <30% (anaemic)
|
PCV <30% (not anaemic)
|
Total PCV <33%
|
Total PCV <30%
|
|
None or Once SP
|
14
(8.4%)
|
153
(91.6%)
|
1
(0.6%)
|
166
(99.4%)
|
167
(100.0%)
|
167
(100.0%)
|
|
Twice SP
|
10
(5.8%)
|
161
(94.2%)
|
4
(2.33%)
|
167
(97.67%)
|
171
(100.0%)
|
171
(100.0%)
|
|
Total
|
24 (7.1%)
|
314 (92.9%)
|
5
(1.5%)
|
333 (98.5%)
|
338 (100%)
|
338 (%)
|
P Value=0.403 (Fisher exact test)-PCV<33%
P
Value=0.4655 (Fisher
exact test)-PCV<30%
Table 8a:
birth weight of babies in the two groups.LBW=
<2500kg
|
Study Group
|
LBW
|
Normal weight
|
Total
|
Mean weight of babies(g)
|
|
None or once SP
|
5
(3.0%)
|
162
(97.0%)
|
167
(100.0%)
|
3120.6±44.76
|
|
Twice SP
|
0
(0.0%)
|
171
(100.0%)
|
171
(100.0%)
|
3255.4±463.17
|
|
Total
|
5
(1.5%)
|
333
(98.5%)
|
338
(100.0%)
|
|
Statistically significant difference in low birth weight
LBW in the two groups
X2= 5.197, P Value (using fisher
exact test) = 0.029,
Also in mean weight of babies, p value=
0.0066
Table
8b: Placenta parasitisation in the two groups
|
Study Group
|
-ve
|
P.G
|
+
|
++
|
Total
|
|
None or once SP
|
38
(22.9%)
|
21
(12.7%)
|
99
(59.6%)
|
8
(4.8%)
|
166
(100.0%)
|
|
Twice SP
|
69
(40.4%)
|
40
(23.4%)
|
62
(36.3%)
|
0
(0.0%)
|
171
(100.0%)
|
|
Total
|
107
(31.8%)
|
61
(18.1%)
|
161(47.8%)
|
8
(2.4%)
|
337
(100.0%)
|
X2 =31.335
P Value=0.000,
4. DISCUSSION
The study was
undertaken to find out if there are statistically significant differences in
the prevalence of malaria parasitaemia, anaemia, low birth weight and placental parasitisation
between pregnant women who took 2 doses of SP and those that had none or once
SP
Social classification
of the two groups were done using protocol for social classification by Olusanya et al (1985), social classes 1-V were derived
using the husband’s occupation and the wife’s education68. There was
no statistically significant association between the social class
in the two groups. Also social class did not significantly affect the anaemia, maternal malaria parasitaemia
and the placental parasitaemia. P values were >
0.05. But the protocol did not address the women that had no formal education
and also who qualifies as ‘middle level’ in the husband’s occupation was not
easy to determine
In the none or once
SP study arm, there were 167 women, out of whom 75.4% had positive MP, 9.6% had
pigments and 15% were negative. While in the twice SP control group, of the 167
women, 44.9% had positive MP, 18% had pigments and 37.1% were negative. This
was statistically significant. X2=32.937, P
value=0.000 Likelihood Ratio=33.660,
The prevalence of
malaria parasitaemia in the twice SP study arm is
comparable to 45% prevalence rate reported by Praise et al (1998) in Kenya in
which SP or chloroquine was used as intermittent
presumptive treatment5. Also in an observational study in Malawi,
Sullivan et al (1999) also found a prevalence of 37% malaria parasitaemia.
A cross sectional
study carried out at University of Maiduguri Teaching Hospital by Bako et al (2009) found a prevalence of 30.7 in the
subjects. 68% of the subjects had used 2 doses of SP25. The reported
prevalence in the above study is lower than observed in the two-study arms in
this particular study. These are geographically distinct population. Here
difference in geographical location and difference in the uptake of insecticide
treated nets could account for the difference. Jos is on the plateau at an
altitude of 1,200m above the sea level26.
The inverse relationship between malaria prevalence and altitude has been
reported elsewhere in Tanzania. In a study in Usambra
mountain in the North eastern Tanzania, a prevalence of malaria in children was
observed to decrease by 5% for every 100m increase in altitude from 82% in the
lowlands (300m) to 12% in the highlands (1700m)69.
It was supported by another study also in Northern Tanzania70. Lower
malaria prevalence in higher altitude is likely to be attributed to low ambient
temperatures that discourage vector transmission71. However, local
variations in seasonality of malaria transmission including vector species
composition, topography, host and parasite genetics and socioeconomic factors
influence the prevalence of malaria parasitaemia in
any given area71.
Nnatu et al (1987)
in a Lagos cross-sectional study of 230 women at delivery reported a prevalence
of 40%72. Here details of antimalarials
and malaria chemoprophylaxis were not given hence the data were difficult to
compare.
Ogbodo et
al (2009) reported a prevalence of
59.9% in a cross sectional study involving 272 women in a rural community in Ebonyi State, where a case was made for the need for
combined preventive approach19. Prevalence rates are comparable
The prevalence of anaemia in the two groups show that out of the 167 in the
none or once SP arm 14 (8.4%) had PCV less than 33% while in the twice SP arm,
out of the total of 171 women, 10 (5.8%)
had PCV less than 33%. It is true that the prevalence of anaemia
is higher in the none or once SP. However this did not
reach a statistically significant level. X2=0.823, L.R= 0.826, P
value=0.364.
The difference was
not also statistically significant when PCV less than 30% was used as cut-off
for anaemia. Using Fisher exact test, P value =
0.4655, Risk Ratio 0.506, C.I= 0.353-2.366.
The prevalence of anaemia in the study arm and control arm are lower than
most reported prevalence rates locally73 and regionally74.
But prevalence rate 2-30% has also been reported75. Difference in
nutritional status, hookworm and schistosomiasis
infestations could account for the differences. Moreover in this study, women
with risk factors known to be associated with anaemia
such as sickle cell disease, multiple pregnancies, women with severe
preeclampsia, women who are not compliant on haematinics
were excluded from the study.
The prevalence of low
birth weight (LBW) in the two groups when also compared showed that of the 167
women in the none or once SP study, 5 had LBW giving a prevalence of 3%. But there was no
recorded case of LBW of the 171 women in twice SP control arm. This was statistically significant. X2=
5.197, L.R = 7.127, using Fisher exact test, P value=0.029
When the mean birth
weights in the two groups were compared, the none or
once SP group was 3120.5988 while the twice SP group was 3255.4035. Again this
was statistically significant. T stastistic=2.7341, P
value=0.0066.
Prevalence of LBW of
9.5% after 2 doses of SP as against 13.3% in group that had no intervention was
reported by reported Challis et al (20004), in a randomized control trial in
southern Mozambique76. Difference could be accounted for by sample
size and difference in other determinant of final birth weights like race,
maternal weight, paternal height, and other medical conditions associated with LBW,
most were excluded from this study.
Also prevalence of
LBW of 8% was also reported in similar cross sectional study in Maiduguri25.
This particular study also found a statistically significant association
between placental parasitisation and LBW. In this
study, all the 5 babies with LBW had placenta malaria parasitisation.
The high perfusion of human placenta makes it easily accessible to malaria
parasites in the maternal circulation making placenta malaria a common finding25.
Placenta malaria parasitisation in the two groups also showed interesting
results. Of the 166 in the none or once SP group 38
(22.9%) was negative, 21 (12.7%) had pigments and 99 (59.6%) had 1+, with 8
(4.8%) having 2++. In the twice SP control arm, of the 171 women, 69 (40.4%) was
negative, 40 (23.4%) had pigments, 62 (36.3%) had 1+. There was none in this
group with 2++. This was statistically significant. X2=31.33, L.R=
34.725, P value=0.000. Also of the 61 placenta blood with malaria pigments
which suggests previous infection, 40 were in the twice SP group while 21 were
in none or once SP group showing that 2 doses of SP in pregnancy is more
effective in the placenta malaria parasite clearance. X2=6.684,
L.R=6.783, Using Fisher exact test, P value =0.011.other authors also agree
that IPT with SP is effective in reducing placenta malaria in our environment77.
The prevalence of
placenta parasitisation is however higher than that
reported by Bako et al (2009) where a prevalence of
33.9% was reported25. Also Ukaga et al(2007) in a multicentre cross
sectional study in Owerri Imo State South eastern
Nigeria found a placental parasitisation of 29.9%78.
The difference could be accounted for by study population, malaria
transmission, or differences in diagnostic methods. Placental malaria prevalence
of 57.69% was reported by Ibhanesebhor and Okolo (1992) in Benin which is comparable to the prevalence
in the none or once SP study arm79. Though
there was no intervention in the form of either IPT or ITNS in the
Benin study.
5. CONCLUSION
The results of the
study showed that there was statistically significant higher prevalence of
maternal malaria parasitaemia in the
none or once SP study group compared to twice SP control group
It also showed that the
prevalence of anaemia before delivery was higher in the none or once SP study group compared to twice SP control
group. But this was not statistically significant
There was also statistically
significant higher prevalence of malaria parasitisation
in the none or once SP study group compared to twice
SP control group
It also showed that
there was statistically significant higher prevalence of low birth weight LBW in the none or
once SP study group compared to twice SP control group
It also showed that
there was statistically significant higher prevalence of Placental parasitisation in the none or once
SP study group compared to twice SP control group
Recommendation.
Following this study, it is recommended that:
1.
Effort toward improving the uptake of at 2
doses of SP should be improved upon through health education of both patient
and health care givers at all levels of care.
2.
The DOT system for the administration of SP
should be resuscitated, thereby curbing the occurrence of non-compliance by
patients
3.
Monitoring and supervision of the IPT programme implementation should be stepped up at all levels
of health care delivery. This will among others ensure not only that the drugs
are available for administration but also that they are administered to the
patient. .
6. REFERENCES
1. WHO
(1998) World health statistics, 1996. WHO Geneva. (Assessed
15th May, 2010.)
2. Shulman
CE, Dorman EK, Cutts F et al. Intermittent sulfadoxine pyrimethamine to
prevent severe anaemia secondary to malaria in
pregnancy; A randomized placebo controlled trial. Lancet 1999;353:632-636
3. WHO. A
strategic framework for malaria prevention and control during pregnancy in
African Region, Geneva; World Health Organization, 2004 AFR/MAL/04/01(assessed
15th May, 2010)
4. Verhoeff FH, Brabin BJ, Chimsuku L, Kazember P, Russel W.B and Broadhead RL.
An evaluation of the effects of intermittent sulfadoxine-pyrimethamine treatment in pregnancy on
parasite clearance and risk of low birth weight in rural Malawi Am. Trop. Med. Parasitol. 1998; 92(2): 141-50.
5. Parise ME, Ayisi JG, Nahlen BL et al. Efficacy of sulfadoxine-pyrimethamine for
prevention of placental malaria in an area of Kenya with a high prevalence of
malaria and human parasitaemia at the first antenatal
clinic visit in a study of malaria treatment and prevention in pregnancy in
rural Malawi. Am J
Trop Med Hyg. 1998; 55: 17–23.
6. Roll
Back Malaria/WHO. The Abuja Declaration and the plan of
action. An extract from the African summit on roll
back malaria, Abuja, April2000. World HealthOrganization2000WHO/CDS/RBM/2000./;http//www.rbm.who.int/doc/abujadeclaration.pdt
(as assessed 15th May, 2010)
7. Steketee RW, Nahlen BL, Parise M,
Menendez C. The burden of malaria in pregnancy
in malaria endemic areas. AM J Trop med Hyg. 2001; 64(1-2) suppl):28-35
8. Newmann RD, Robalo M, Quakyi I. Malaria during pregnancy: Epidemiology, current
preventive strategies and future directions. Emerging
Infection. November 2004.
9 Steketee RW, Wirima JJ, and Campbell CC. Developing effective strategy
for malaria prevention programmes for pregnant
African women. Am J Trop Med Hyg. 1996; 55 (1 suppl):95-100.
10. World
Health Organization. A strategic framework for malaria prevention and control
during pregnancy in the Africa Region – Brava ville,
Republic of Congo: Regional office for
Africa, WHO 2004. AFR/MAL/04/01 ( as assess 15th May, 2010.)
11. Verhoeff FH, Brabin BJ, Chimsuku L, Kazember P, Russel W.B and Broadhead RL . An evaluation of the effects of
intermittent sulfadoxine-pyrimethamine treatment in
pregnancy on parasite clearance and risk of low birth weight in rural Malawi.
Am. Trop. Med. Parasitol. 1998; 92(2): 141-50.
12. Okonofua FE, Abejide OR. Prevalence of malaria in pregnancy in Nigeria women. J. of Obst. and
Gynae,1364-6893.1996; 16(5): 311-315
13. McCormick
MC. Determinants of low birth weight to infant mortality and childhood
mortality. N Engl J Med 1985; 312:82-90
14. World
Health Organization /UNICEF 2003: Africa Malaria report 2003, World Health
Organization Geneva, 2004. WHO/CDS/MAL/2003, 1093.
(Assessed 16th may, 2010)
15. Berstrom SA Furmadesj Schwalbalch O Ray M. Materno–fetal
transmission of pregnancy malaria and immunoparasitological
study on 202 patient
in Maputo. Gynecol Obstet. Invest. 2004; 35 :103-107
16. Snow
RW, Gou WE, Korenromp EL. Paediatric
mortality in Africa: Plasmodium falciparium as a
cause or risk? Am J. Trop. Med. Hyg.
2004; 71: 16-24
17. Van
den Broek NR, White SA Neilson JP. The
relationship between asymptomatic human immunodeficiency virus infection and
the prevalence and severity of anaemia in pregnant
Malawian women. Am. J. Trop. Med. Hyg. 1998;
59: 1004-1007
18. World
Health Organization. Prevention and management of severe anaemia
in pregnancy: Report of a Technical Working Group.Geneva:1994 WHL/FHE/MSM/93.3
19. Ogbodo SO, Nwagha UI, Okaka ANC, Ogenyi SC, Okoko RO, Nwagha TU. Malaria parasitaemia among pregnant women in a rural community of
eastern Nigeria; need for combined measures. Nig J Physio. Sci. 2009; 24(2): 95-100.
20. Bouyou-Akotet MK, Lonete-Collard
DE, Mabika-Manfoumbi M, Kendjo
E, Matsiegui PB, Mavoungou
E. et al. . Prevalence of Plasmodium falciparum infection
in pregnant women in Gabon. Malaria J 2003; 2: 18.
21.
Nwagha UI, Ugwu VO, Nwagha TU, Anyaehie USB.
Asymptomatic Plasmodium parasitaemia in
pregnant Nigerian women: almost a decade after Roll Back Malaria. Trans. Roy. Soc. Trop. Med. Hyg. 2009; 103: 16-20.
22. Okonofua FE, Abejide OR. Prevalence of malaria in pregnancy in Nigeria women. J. of Obst and Gynae,
1364-6893. 1996; 16(5): 311-315
23. Ojo T, Kuti O, Orji E, Oguniyi S, Sule SS. Comparative
study of the efficacy of pyrimethamine
chemoprophylaxis and intermittent preventive treatment using sulfadoxine pyrimethamine in the
prevention of malaria in the pregnancy in south western Nigeria. J. Chinese Clini Medi. 2007; 2: 9
24. Mokuolu OA, Falade CO, Orogade AA et al. Malaria at parturition in Nigeria
.Current status and delivery outcome. Inf. Dis. In Obstet.
And Gynecol. 2009; article ID 473971,7 pages
25. Bako BG, Audu BM, Geidam AD. Et al. Prevalence risk factors and effect of
placental malaria in the UMTH Maiduguri North Eastern Nigeria: A cross
sectional study. J Obstet Gynaecol. 2009; 29(4): 307-310
26. Daniel,
Jos and Plateau State diff missen 2002. Available at http://www.widernet.org/josproject/josplateauintml
27. Steketee RW, Nahlen BL, Parise ME, Menendez C. The burden of
malaria in pregnancy in malaria-endemic
areas. Am J Trop Med Hyg 2001;64:28—35.
28. McCormick
M. The contribution of low birth weight to infant and
childhood morbidity. New Engl J Med 1985;312:82—90.
29. Guyatt HL, Noor AM, Ochola SA,
Snow RW. Use of intermittent presumptive treatment and insecticide treated bed
nets by pregnant women in four Kenyan districts. Trop Med Int
Health 2004;9:255—61
30. Warhurst
DC, Williams JE . "Laboratory diagnosis of
malaria". J Clin Pathol 1996 ;49 (7): 533–38.
31. Chen Q, Schlichtherle
M, Wahlgren M .Molecular aspects of severe
malaria. Clin. Microbiol. Rev. 2000; 13 (3): 439–50.
32. Lindsay S, Ansell J, Selman C,
Cox V, Hamilton K, Walraven G (2000). Effect of pregnancy on exposure to malaria mosquitoes. Lancet 355 (9219): 1972.
33. Van Geertruyden
J, Thomas F, Erhart A, D'Alessandro U.. The contribution of malaria in
pregnancy to perinatal mortality. Am J Trop Med Hyg. 2004 ;71 (2): 35–40.
34. Elliott
SR, Brennan AK, Beeson JG et al . Placental
malaria induces variant antibody of the cytophylic
subtype immunoglobulin G1 (IgG1) and IgG3 that correlate with adhesion
inhibitory activity. Infection
and Immunity 2005; 73:5903-5907.
35. Fried
M, Nosten F, Brockman A, Brabin
BJ, Duffy PE .
Maternal antibodies block malaria. Nature 1998; 395: 851–852.
36. Duffy
PE, Fried M.
Malaria during pregnancy; parasites, antibodies and chondroitin sulphate- A. Biochem Soc Trans 1999; 27: 478-482.
37. Nwagha UI, Ugwu VO, Nwagha TU, Anyaehie
USB. Asymptomatic Plasmodium parasitaemia in
pregnant Nigerian women: almost a decade after Roll Back Malaria. Trans. Roy. Soc. Trop. Med. Hyg. 2009; 103: 16-20.
38. Klufio CA . Malaria
in pregnancy. PNG Med J 1992;35(4):
249-257.
39. Shulman CE, Graham WJ, Jilo
H, Lowe BS, New L, Obiero J. et al . Malaria
is an important cause of anaemia in primigravidae: evidence from a district hospital in coastal
Kenya. Trans. Roy. Soc. Trop. Med. Hyg.
1996; 90(5): 535-539.
40. Crawley
J, Hill J, Yartey M et al. From evidence to action?:challenge to policy change and programme
delivery for malaria in pregnancy. Lancet Infect. Dis. 2007; 7: 145-155.
41 Riley
EM, Schneider G, Sambou I, Greenwood BM. Suppression of cell mediated
immune responses to malaria antigens in pregnant Gambian women. Am J Trop Med Hyg 1989 ;40:141-144.
42. Rasheed FN, Bulmer
JN, Dunn DT, Menendex
C, Jawla MFB., Jepson, A. et al . Suppressed peripheral blood and placental blood lymphoproliferative
responses in first pregnancies: relevance to malaria. Am J
Trop Med Hyg 1993 ; 48:
154-160.
43. Desai
M, ter Kuile FO, Nosten F, McGready R, Asamoa K, Brabin B, Newman RD: Epidemiology and burden of malaria in pregnancy.
Lancet Infect Dis 2007; 7:93-104.
44. Bouyou-Akotet MK, Lonete-Collard
DE, Mabika-Manfoumbi M, Kendjo
E, Matsiegui PB, Mavoungou
E. et al. Prevalence
of Plasmodium falciparum infection in pregnant women in Gabon.
Malaria J
2003; 2: 18.
45. Deen JL, Von Seidlein L, Pinder M, Walraven GEE, Greenwood
BM. The safety of the combination artesunate
and pyrimethamine-sulfadoxine given during pregnancy.
Trans Roy Soc
Trop Med Hyg.2001; 95(4):
424-428.
46. World
Health Organization. Basic malaria microscopy. Geneva,
Switzerland, 1991, 67-8.
47. Redd
S, Kazembe P, Luby S, et
al. Clinical algorithm for treatment of Plasmodium falciparum malaria in
children. Lancet 2006; 347
(8996): 80.
48. Snounou G, Viriyakosol S, Zhu XP,
Jarra W, Pinheiro L, do
Rosario VE, et al. High sensitivity of detection of human malaria parasites by
the use of nested polymerase chain reaction. Mol Biochem Parasitol 1993; 61:
315-20
49. Snounou G, Viriyakosol S, Jarra W, Thaithong S, Brown KN.
Identification of the four human malaria parasite species in field samples by
the polymerase chain reaction and detection of a high prevalence of mixed
infections. Mol Biochem Parasitol
1993; 58: 283-92.
50. Färnert A, Arez AP, Babiker HA, Beck HP, Benito A, Björkman
A, et al. Genotyping of Plasmodium falciparum infections by PCR: a Comparative Multicentre Study. Trans R Soc
Trop Med Hyg 2001; 95: 225-32.
51. Mens
PF, Schoone GJ, Kager PA, Schallig HD. Detection and identification of human
Plasmodium species with real-time quantitative nucleic acid sequence-based
amplification. Malaria Journal 2006; 5(80): 80.
52. Imwong M, Pukrittakayamee S, Looareesuwan S, Pasvol G, Poirreiz J, White NJ, et al. Association of genetic
mutations in Plasmodium Vivax dhfr
with resistance to sulfadoxine-pyrimethamine: Geographical
and clinical correlates. Antimicrob Agents Chemother 2001; 45: 3122-7.
53. Imwong M, Pukrittayakamee S, Rénia L, Letourneur F, Charlieu JP, Leartsakulpanich U.
Novel point mutations in the dihydrofolate reductase gene of Plasmodium vivax:
evidence for sequential selection by drug pressure. Antimicrob
Agents Chemother 2003; 47: 1514-21.
54. Shiff CJ, Minjas J, Premji Z. The ParaSight-F test: a
simple rapid manual dipstick test to detect Plasmodium falciparum infection. Parasitol Today
1994; 10: 494-5.
55. Shiff CJ, Premji Z, Minjas JN. The rapid manual ParaSight-F test. A new diagnostic
tool for Plasmodium falciparum infection. Trans R Soc Trop Med Hyg 1993; 87: 646-8.
56.
Moody A, Hunt-Cooke A, Gabbett E, Chiodini
P. Performance of the OptiMAL malaria antigen capture
dipstick for malaria diagnosis and treatment monitoring at the Hospital for
Tropical Diseases, London. Br J Haematol 2000;
109:891-4.
57. Moody AH, Chiodini
PL. Non-microscopic method for malaria diagnosis using OptiMAL
IT, a second-generation dipstick for Malaria pLDH
antigen detection. Br J Biomed Sci
2002; 59: 228-31.
58. Murray CK, Bell D, Gasser RA, Wongsrichanalai C. Rapid diagnostic testing for malaria.
Trop Med Int Health 2003; 8: 876-83.
59.
Srinivasan S, Moody AH, Chiodini
PL. Comparison of blood-film microscopy, the OptiMAL
dipstick, Rhodamine-123 fluorescence staining and PCR, for monitoring
antimalarial treatment. Ann Trop Med Parasitol 2000;
94: 227-32.
60. Mayxay M, Pukrittayakamee S, Chotivanich K, Looareesuwan S,
White NJ. Persistence of Plasmodium falciparum HRP-2 in successfully treated
acute falciparum malaria. Trans R Soc Trop Med Hyg 2001; 95:
179-82.
61.
Baird JK, Purnomo, Jones TR. Diagnosis of malaria in
the field by fluorescence microscopy of QBC capillary tubes. Trans R Soc Trop Med Hyg 1992; 86: 3-5.
62. Voller A. The immunodiagnosis
in malaria. In: Wernsdorfer WH, Mc Gregor I. eds. Malaria
Principles and practise of malariology. Churchill Livingstone, Edinburgh, Scotland; 1998, 815-25.
63. Ismail
MR, Ordi J, Menendez C, Ventura PJ, Aponte JJ, Kahigwa E, Hirt R, Cardesa A, Alonso PL. Placental pathology in malaria: a histological, immunohistochemical, and quantitative study. Human Pathol.
2000; 31: 85-93.
64. Romagosa C, Menendez C, Ismail MR, Quintó
L, Ferrer B, Alonso PL, Ordi
J. Polarisation microscopy increases the sensitivity of hemozoin and Plasmodium detection in the
histological assessment of placental malaria. Acta Trop 2004; 90:277-284.
65. Shulman
C, Dorman EK. Reducing childhood mortality in poor Countries:
Importance and prevention of malaria in pregnancy. Trans R Soc Trop Med Hyg 2003; 97:30-35.
66.
Sullivan AD, Nyirenda T, Cullman T et al. Malaria
infection during pregnancy: intrauterine growth restriction and preterm
delivery in Malawi. J infect Dis 1999; 179: 1580-1583.
67.
Araoye MO. Sample size determination. In: Araoye MO (ed)
Research methodology with statistics for Health and Social Sciences. Nathadex pub.
2003; 6 :115-121
68.
Olusanya O, Okpere EE, Ezimokhai M: The importance of social class in voluntary
fertility control in a developing country. W Afr J
Med 1985; 4(4): 205-12.
69. Bodker R, Msangeni
HA, Kisinza W, Lindsay SW. Relationship between the
infectivity of exposure to malaria parasites and infection in the Usambara Mountains,
Tanzania. Am J Trop Med Hyg. 2006; 74: 716–723.
70. Drakeley CJ, Carneiro
I, Reyburn H et al . Altitude- dependent and -independent variation in Plasmodium falciparum prevalence in northeastern Tanzania. J of Infect Dis. 2005; 191: 1589– 1598.
71. Khaemba BM, Mutani
A, Bett MK. Studies of Anopheline
mosquitoes transmitting malaria in a newly developed highland urban area: a
case study of Moi University and its environs. East Afri
Med J.
1994;
71: 159–164.
72.
Nnatu E, Anyiwo CE, Nwobu RU. Malaria parasitaemia
at delivery in Nigeria. East Afri
Medi J. 1987; 64: 44-48.
73.
Erhabor O, Adias TC, Hart
ML. Effect of falciparium malaria on the indices of anaemia among
pregnant women in Niger- Delta of Nigeria. J Clin Med
Research 2010; 2 (3): 035-041
74
Van Den Broek NR, Rogerson
SJ, Mhango CG, Kambala B,
White SA, Molyneux ME. Anaemia
in pregnancy in Southern Malawi: prevalence and risk factors. BJOG 2000; 107: 445-451
75.
Guyatt HL, Snow RW. The epidemiology and burden of
plasmodium falciparium malaria related anaemia among pregnant women in sub- Saharan African. Am J Trop Med Hyg Int Health 2000; 64(1): 36-44
76.
Challis K, Osman NB, Cotiro M, Nordahi
G, Dgedge M, BertrÖm S.
Impact of a double dose of sulphadoxine-pyrimethamine to reduce prevalence of
pregnancy malaria in Southern Mozambique. Trop Med Int
Health 2004; 9(10):1066-1073
77.
Falade CO, Yusuf BO, Fadero
FF, Mokulo OA, Hamer DA, Salako LA. Intermittent preventive treatment is effective
in preventing maternal and placental malaria in Ibadan, South Western Nigeria. Malaria J. 2007; 6: 88-90.
78.
Ukaga CN, Nwoke BEB, Udujih OS et al. placental malaria in Owerri, Imo State
South eastern Nigeria. Tanzania Health Research Bulletin 2007; 9: 3.
79.
Ibhanesebhor SE, Okolo AA.
Placental malaria and pregnancy outcome Int J of Gynae and obste 1992; 32:
247-252.
|
Cite this Article: Ozele, KC; Ozele, N (2022). Anaemia in Pregnancy and Malaria Parasitaemia
in Women at Delivery after 2 Doses of Sulfadoxine-Pyrimethamine
Combination in Jos. Greener Journal of
Medical Sciences, 12(1): 06-24.
|
APPENDIX
APPENDIX A
CONSENT FORM
I, Dr. Ozele, Kingsley Chukwuka of the Department of Obstetrics and Gynaecology,
JUTH, wish to carry out a research on anaemia in pregnancy (low blood level)
and malaria parasitaemia (malaria parasite in the
blood) in women at delivery after 2 doses of sulfadoxine-pyrimethamine
combination (fansidar) at the Jos University Teaching
Hospital, Jos, Plateau State.
Before you decide if
you would like to take part or not, please read the following carefully.
WHAT IS THE STUDY ABOUT?
The study is aimed at
finding out how effective intermittent preventive therapy using sulphadoxine-pyrimethamine in reducing anaemia in pregnancy
and malaria parasitemia
WHAT WILL BE DONE TO YOU IF YOU PARTICIPATE IN THIS
STUDY?
About 5mls of blood
will be collected from your vein and taken to the laboratory and analysed for
malaria parasites and to know your blood level (pack cell volume)
WILL THE INFORMATION BE CONFIDENTIAL? YES
The information
collected during this study will be stored and analysed without including your
name. Only your Doctors will know that
the information is related to you. The
results of the study may be published in medical literature but your identity
will not be revealed.
WRITTEN CONSENT FORM
I……… (Initials please) have read and understood all
the information given to me about my participation in this study and I have
been given the opportunity to discuss it and ask questions. All my questions have been answered to my
satisfaction and I voluntarily agree to take part in this study. I understand that I will receive a copy of
this signed written informed consent form. I authorize the release of my
medical records to the investigator, regulatory authorities and ethical
committee as may be required.
Signature / thumb
print of subject …………………Date…………
Initials of subject
…………………
Signature / thumb
print of witness …………………Date…………..
Initials of witness
………………..
Signature of
investigator ………………………… Date…………………
Initials of
investigator …………….
APPENDIX B
PRO FORMA
ANAEMIA IN PREGNANCY AND MALARIA PARASITAEMIA IN WOMEN AT DELIVERY AFTER
2 DOSES OF SULFADOXINE-PYRIMETHAMINE COMBINATION AT THE JOS UNIVERSITY TEACHING
HOSPITAL, JOS, PLATEAU STATE
1. Serial No ……………..
2. Date…………………..
3. Hospital No ……………..
4. Age (years) ………………
5. Ethnicity (a) Hausa/Fulani (b)Igbo (c)Yoruba
(d) Others (specify) …………………….
6. Place of residence……………………..
7. Level of education (a) none (b) primary
(c) secondary (d) tertiary
8. Occupation (a) housewife (b) student (c)
trader (d) self employed
(e) Civil servant
(specify) ……………….
9. Husband’s level of education (a) none
(b) primary (c) secondary (d) tertiary
10. Husband’s occupation (a) unemployed (b) student (c) trader
(d) Self employed (e) civil servant (specify) ……………….
11. Parity……………….
12. Gestational age at delivery (weeks)………………
13. Maternal height………………………
14. Maternal weight in labour. ……………..
15. When last did you take a worm expeller?
Section 2
Treated for malaria
in this pregnancy? Yes ….NO……..
If yes how many times
……….
What was used in
treatment……..
Sleep under
insecticide treated net? ………..
Section 3
Maternal packed cell
volume…………….
Maternal malaria parasitaemia ………………….
Maternal malaria
parasite density……….
Maternal malaria
parasite species…………………
Placenta malaria parasitaemia....................
Section
4
Weight of baby ………………………………………….
Weight of placenta…………………………………
WORK PLAN
30th
October, 2009: Passed Part 1 Fellowship examination of the West African College
of Surgeons, Faculty of Obstetrics and Gynaecology.
April 2010:
Dissertation topic selected .Supervisors given drafts of proposal for inputs
and correction
June 2010:
Proposal sent to JUTH ethical committee for clearance/approval
3rd
August, 2010: Approval from JUTH Ethical Committee
15th
September, 2010: Dissertation proposal sent to the College
15th
January, 2011: Dissertation proposal received from College. Accepted with major
revision
April
2011: Corrected copy sent back to the College
June 2011
to December 2011: Sample and Data collection and analysis of data:
April
2012: Proposed date for Part 11 Fellowship Examination

