INTRODUCTION
The
Transsphenoidal surgery (TSS) approach has largely been accepted as the gold
standard for resection of sellar/parasellar lesions because of its minimally
invasive appeal.1 Lesions in the cranial bases from the
cribriform plate to the odontoid can be reached via the extended approaches. The use of the microscope in TSS
predates the endoscopic application. However, qualities like having a wide
panoramic view, and better illumination have made the endoscope the preferred
instrument for approaching the complex skull base despite its steep learning
curve.2
Indocyanine green (ICG) fluorescence is an
emerging intraoperative-imaging modality in surgery.3,4 Its application in neurosurgery,
although still evolving, has revolutionized vascular neurosurgery vis a vis
craniotomy for intracranial aneurysms and other vascular malformations. Given
its appealing clinical safety profile of being nonionizing and nontoxic3 as well as easy intraoperative
usability, skull base endoscopists are beginning
to explore its applicability in the endoscopic
endonasal approach (EEA).
Technical
considerations of ICG Endoscopy
Certain
requirements must be in place to perform an ICG examination during EEA. These
include the ICG compound, a high-definition (standard) endoscope and a custom
endoscope with near-infrared light source.
Indocyanine green (C43H47N2NaO6S2)
is a water-soluble tricarbocyanine dye with an absorption and fluorescence
spectrum in the near-infrared (NIR) region. It easily binds to plasma proteins
after intravenous injection, distributing rapidly in the blood with maximum
absorption and fluorescence wavelengths of 780nm and 820nm respectively.5 ICG when given as a bolus injection
(12.5–25 mg) in the peripheral vein is relatively safe, but to be avoided in
patients with iodine hypersensitivity and used with caution in patients with
chronic kidney impairment.6
During
EEA, the nasal and sphenoid stages are usually done in the standard fashion
under white light with high-definition (standard) endoscopes. However, to
perform an ICG examination during the sellar stage, a custom ICG endoscope is
needed in addition.
The custom endoscope is a near-infrared (NIR)
endoscopic system with technical components designed to combine excitation,
emission, and observation of the ICG tracer drug.4
NIR-optimized endoscope has a photodynamic diagnostic (PDD) 3-chip
camera head, and an in-built optical excitation filter to block both ambient
and excitation light so that only ICG fluorescence is captured.4–6 However, some custom endoscopes have
been modified such that the light source can be converted from standard white
light to near-infrared light (ICG mode) with a foot switch.4
Roles
in Endoscopic Endonasal Approach
- Confirmation of
Anatomical landmarks and Neurovascular structures.
Hide
et al5 highlighted the superiority of ICG
over intraoperative neuronavigation and Doppler sonography in the confirmation
of anatomical landmarks during EEA. ICG gives clearer information on vessels
hidden behind the dura mater such as internal carotid artery (ICA), cavernous
and inter-cavernous sinuses, or vessels buried
in bone and tumour, therefore providing for a better orientation. Unlike
neuronavigational systems which have low spatial resolution power for the
detection of vessels that are not clearly defined on computed tomography and
magnetic resonance images, the ICA and patent cavernous sinus are detected with
the ICG endoscope in real-time and at high resolution with a strong
fluorescence signal through the dura mater and the covering thin bone.
ICG
fluorescence signals are strong enough to outline abnormal tumour vessels, and
identify the hypophyseal arteries and small perforators to the brain, optic
nerves, chiasm, and pituitary stalk, confirming their patency during and after
tumour resection.5
With the real-time observation of the blood supply to the optic nerves
and pituitary, preservation of their function postoperatively can be reasonably
predicted. Conversely, although intraoperative doppler ultrasonography can
reveal arterial flow in real-time, it cannot demonstrate the shape of concealed
arteries nor obtain information on the venous systems because of its low
spatial resolution.5 Endoscope-integrated ICG
video-angiography can be useful in verifying the patency of vessels hidden from
microscopic and doppler ultrasonography views.
Hide
and associates5 found the application of ICG during
EEA for resection of craniopharyngioma quite useful in identifying the
pituitary stalk which has a strong fluorescent signal and distinguishing it
from the weak signal of the tumour. Hitherto, this discrimination between the
pituitary stalk and craniopharyngioma would have been difficult resulting in a
higher incidence of damage to the stalk with the attendant sequelae.
- Intraoperative localization of
microadenomas.
Another
important role of ICG in EEA is the intraoperative localization of pituitary
microadenomas. Conventional imaging modalities like the computed tomography
(CT) scan and magnetic resonance imaging (MRI) have low sensitivity in
visualizing pituitary microadenomas. Although MRI dynamic studies and inferior
petrosal sinus sampling (IPSS) have improved the preoperative localization of
microadenomas, the accuracy of localization is still less than 50 per cent and
80 per cent respectively.7,8
The
difference in capillary microvascular densities of pituitary adenomas and
normal glands has been well established and is being explored in the ICG
technique. Following ICG injection, the normal gland and microadenomas can be
differentiated in real-time during surgery because the normal gland has an
increased fluorescence while the adenoma fluoresces less. This was explained by
Kovacs and associates9 who observed that pituitary adenomas have a
reduced capacity to stimulate angiogenesis and have fewer vascular densities
compared to normal glands. Histologically, adenomas have fewer capillaries and
endothelial cells compared to non-tumorous adenohypophysis.9 In light of the foregoing, the use of
intraoperative ICG video angiography facilitates early identification and
complete resection of microadenomas while limiting damages to the normal
gland.
In
addition, both tumoral and en passage vessels are more easily identified; brain
shift and time-related changes during tumour resection are accounted for unlike
in the use of a neuronavigational system.
Overall, these contribute to the improved degree of tumour resection, fewer
complications and better outcomes generally.6,10
- Intraoperative
assessment of vascularized intranasal flap perfusion and viability.
ICG is popular among plastic reconstructive
surgeons in the intra and postoperative assessment of free flaps viability. It has
shown promise in the assessment of the perfusion of vascularized nasoseptal
flaps which is the primary reconstructive option for skull base defects during
EEA.11 This is especially useful in
situations where there may be suspected compromise either in repeat EEA
procedures or following intraoperative iatrogenic injuries like drill injuries.
Geltzeiler and associates11 evaluated the relationship between
ICG enhancement of intranasal flap during EEA and postoperative clinical
outcomes like flap necrosis. Of significance was the finding that, when both
the flap body and pedicle enhanced with ICG intraoperatively, the rate of
postoperative flap necrosis was zero per cent, making it a good tool for
predicting flap viability.
Challenges
in a Resource-limited Developing nation
Endoscopy
application in neurosurgical procedures has come of age. It is however, still
in its budding stages in Nigeria and other sub-Saharan countries.12,13 Nigeria is a developing nation,
largely made up of low-income communities, with limited access to basic
amenities like electricity and well-equipped health care systems as well as
standard neurosurgical facilities. Nigeria has about 80 practicing
neurosurgeons to cater for its population of 200 million, giving a ratio of one
neurosurgeon to 2.7 million Nigerians.14
Despite the
global trend towards the transsphenoidal approach and EEA in particular,1,2 most neurosurgeons
in Nigeria still approach sellar lesions via the transcranial route. This is
because of the numerous challenges associated with setting up and running a
neuroendoscopy unit in Nigeria. These challenges are institutional, technical,
and patient-related.
Endoscopic pituitary surgery is a technical
venture with arguably a higher cost-per procedure compared to the alternative
microscopic transsphenoidal and transcranial approaches.2 It requires huge investments in
equipment procurement, maintenance and personnel training to set up and support
the services. To carry out an intraoperative ICG examination during an EEA,
additional requirements like ICG-compatible endoscopes with NIR visualization, apart from the traditional
endoscopes and ICG injections are needed. All these come at a huge cost and are
currently not available in Nigeria.11,12
There is no political will from the
government, and the private sector is not motivated enough to invest such huge
resources needed in setting up and managing neurosurgical services in general
and neuroendoscopy in particular.12 Compounding the situation, is the
uneven distribution of the health resources in Nigeria due to the low public
health priority given to neurosurgery.
EEA
is a technically challenging procedure, requiring sub-specialization and
intensive training of personnel. The low number of practitioners in the
neurosurgical workforce and the ongoing problem of brain drain in Nigeria makes
sub-specialization in neuroendoscopy difficult.
The average neurosurgeon is preoccupied and overwhelmed with trauma,
infections and other general neurosurgical conditions that neuroendoscopy
appears like a luxury. EEA requires a
steep learning curve which entails large case volumes before one can attain
proficiency. To the authors’ knowledge, there are no neuroendoscopy training
facilities in the subregion, and with a poor referral system in Nigeria, concentrating
a large volume of cases in one center for training is a difficult task.
Patient-related challenges include financial reasons,
illiteracy, and ignorance. Most patients pay out of pocket for healthcare
services in Nigeria due to a lack of insurance coverage. With the high-cost
requirement for the setup of neuroendoscopy services and the absence of
government subsidies, the cost of treatment is expected to be higher than the
traditional transcranial procedure. Illiteracy and ignorance as well as negative
cultural beliefs are important factors that affect the acceptability of
neuroendocopic procedures.12
Addressing these challenges requires a
multifaceted approach. More advocacy with the government and private sector to
show commitment to neurosurgery with legislative back-up. Specialized regional
neuroendoscopy centers should be set up where resources can be concentrated,
and proper referral systems adopted to build a large patient volume for
effective service delivery and training of personnel locally. Endoscopic skill
acquisition laboratories should be set up to facilitate learning outside the
operating rooms.2,12 Younger colleagues
and medical students should be encouraged to subspecialize in neurosurgery by
providing an enabling environment and mentorship.
Case
Illustration
A 77-year-old Japanese woman presented with a four-year history of
uncontrollable secondary diabetes mellitus (Hb A1c 10.3%) attributed to
hormonal disturbance with cortisol of 24.7µg/dl (5 -15 µg/dl), ACTH 88.7pg/ml
(7.2 -63.3pg/ml). This was complicated by diabetic ketoacidosis (RBS of
406mg/dl) and subsequently
electrolyte imbalance (Potassium 2.4mmol/l). Other associated co-morbidities were osteoporosis, hypertension,
dyslipidemia and paroxysmal atrial fibrillation.
Examination findings were blood pressure of 141/93mmHg, hirsutism and
thin skin. Visual
assessment was essentially normal as well as other neurological exams. Other hormonal assessments showed normal thyroid
function and growth hormone but slightly elevated prolactin – 23.7ng/ml (3.6
-16.3ng/ml).
A dynamic contrast-enhanced MRI study highlighted the pituitary
microadenoma (Fig 1). Inferior petrosal sinus sampling confirmed an ACTH-
secreting pituitary adenoma on the left side (peripheral ACTH - 11.6pg/ml,
right IPS- 26.9, left IPS – 44).
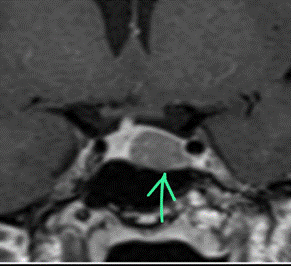
Figure 1. Coronal section, T1 weighted post-contrast dynamic
sequence image of the pituitary fossa showing the tumour, a rounded region of
delayed enhancement (green arrow) compared to the rest of the gland.
She was counselled for total
resection of the tumour through an Endoscopic endonasal approach (EEA). EEA was
performed through the right paraseptal route with the left rescue flap using
the standard endoscope (Fig. 2). ICG was
administered and a custom ICG-integrated endoscope was introduced. The tumour
was visualized as appearing non-fluorescent compared with the normal pituitary
gland (Fig. 3). The adjourning ICA was also visualized (Fig. 4). A soft whitish tumour was resected after
capsulotomy. The anterior and posterior lobes were confirmed and remained
fluorescent with ICG. Pseudo capsulectomy was added. After total resection, hemostasis
was confirmed and the sellar floor was repaired.

Figure 2. Standard
endoscopic view of the sellar floor with the bone removed. Inset of the preop
coronal MRI image.
Post-operatively, the patient’s
serum cortisol and ACTH were normalized as summarized in Table 1. Potassium
levels were also normalized, and BP and blood sugar levels were controlled.
Histological examination confirmed pituitary adenoma. The patient remained free from any symptoms suggestive of
endocrinopathy at six months follow-up visit and no tumour recurrence (Fig 5).
Comment: This case highlights
the advantages of using ICG assistance in the intraoperative localization and
subsequent total resection of a functioning pituitary microadenoma via the EEA
approach in a technologically advanced referral center in Japan, a high-income
society. A complete tumour resection is required in achieving resolution of
hypercortisolism. This ICG technology offers that assistance and precision needed
intraoperatively to separate the tumour from the normal gland as well as
preserve normal glandular functions.
This may be extrapolated to craniopharyngioma
resections where the ICG technology will be helpful in separating the tumour
from the pituitary stalk to achieve gross total resection with less risk of
recurrence as well as preservation of normal pituitary glandular and stalk
functions,
Limitations: ICG use in EEA is a
relatively new and expensive technology. As would apply to all new technologies,
there is lack of class I data associated with its use. However, the preliminary
reports are promising, with its utility and practicality continually being
reviewed.
CONCLUSIONS
The
role of ICG video angiography in EEA is still emerging compared to other
non-invasive intraoperative imaging modalities but shows great promise.
However, these modalities are best used to complement each other rather than in
isolation to achieve the desired outcomes.
The challenges in Nigeria and other
developing countries are enormous and appreciated by the neurosurgical
practitioners who are willing and eager with the help of the government and
other stakeholders to explore these advances in neuroendoscopy for the benefit
of the Nigerian patient.
REFERENCES
1. Alander JT, Kaartinen I, Laakso A, et al. A Review
of Indocyanine Green Fluorescent Imaging in Surgery. Int J Biomed Imaging.
2012;(Oct;2012).
2. Booth GL,
Redelmeier DA, Grosman H, Kovacs K, Smyth HS, Ezzat S. Improved Diagnostic
Accuracy of Inferior Petrosal Sinus Sampling over Imaging for Localizing
Pituitary Pathology in Patients with Cushing ’ s Disease. J Clin Endocrinol
Metab. 1998;83(7):2291-2295.
3. Bruneau M, Appelboom G. Endoscope-integrated ICG
technology : first application during intracranial aneurysm surgery. Neurosurg
Rev. 2013;36:77-85.
4. Emejulu J; Malomo A; Oremakinde A; Onyia C; Opara O;
Iroegbu-Emeruem L; Otorkpa E; Komolafe E; Bankole, F; Shehu B. Developing a
Guideline for Neurotrauma in Nigeria. World Neurosurg.
2020;138(June):e705-e711.
5. Fujimura M, Ikeda H, Takahashi A, Ezura M, Yoshimoto T,
Tominaga T. Diagnostic value of super-selective bilateral cavernous sinus
sampling with hypothalamic stimulating hormone loading in patients with
ACTH-producing pituitary adenoma. Neurol Res. 2005;27(January):11-15.
6. Geltzeiler M,
Carolina A, Nakassa I, et al. Evaluation of Intranasal Flap Perfusion by
Intraoperative Indocyanine Green Fluorescence Angiography. Oper Neurosurg.
2018;15(6):672-676. doi:10.1093/ons/opy002
7. Hide T, Yano S,
Shinojima N, Kuratsu J. Usefulness of the indocyanine green fluorescence
endoscope in endonasal transsphenoidal surgery. J Neurosurg.
2015;122(May):1185-1192.
8. Idowu O,
Doherty A, Tiamiyu O. Initial experience with endoscopic third ventriculostomy
in Nigeria , West Africa. Child’s Nerv Syst. 2008;24:253-255.
9. Ishioka HN. Recent Evolution of Endoscopic Endonasal
Surgery for Treatment of Pituitary Adenomas. Neurol Med Chir (Tokyo).
2017;Apr(57(4)):151.
10. Jugenburg M, Kovacs K, Stefaneanu L, Scheithauer BW.
Vasculature in nontumorous hypophyses, pituitary adenomas, and carcinomas: A
quantitative morphologic study. Endocr Pathol. 1995;6(2):115-124.
11. Kassam AB, Prevedello DM, Carrau RL, et al. Endoscopic
endonasal skull base surgery: analysis of complications in the authors’ initial
800 patients. J Neurosurg. 2011;114(June):1544-1568.
12. Litvack ZN, Zada G, Laws ER. Indocyanine green
fluorescence endoscopy for visual differentiation of pituitary tumor from
surrounding structures. J Neurosurg. 2012;116(5):935-941.
13. Mclaughlin N,
Eisenberg A, Cohan P, Chaloner C, Kelly D. Value of endoscopy for maximizing
tumor removal in endonasal transsphenoidal pituitary adenoma surgery. J
Neurosurg. 2013;118(March):613-620.
14. Uche EO, Okorie E,
Emejulu J, Ajuzieogu O, Uche NJ. Challenges and outcome of cranial
neuroendoscopic surgery in a resource constrained developing African country. Niger
J Clin Pract. 2016;19:811-815.
|
Cite this
Article: Iroegbu-Emeruem, L; Mbadugha, T; Ogiwara, T
(2024). The Role of Indocyanine Green Fluorescence in Endoscopic Endonasal
Approach: Challenges in a Resource-Limited Developing Nation. Greener Journal of Medical Sciences, 14(1): 8-14.
|





