By Nnoka, VN;
Edward, UF; Enebeli, SK; Woha,
BJ; Daka, IR (2023).
Kargasok Tea Consumption and Liver Toxicity in Rats
Nnoka VN1, Edward UF1, Enebeli Sarah Kelechi1, Woha
Boobondah James2, & Daka Iyaeneomi Ransome1
Department of Pharmacology & Therapeutics, College of
Medical Sciences, Rivers State University1
Department of Medical Biochemistry, College of Medical
Sciences, Rivers State University2
|
ARTICLE INFO
|
ABSTRACT
|
|
Article No.: 040223033
Type: Research
Full Text: PDF, HTML, PHP, EPUB
DOI: 10.5281/zenodo.7801966
|
Background - Kargasok tea (kombucha) is a widely consumed tea
(even in Nigeria) made from a symbiotic culture of bacteria and fungi. While
many persons consume kargasok tea because of its
potential benefits like anti-cancer effects, cure for chicken pox,
anti-hypertensive, improvement of poor eye sight, treatment of insomnia etc, some studies have shown that some toxicity exist
following acute and/or chronic consumption. The aim of this study is to
determine the toxicity of kargasok tea on the liver
of rats.
Method - The median lethal dose (LD50)
done earlier using this tea did not produce any mortality even with a dose as
high as 5000mg/kg. The study was carried out using a total of 50 wister rats which was divided into 4 groups. Group 1 (3
rats) was used as control while group 2 to 4 (12 rats each) was the test
group. Kargasok tea extracts was administered to
the rats in groups 2 to 4 for a period of 28days with group 2 receiving
250mg/kg of the extract, group 3 received 500mg/kg while group 4 received
high doses (1000mg/kg). The rat liver was harvested (after sacrifice) every
week under the effect of chloroform anaesthesia for histopathological
evaluation. Also, blood was collected for LFT. The mean values of the
biochemical parameters were evaluated for significant differences using SPSS
23. ANOVA test was employed for calculation of means and standard error
determinations.
Results - Results revealed that the rats that
consumed kargasok tea had increased liver enzymes.
Also, there was histologically distorted liver from the rats that consumed kargask tea. This showed that kargasok
tea has the potential of causing damage to the liver and kidney especially
upon chronic consumption.
Conclusion - Consumers of kargasok tea need to be wary of its deleterious effects
especially from chronic intake as it has been shown to possess the potential
of causing damages to vital organs of the body especially the liver.
|
|
Accepted: 03/04/2023
Published: 05/04/2023
|
|
*Corresponding
Author
Edward UF
E-mail: ucheawaji.edward@ ust.edu.ng
|
|
Keywords: Kargasok tea, Kombucha,
Liver, Toxicity, Rats.
|
|
|
|
INTRODUCTION
Kargasok tea is a symbiotic
culture of bacteria and yeast (SCOBY). Yeast/bacteria fungal symbiont, zoogleal mat, T'chai from the sea, Teekwass, Tschambucco, Zoogleal mat, and
Champagne of life are other names for this beverage.
Kargasok tea is a fermented
tea beverage produced by adding sugar to black tea and letting it to ferment.
The Symbiotic Culture of Bacteria and Yeast (SCOBY) produces
several metabolic products, such as acetic acid and other organic acids, that
are antibacterial in nature and prevent harmful bacteria from infecting the
beverage1.
The fermentation of Kargasok tea is facilitated by a mutualistic connection
between bacteria and yeasts2. It's not as easy to pinpoint the exact
bacterial makeup of kombucha because of how
changeable the bacterial composition of kombucha is.
The inoculum used to brew the tea might have come from a variety of other
places. There are plenty of prokaryotes in the kombucha
culture with the most abundant belonging to two genera – Gluconobacter and Acetobacter.
Acetobacter xylinum is
regarded as the basic bacterium3,4,5. The
cellulose layer's microflora, as reported by Sievers et al. (1995), includes both Zygosaccharomyces
and A. xylinum sp.5 Acetobacter
xylium, A. pasteurianus, A.
aceti, and Gluconobacter oxydans are the most common acetic acid bacteria in the tea
fungus, as described by Liu et al. (1996)6.
Kombucha tea has a plethora
of different yeast species including Koleckera, Pichia, Mycoderma, Saccharomyces,
Saccharomycodes, Schizosaccharomyces,
Zygosaccharomyces, Brettanomyces/Dekkera, Candida, Torulospora.3
Following the
chemical analysis of Kargasok tea, the presence of
the following have being identified – sugars, organic acids such as gluconic acid, citric acid, acetic acid, L-lactic acid,
malic acid, tartaric acid, Vitamins B1, B2, B6,
B12, and C, amino acids, lipids, proteins, polyphenols, as well as
yeast and bacterial metabolites.3,7,8,9
For the preparation
of kargasok tea, about 5 grams of tea leaves are
added to 1 litre of water (5g/L), followed by a
similar amount of sugar (50g for 1 L of water), which functions as a substrate
for the tea fermenting yeasts and bacteria throughout the kargasok
tea making process. Before the SCOBY or a tiny amount of ready-to-drink kargasok tea (starter culture) may be added, the beverage
has to be at a temperature near to 20°C. To avoid unjustified contamination by
undesirable microorganisms, it is crucial to always operate in a clean
atmosphere and only use sterilised tools throughout
the process.1
It is also vital to
control the levels of pH during the fermentation process. Preferably, the
process should be stopped when the pH gets to 4.2 so as to avoid excessive
production of acetic acid which may prove counterproductive.
Kargasok tea fermentation
might vary from culture to culture, kombucha can be fermented in as little as three days and as
long as sixty days.1
Numerous positive
benefits of drinking Kargasok tea have been observed.
According to Dufresne and Farnworth
(2000), the health benefits of drinking Kargasok tea
include: removing toxins from the blood, lowering cholesterol levels, slowing
the progression of atherosclerosis, reducing the risk of stroke in hypertensive
patients, amongst others.10
Antimicrobial11,
12, Antioxidant13 and
Hepatoprotective14, 15 benefits of kargasok
tea have been observed in different studies by researchers.
It is worth noting
that concerns about the safety of herbal medicines and supplements like kargasok tea have increased in recent years along with
their popularity. The risks associated with kargasok
use are nothing new. The increasing consumption of kargasok
in the 1950s coincided with the proliferation of cancer-causing rumours regarding the Russian tea Kwass,
which is quite similar to kargasok. Sunghee et al. (2009) demonstrated lactic acidosis in an HIV
patient who consumed kargasok tea within 12 hours of
presentation.16 Srinivasan et al. (1997)
reported that four people who drank kargasok tea
suffered hepatic and allergic issues.17 After consuming kargasok tea for a month, a 58-year-old woman exhibited
signs of cholestatic liver injury, as evidenced by a
high alkaline phosphatase to aminotransferase ratio and an increased bilirubin
level. Biopsy results confirmed drug-induced hepatotoxicity.18
Aim
To determine the
toxicity of kargasok tea on the liver of rats
MATERIALS
AND METHODS
Animals
Fifty (50) 200g to
360g of male (30) and female (20) wistar rats were
used. They were sourced from the University of Port Harcourt animal house and
allowed to acclimatize for two weeks at the Department of Pharmacology
Laboratory of the University of Port-Harcourt, Rivers State, Nigeria.
Kargasok tea
Kargasok tea was prepared by
fermenting sugared black tea with a SCOBY (Symbiotic Culture of Bacteria and
Yeast). This was in turn orally administered to the animals.
Ethical clearance
Ethical commendation
was obtained from the ethics committee of the University of Port Harcourt. The
animals were handled according to the guidelines of National Institute of
Health guide for care and use of Laboratory Animals.
Experimental Design
This study was
carried out using a total of 50 rats. They were divided into four (4) groups
(group 1 to group 4). Group 1 was used as the control while group 2 to 4 as
test group. Group 2 to 4 had 12 rats each while group 1 had 3 rats only. Tea
extracts was administered to the animals in groups 2 to 4 for a period of
28days with group 2 receiving low doses, that is
250mg/kg, group 3 receiving moderate doses, that is 500mg/kg while group 4
received high doses, that is, 1000mg/kg. The tea was administered once daily.
The liver was
collected from the animals every week under the effect of diethyl ether
anesthesia for histopathological evaluation. Also,
blood samples were collected during this period of sacrifice for the evaluation
of liver function tests (LFT).
Statistical analysis
The mean values of
the biochemical parameters were evaluated for significant differences using
SPSS version 23 (IBM, United States of America). ANOVA test was employed for
calculation of means and standard error determination. P-values were accepted
at ≤ 0.05 significance level.
RESULTS
Figure 1 shows a
graph of mean values of Aspartate transaminase (AST).Compared with normal
control, the AST values of the rats that consumed low dose (250mg/kg) of kargasok tea were higher for week 3 and 4. The AST values
of rats that consumed medium (500mg/kg) and high dose (1000mg/kg) of the tea
were higher for week 2 through to week 4. Statistically significant increases
were found in medium and high dose week 3, all doses for week 4 (p≤0.05).
This showed that increasing the dose of the tea led to an earlier increase in
the AST values. Also, the AST values increased proportionally to the number of
weeks of consumption of the tea.
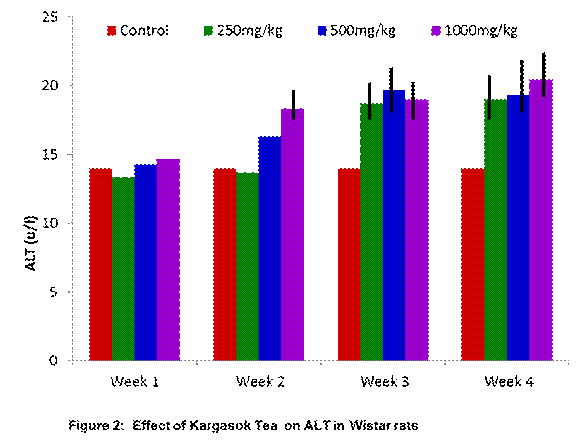
Figure 2 shows a
graph of mean values of Alanine transaminase (ALT). When compared with normal
control, the ALT values of the rats that consumed low dose (250mg/kg) of the
tea were higher for week 3 and week 4; while that of rats that consumed medium
(500mg/kg) and high dose (1000mg/kg) of the tea were higher from week 1 through
to week 4. Statistically significant increases were seen in high dose week 2,
all doses for week 3 and week 4 (p≤0.05). This showed that an increase in
the dose of the tea also led to an increase in the AST level. Also, the more
the number of weeks spent on consumption of the tea, the higher the AST value
was.
Figure 3 shows the
mean values of alkaline phosphatase (ALP).
When compared with
the normal control, the ALP values of the rat that consumed low dose
(250mg/kg), medium dose (500mg/kg) and high dose (1000mg/kg) of the tea were
higher from week 2 through to week 4; although a statistically significant
difference was only observed in high dose, week 4 (p≤0.05). It was also
noticed that this increase was directly proportional to the number of weeks.
Figure 4 shows the mean
values of total bilirubin. There was a slight increase in values among the rats
that consumed medium dose of the tea in week 1, 3 and 4 and also high dose week
2 and 4; however these slight increases were statistically insignificant.




Figure
5:
Photomicrograph of the rat liver showing (A)
control - histologically normal liver with intact hepatocytes (H), Sinusoids
(S) (B) low dose wk 1 (C) low dose wk 2 (D) low dose wk 4- histologically
distorted liver with congested central vein (CV), hepatocytes with different
grades of steatosis.

Figure
6: Photomicrograph
of rat liver showing (A) Medium dose
wk 1 – histologically normal rat liver with patent
central vein (CV), intact hepatocytes and sinusoids (S). (B), (C) and (D) – medium dose wk 2, medium dose wk 3 and medium
dose wk 4 respectively, all showing histologically distorted liver with
congested central vein (CV), different grades of steatosis
(ST).

Figure
7: Photomicrograph
of rat liver showing high dose wk 1 (A),
high dose wk 2 (B), high dose wk 3 (C) and high dose wk 4 (D). These photographs show
histologically distorted liver with congested central vein (CV) and different
degrees of steatosis (ST).
DISCUSSION
From the result of
the liver function tests (LFT), it was observed that the liver enzymes (ALT,
AST & ALP) were increased following consumption of kargasok
tea with ALT and AST showing a greater increase when compared with ALP. These
liver enzymes are usually increased in pathologic conditions of the liver, although
there are non-hepatic causes of elevated ALT and AST like in cases of
hemolysis, myopathy and even exercise; from the result, ALT and AST showed a
greater increase relative to ALP. This is said to occur when there is a
hepatocellular cause for the liver pathology as against a very elevated ALP
relative to ALT and AST which would signify a cholestatic
or hepatobilliary cause.19 Therefore from
this study, it can be inferred that the rats which consumed Kargasok
tea for at least 2 weeks and beyond irrespective of the dose showed signs of
hepatocellular injury. The laboratory results were also backed up by results of
histology which showed distorted liver, hepatocytes with different grades of steatosis with a congested central vein. This is in keeping
with an inflamed liver. This assertion is supported by Srinivasan
et al (1997) who reported onset of hepatic complications in four patients
consuming kargasok tea and that these symptoms ceased
when consumption was halted.17 Gedela et
al (2016) has also observed hepato-toxicity from a
liver biopsy following consumption of kargasok tea.18
Perron et al (1995) also observed elevated liver
enzymes in a 53 yr old college professor who drank half cup of kombucha tea for two weeks of which symptoms and laboratory
values returned back to normal after cessation of the drink.20
CONCLUSION
Consumers of kargasok tea need to be wary of its deleterious effects
especially from chronic intake as it has been shown from this study to possess
the potential of causing damages to vital organs of the body especially the
liver.
The authors share no
conflict of interests.
REFERENCES
1.
Watawana, M.I., Jayawardena,
N., Gunawardhana, C.B., & Waisundara,
V.Y. (2015). Health, wellness and safety aspects of the consumption of kombucha. Journal of Chemistry, 2015, 1-11.
2.
Jarrell
J., Cal T., Bennett J.W. (2000). The kombucha
consortia of yeasts and bacteria. Mycologist, 14: 166-70.
3.
Jayabalan, R., Malbasa, R.V., Loncar, E.S.,
Vitas, J.S., & Sathishkumar, M. (2014). A review on kombucha Tea-microbiology, composition, fermentation,
beneficial effects, toxicity and tea fungus. Comprehensive Reviews in Food
Science and Food Safety, 13, 538-550.
4.
Roussin M.R. (1996). Analysis of kombucha ferments:Report
on growers. Information Resources.
5.
Sievers M., Lanini
C., Weber A., Schuler-Schmid U., & Teuber M. (1995). Microbiology and fermentation balance in
a kombucha beverage obtained from a tea fungus
fermentation. Syst Appl Microbiol, 18: 590-4
6.
Liu
C.H., Hsu W.H., Lee F.L., & Liao C.C (1996). The isolation and
identification of microbes from a fermented tea beverage, Haipao,
and their interactions during Haipao fermentation. Food Microbiol,
13:407-15.
7.
Vitas
J.S, Malbasa R.V, Grahovac
J.A, & Loncar E.S (2013). The antioxidant
activity of kombucha fermented milk products with
stinging nettle and winter savory. CI&CEQ, 19:129-39.
8.
Velicanski A.S, Cvetkovic D.D, & Markov S.L (2013). Characteristic of kombucha fermentation on medicinal herbs from Lamiaceae family. Roum Biotecnol Lett, 18: 8034-42.
9.
Yavari N, Assadi
M.M, Larijani K, & Moghadam
M.B, (2010). Response surface methodology for optimization of glucoronic acid production using kombucha
layer on sour cherry juice. Aust J Basic Appl Sci 4(8):3250-6
10.
Dufresne C, & Farnworth E. (2000). Tea, kombucha,
and health:a review. Food
Res Int, 33:409-21
11.
Greenwalt C.J, Ledford R.A,
& Steinkraus K.H. (1998). Determination and characterization of the
antimicrobial activity of the fermented tea kombucha.
LWT Food Sci Technol,
31:291-6.
12.
Sreeramulu, G., Zhu, Y., Knol, W (2000). Kombucha
fermentation and its antimicrobial activity. Journal of Agriculture and Food
Chemistry, 48 (6): 2589.
13.
Jayabalan, R., Subathradevi, P., Marimuthu, S., Sathishkumar, M., & Swaminathan,
KSM (2008). Changes in free radical scavenging ability of kombucha
tea during fermentation. Food Chemistry. 109; 227-234
14.
Pauline
T, Dipti P, Anju B, Kavimani S, Sharma S.K, Kain A.K,
Sarada S.K, Sairam M, Ilavazhagan G, Devendra K & Selvamurthy W. (2001). Studies on toxicity, anti-stress and
hepato-protective properties of kombucha
tea. Biomed Environ Sci, 14:207-13.
15.
Murugesan G.S, Sathishkumar M, Jayabalan R, Binupriya A.R, Swaminathan K, Yun
S.E. (2009). Hepatoprotective and curative properties
of kombucha tea against carbon tetrachloride-induced
toxicity. J Microbiol Biotechnol,
19(4);397-402.19420997.
16.
SungHee Kole A,
Jones H.D, Christensen R, & Gladstein J (2009). A
case of kombucha tea toxicity. J Intensive Care Med,
24(3):205-207.19460826.
17.
Srinivasan R, Smolinske S, and Greenbaum D
(1997). Probable gastrointestinal toxicity of kombucha
tea: is this beverage healthy or harmful?. Journal of
general internal medicine, 12 (10), 643- 645.
18.
Gedela M, Potu K, Gali VL, and Alyamany K (2016). A
case of hepatotoxicity related to kombucha tea
consumption. South Dakota Journal of Medicine, 69(1):26-8.
19.
Lala V, Goyal A,
Minter DA (2021). Liver Function Tests. In:StatPearls.
Treasure Island (FL): Statpearls Publishing.
20.
Perron AD, Patterson JA, and Yanofsky NN (1995). Kombucha
“mushroom” hepatotoxicity. Ann Emerg Med,
26(5):660-1. Doi: 10.1016/s0196-0644(95)70028-5.
|
Cite this
Article: Nnoka, VN;
Edward, UF; Enebeli, SK; Woha,
BJ; Daka, IR (2023). Kargasok
Tea Consumption and Liver Toxicity in Rats. Greener Journal of Medical Sciences, 13(1): 74-82. https://doi.org/10.5281/zenodo.7801966.
|







