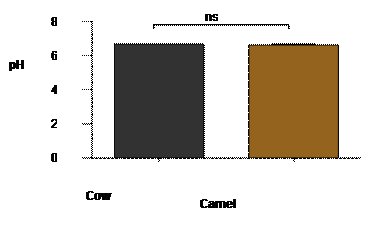INTRODUCTION
Cheese
can be defined as a consolidated curd of milk solids in which milk fat is entrapped
by coagulated casein (Adam and Moss, 2000). This description comprises a
heterogeneous group of fermented milk-based products, which are elaborated worldwide
and present a broad range of flavours and forms. The
main ingredient in cheese is milk. Cheese is made using cow, goat, sheep,
buffalo or a blend of these milks. The type of coagulant used depends on the
type of cheese desired. For acid cheeses an acid sources such as acetic acid
(the acid vinegar) or gluconodelta-lactone (a mild
fool acid), is used. For rennet cheese, calf rennet or, more commonly, rennet
produced through microbial source, calcium chloride is sometimes added to the
cheese to improve the coagulation properties of the milk (cheese production/milk Facts.info). Aspartic
proteases, also known as acidic proteases have been isolated from diverse
sources, including virus, bacteria, fungi plants and animals (Chen et al., 2009). They are use as
milk-coagulating enzyme for the manufacture of cheese. Hence some of the
aspartic proteases are termed as microbial rennet like milk clotting enzymes
that catalyse milk coagulation substituting calf
rennet, which is the main enzyme employed in cheese production (Patilpallavi et al.,
2012). Milk coagulation is a basic step in cheese manufacture using Mucor and Penicillum
strains mainly as adjuncts in different types of cheese, and in these recent
years other strains like Rhizopus
prove interesting as producers of rennet – like
enzymes (Abebe et
al., 2014). The worldwide increase of cheese production coupled with a
reduced supply of calf rennet has prompted a search for calf rennet substitutes
including microbial and plant rennet (Cavalcanti et al., 2004). The finding of this research would create awareness and job opportunities
in dairy industries for teaming population. It would also developed a protocol
of preserving milk through cheese production aver along period of time.
Therefore, this study aimed at production of cheese from camel and cow milk
and combination of both using the extracted enzymes (EE) produced and compare
with commercial rennet. And further evaluate sensory perception of cheeses
produced using Hedonic scale for individual responses.
MATERIALS AND METHODS
Collection and Analysis
of milk Sample for cheese production
Ten
(10) liters each of camel and cow milk were collected early morning from free
range camel and cow herds at a Fulani settlement in Sokoto.
The milks were sieved in a double layered cheese cloth and poured in plastic
containers surrounded with ice block and transported to laboratory, for pH
determination, mineral analysis and cheese production.
Production and
Purification of Enzymes for Cheese Making
Aspergillus specie was subjected to Solid State
Fermentation (SSF) according to the method of sirividya
(2012) with a little modification using Banana Peel Powder (BPP) as substrates
to produce protease that was used for comparative cheese making. After
fermentation, enzyme protease was extracted using the method of Sirividya (2012) and purified according to the method of Ramachandra and Arustselve
(2013).
Assay of enzyme and
Protease activity (Milk clotting activity)
The
Milk clotting activity (MCA) of the enzyme extracted was measured by method
described by Otani et al, (1991) and the milk-clotting activity was calculated by
using the formula of Kawai and Mukai (1970). Protease
activity of each protease was measured using the method of Kunitz
(1947) which was adopted by kademi et al. (2013).
Soft Cheese Making and
yield determination
Partially
purified enzymes of Aspergillus
tamarii
(EE) were used for the preparation of comparative milk curds. A 500ml of fresh
milk each of cow (CWM) and camel (CM) were poured separately into eighteen (18)
experimental buckets which were labeled according to the type of milk and coagulant.
The cheese produced from each experiment was weighed on an electrical weighing
balance; percentage cheese yield was thus calculated using the formula:
Grams
of cheese produced X 100
Grams
of milk used
Akinloye and Adewomi,
2016
Sensory Evaluation of
the Soft Cheeses Produced
Each
produced cheese was examined after 72 hours of ripening and sixty member
trained panelists were employed for the sensory tests. Evaluation was based on
five attributes: colour, taste, flavor, texture and
overall acceptability, scores were based on 9 - point hedonic scale (Badmos and Joseph, 2014)
RESULTS
Figure
1 revealed that, there is no significant difference between pH values 6.7 and
6.6 of cow and camel milk.
DISCUSSION
pH result for CWM 6.7 is within the East
African community 2006 reference range of 6.6 to 6.8, it is also very close to
the mean pH of cow milk samples reported by Gwandu et al, (2018). While pH 6.6 for CM is
within the range of pH (6.4-6.7) reported by Wangoh
(1997) for fresh camel milk, which further stated that it was similar to that
of sheep milk, but slightly lower than that of Bovine milk.
The MCA and MCT of the supernatant
enzymes from BPP treated with A. tamari
supernatant proved to have a highest MCA of 351.2SU/mL with a short clotting time
of 382 sec. The shortest clotting time
of A. tamari is related to the
findings of Benlounissi et al., (2012) who reported that A. tamari and A. niger fermented industrial
whey of cheese to clot milk within 5 minutes. Mirian et al., (2011), also reported high
clotting activity from A. ochraceous. Sethi et al, (2016) identified banana peel (Musa paradisiaca L.
Family: Musaceae) as most suitable for proteases
(pectinase) biosynthesis with activity of 6500±1116.2 µg/L. The milk clotting
activity 351.2 SU/ML from Aspergillus tamarii strain is also related MCA of 398.16 SU/ML from M. circinelloides
was reported by Bensmail et al. (2019) using wheat bran as substrate.
The PA generated 0.35 U/ML for the
enzymes of A. tamari on treatment
with BPP are very low when compared with the findings of Rodate
et al., (2011). Protease activity of
13.82U and 20.90U for Aspergillus ochracious
and Aspergillus dimorphicus
isolated from fruits and use casein as substrate to assay for their PA at 30oC,
pH 7.0 for 2 hours. The time coarse and pH could be a reason for the variation,
that is 30 minutes, pH
6.0 (used in this research)
The soft cheese
produced from CM + CR was extremely white and CM + EE cheese was also white.
This is contrary to the report of Shahein et al., (2014) who noted no
difference in appearance of CM+BM (buffalo milk) and their individual
respective milks soft cheese. The difference in the cheese texture could be
attributed to the initial composition of the milk, e.g., high acidity, proteins
and total solids contents generally make cheese harder and less easily deformed
(Kehagis et
al., 1995). The reason why cheese produced from CWM and combinations was
more formed and a bit harder than the cheese produced from CM was because the
protein and total solid contents were more in the CWM than in the CM (as
indicated in physiochemical properties of the two milks). The tastes of the
cheeses were typically slighter to moderately sour for all except the product
produced from CM + CR, which was having a kind of bitterness prior to swallowed
and remained some times after swallowing. However, Ramet
(1994) highlighted that temporary bitterness has been noted in some soft and
semi-hard cheeses. The defect has been detected mainly after the cheese has
been swallowed, adding that bitterness in dairy products might be caused by
factors such as alkalis of ingested plants, salts of external origin (mainly
calcium and magnesium chlorides) and carbonates or bitter peptides (generated
by casein hydrolysis) and presence of proteolytic
residues, which accumulate when the pH of the cheese is low and a high residual
proteolytic activity from clotting enzymes remains in
the curd.
The soft cheese produced from CM + CR
is extremely white; CM + EE cheese is also white, this is contrary to the
report of Shahein et
al., 2014 who noted no difference in appearance of CM+BM (Buffalo milk) and
their individual respective milks soft cheese. The sensory evaluation by 60
panelists has revealed that individual CM cheese was the least choice where by
CM + EE occupied 5th position and CM + CR 6th position by
kruskal Wallis mean ranking. While Siddiq et al.,
(2016) reported that mixing CWM with CM in the ratios 1:1 to produced Jibnabeida cheese did not decrease the acceptability of the
final product.
CONCLUSION
The
strain Aspergillus
tamarii isolated in this study is versatile producer
of extra-cellular protease that produced EAs at pH 5.0 (0.309±0.21 U/mL), 6.0
(0.351±0.096U/mL) and 8.0 (0.146±0.020). Hence, it could be exploited as
bioactive agent and could further be checkmate for further industrial usage.
The study revealed that protease of Aspergillus tamari
had produced cheese which is the best of panelist choice from cow milk and
ranked 1st by Kruskal Wallis test.
Findings in this study indicated that in terms of cheese production CM yield a
better product when combined with CWM at 1:1 ratio. There is need for soft
cheese producers to use coagulant that promote taste and texture as they are
the attribute that indicated significant difference in the sensorial evaluation
of the cheeses produced.
ACKNOWLEDGEMENT
We wish to
express our sincere gratitude to Tertiary Education Trust Fund (TETFUND, Abuja)
for sponsorship of this research work.
CONFLICT OF INTEREST
The authors have declared that no competing interests exist.
REFERENCES
Abebe B, Abraham S, Genet, A., Hiwot, G., pavlos, K. and Mekse, A. (2014) Isolation, optimization and
characterization of bacteria from soil water in Gondar town, North West Ethiopia
International Journal of Bacteriology,
Virology and immunology 1 (3):020 – 024
Adam,
M.R, and Moss, M.O (2000) Food Microbiology (Second edition). The Royal Society of Chemistry
Cambridge Aftab, Md. U., Hassan, Md.M.,and rasheed, N (2010) Isolation and identification of
pathogenic Escherichia coli, Klebsiella spp. and
Staphylococcus spp. in Raw Milk Samples collected from
different areas of Dhaka City
Akinloye, A. M., and Adewumi, O. O. (2016). Effects of local coagulants on the
yield of cheese using cow and sheep milk. International Journal of
Development and Sustainability, 3, 150–161.
Badmos A.H and Joseph, K (2014) Effect of
supernatants of various cereal and cereal mixture on yield and acceptability of
soft Cheese Best Journal 11(2): 1-5
Benlounissi, A., Mechakra-maza, A., Gherib, Z.,
Mahfouz, M., Loic, J. and Christope,
A. M., (2012).
Milk-clotting fungus enzyme production: A sustainable development approach
based on whay recycling. African Journal of Biotechnology.2 (8); 1997-2005
Bensmail; boudjema, K. and Naimi-fauzourane
F. (2020). Production of extracellular Rennen-like
enzyme by a newly isolate mucorcircinellolder (von tieghem) and its application in camembert cheese making.
Journal of applied biotechnology reports 7(1);16-24 doi 10.30491/JABR.20202.105914
Cavalcanti M.T.H, Teixeira
MFs, Linafiho J.L, and Proto ALF (2004) partial
purification of new milk – clotting enzyme produced by nocardiopsis
sp. Biores. Techn. 93, 29-38
Chen,
J., Ouyang, Y., Wang, L, Xie,
W. and Zheng, Q (2009) Aspertic
Proteases gene family in rice: Gene structure and expression, predicted protein
features and Phylogenic relation.
Journal of Gene 442:108-18.
Gwandu S. H., Nonga
H. E., Mdegela, R. H., Katakweba
A. S., Suleiman T. S and Ryoba R (2018). Assessment of
Row Cow Milk Quality in smallholder Diary farms in Pemba Island Zanzibar, Tanzaniya. Veterinary
Medicine International 20(8); 18-24
Kademi, F., Abachi, S., Mortazavi, A., Ehsani, M. A. Tabatabaci, M. R.
and Malekzadeh, F. A (2013) Optimization of fungal
rennet production by
local isolate of Rhizomucor nainifalensis under solid substrate fermentation system. IQSR Journal of Pharmacy and Biological Sciences 5(2); 115-121
Kawai,
M. and Mukai, N. (1970). Studies on
Milk-clotting enzymes produced by Basidiomycetes.
L. screening tests of Basidiomycetes for the
production of milk clotting enzymes. Agric Biology and
Chemistry, 34:159-163
Kehagis C., Kovlovris
S., Samano A. Mallious S.
and Kovmoustsos G (1995) Effect of various starters
on the quality of cheese in Brine: Food
Microbiology 12:413-420
Kunitz, M. (1947) Crystalinesoyabean trypsin inhibitor II. General properties.
Journal of General
Physiology. 30:291-310
Mirian, P.R, Disney, R.D, Daniel M.V and Rosena, F (2011) Filamentous Fungi Isolated from coffee
fruit (coffee grubics L.) Acta Scientiarum AhUmming,
C.Masamba,gronomy Mariya 33(3) 457-464.
www.sci.br>pdf>asagr
Otani, H, Mitsuhiro, I.
and Akiyoshi (1991). The screening of
trees having milk activity. Animal
Science Technology, (62): 417
Patilpallavi, M., Kulkarni,
A, and Kingepallari, T. (2012) Production of milk –
clotting enzyme from Aspergillusoryzae under solid –
state fermentation using mixture of wheat bran and rice bran int. J. of scientific and research publications 2
(10)
Ramachandra, N. and Arutselvi,
R. (2013) partial purification and characterization of protease enzyme from Nomuraerileyi International Journal of pharmaceutical
sciences and research 12(6); 1-7
Ramet J. P (1994). The scientific aspect and specific
technology for the production of camel milk cheese. Proceedings of the Workshop on Domendary and
camel milk,
Rodate, M. P., Dias, D. R., Vilela, D. M., and Schwan, R. F. (2011). Proteolytic activities of bacteria, yeasts and filamentous
fungi isolated from coffee fruit (Coffeaarabica L.). Acta Scientiarum and
Agronomy, 33(3), 457-464.
Sethi, B. K., Nanda, P. K., and Sahoo, S. (2016). Enhanced production of pectinase by Aspergillusterreus
NCFT 4269.10 using banana peels as substrate. Biotechnology,
6(1) 36.
Shahein, M. R., Hassanein, A. M., & Zayan, A. F. (2014). Evaluation of
soft cheese manufactured from camel and buffalo milk. World J. Dairy Food Sci, 9, 213-219.
Siddiq, S. M., Sulieman,
A. M. E., Salih, Z. A., & Abdelmuhsin,
A. A. (2016). Quality characteristics of white cheese (Jibnabeida)
produced using camel milk and mixture of camel milk and cow milk. International
Journal of Food Science and Nutrient, 6, 49-54.
Sirividya, S. (2012)
production and characterization of an acid protease from a local Aspergillus Sp.
By solid- state fermentation scholars research
library; Archives of applied Science
Research, 4(1): 188 – 199.