By
Aladekoyi, G; Ajala, OA; Shakpo, OA; Adesina, TA (2024).
The Effect of Shea
Butter Oil and Moringa Seeds Oil Waxing on The Post-Harvest Life of Cucumber Fruit (Cucumis sativus L.)
Aladekoyi, Gbenga1;
Ajala, Olufunmilola A.2; Shakpo, Olubunmi I.2; Adesina, Tosin A3
1Department of Science
Laboratory Technology, Rufus Giwa Polytechnic, Owo, Ondo State, Nigeria.
2Department of Science Laboratory
Technology, Rufus Giwa Polytechnic, Owo, Ondo State, Nigeria.
3Department of Nutrition
and Dietetics, Rufus Giwa Polytechnic, Owo, Ondo State, Nigeria.
|
ARTICLE INFO
|
ABSTRACT
|
|
Article No.: 062424088
Type: Research
Full Text: PDF, PHP, HTML, EPUB
DOI:
10.15580/gtfsn.2024.1.062424088
|
The ripening of a Fruit is a genetically programmed process which
leads to an assortment of metabolic and physiological variations and changes
that permanently alter its characteristics. Post-harvest study was carried
out on Cucumber
Fruit (Cucumis sativus L.) for
fifteen days using Shea Butter Oil and Moringa Seeds Oil and their
composite in ratios of 20:80 and 80:20 Waxing in day one and studied their
physicochemical properties for ripening for a period of fifteen days. Though, Fruit maturation and ripening depends on
species and can either be climacteric or non-climacteric, but both cases
depends on metabolic shift from normal development conditions toward the
fully mature state, and this can be checked by waxing. a credible measure for preserving Cucumis sativus L was established at fifteen days with the application of 80 %
Shea butter oil and 20% Moringa oil composition. However, at 100% Shea butter
oil also showed a good preservative measures, but may not exceed fifteen days
before deterioration sets in. Application of 80 % Shea butter oil and 20%
Moringa oil composition has proved to be the best waxing method for
post-harvest treatment of Cucumis
sativus L. The observed
preservative measures elucidated in this research for cucumber post-harvest
for fifteen days validate their qualities for consumption with good retention
of their nutritional composition.
|
|
Accepted: 26/06/2024
Published: 05/07/2024
|
|
*Corresponding Author
Aladekoyi,
Gbenga,
E-mail: gbengu7@ yahoo.com,
gbenga_aladekoyi@ rugipo.edu.ng
Phone: +2348034651804
|
|
Keywords: Cucumber,
Post-Harvest, ripening, waxing, Quality.
|
|
|
|
INTRODUCTION
Nigeria
grows cucumbers (Cucumis sativus L.)
year-round because of their many applications, making them one of the most significant
and adaptable vegetables. Nigerian consumers' demand for cucumbers has been
rising recently as a result of growing knowledge of their numerous health
advantages, which include improved skincare (Umeh and Ojiako, 2018).
Inappropriate agronomic practices, pests, diseases, and
high fruit perishability—exacerbated by climate change—severally limit
achievable yields and overall productivity in farmer's fields, despite the
growing recognition of cucumbers as one of Nigeria's major vegetables. In general,
increased rainfall variability and temperature fluctuations brought on by
changing weather patterns that resulted in a changing climate have threatened
agricultural productivity (Somarribaa et
al., 2013).
Protected cultivation, an agro-technology, is one way to
address this. It entails covering the crop to enable control over macro- and
microenvironments, promoting optimal plant growth and development, extending
the duration of growth, inducing earliness, and improving yield and quality
(Gruda and Tanny, 2014, 2015). Nigeria, a country where seasonality and produce
perishability are persistent problems, has good opportunities for vegetable
production thanks to greenhouses, which are protected cultivation methods.
Due to its high value and low volume, cucumbers can be
grown on a commercial scale in greenhouses, which can increase growers' income
through increased productivity. According to Smitha and Sunil (2016), protected
cultivation is the most effective way to boost cucumber production because it provides
a less restrictive environment for plant growth and development than open field
conditions.
1.1 Taxonomy and adaptation of cucumber
One of the
most significant plant families is the Cucurbitaceae, which includes the
cucumber (Cucumis sativus L.). There
are 750 species and 90 genera in the Cucurbitaceae family. About 40 species
make up the genus Cucumis, three of which are widely cultivated: C. anguria L., C. sativus (cucumber), and C. melo L. (cantaloupe). Cucurbita pepo L., C. mixta Pang., C. moschata
Poir., and C. maxima Duch. are some of the other significant crop plants in
the Cucurbitaceae family, along with watermelon (Citrullus vulgaris), muskmelon (Cucumis
melo L.), squash, and pumpkin (Golabadi et
al., 2012).
1.2 Uses of cucumber
In Nigeria, cucumbers are used to make vegetable salads,
stews, and sandwiches. They can also be eaten raw as a relish. Because cucumber
fruit is primarily composed of water, bodybuilders are advised to use it as a
natural diuretic. It has low potassium and vitamin C content (Keith, 1999).
There is some vitamin A in the skin. In addition, the fruit is utilized to make
body lotions, shampoos, and face masks. When consumed regularly, it helps treat
kidney diseases and lower high blood pressure to more healthy levels. Its juice
is frequently advised as a silicon source to enhance skin health and tone.
1.3
Nutritional value of cucumber
According to Keith (1999), potassium, pantothenic acid,
magnesium, phosphorus, copper, manganese, and vitamins A, C, K, and B6 are all
abundant in cucumbers. Also, Steven et
al. (1985) stated that cucumbers' ascorbic acid and caffeic acid can lessen
skin irritation and swelling.
In addition to its high
potassium and magnesium content, fiber-rich skin, and ability to help eliminate
uric acid, cucumbers are good for people with arthritis because they lower
blood pressure and support healthy nutrient functions. Cucumbers' high
magnesium content also has good levels of flavonoids, which are potent
antioxidant compounds that relax muscles and nerves while maintaining healthy
blood circulation (Keith, 1999). Cucumbers ('Manar'), in contrast to European
cucumbers, are much smaller, typically weighing less than 100 g and measuring
between 125 and 175 mm in length.
1.4 Production of cucumber
Africa's
cucumber harvest season varies depending on the region and type of production
method. According to Obeng-Ofori et al.
(2007), the production system can be either an open-field system (21 pickling
cucumbers and fresh market) or a protected cultivation system (fresh market
only). Producing in open fields will shorten the harvest season and lower the
commodity's quality. On the other hand, production in protected culture systems
can produce fruit of superior quality and prolong the harvest season throughout
the year. According to Mrema and Rolle (2002), the harvest season in Africa is
lengthy for field-grown cucumbers, lasting from mid-September to June,
depending on the growing region.
1.5
Production constraints of cucumber
Africa's developing
nations face an immediate challenge from the growing demand for cucumbers. Lack
of sufficient rain, a lack of skilled labor, inexperienced management, and
improper cultivation and transportation methods are the primary obstacles to
cucumber production in Northern Nigeria (Ashby, 2000). Cubbit downy mildew is
caused by pseudoperonospora cubensis
(Mrema and Rolle, 2002) Rostovtsev. It is among the most severe diseases
affecting cucurbit crops in the world. Cucumis
melo L., cucumber (Cucumis sativus
var. sativus L.), squash (Curcurbita
ssp.), watermelon [Citrullus lanatus],
and melon (Cucumis melo L.) are among
the crops infected by it.
Globally distributed Pseudoperonospora cubensis is a major cause of yield losses in the
USA, Europe, and Asia. It has been discovered in more than 70 nations and a
variety of habitats, from semi-arid to tropical (Cohen, 1981). In 20 genera, it
can infect more than 50 distinct species.
P. cubensis overwinters in warmer climates because it cannot withstand
freezing temperatures, unlike its cucurbit hosts. Agricultural productivity is
also hampered by intercropping, inadequate planting techniques, inadequate
weeding, inadequate irrigation methods, and poor soil preparation.
1.6 Soil and
climatic requirement of cucumber
The range of
temperatures for optimal growth is 20 to 25 ℃; below 16 ℃ and above 30 ℃, growth is
reduced. Nonetheless, studies have indicated that cucumbers' heightened female
tendencies are fostered by cooler temperatures and shorter days. Because the leaves
have a large surface area, the plant needs high humidity (Eifediyi and Remison,
2010).
1.7
Cultivation of cucumber
To grow
cucumber seedlings that will be transplanted in the field, it is best to sow
the seeds in nursery bowls or nursery beds. Typically, the seeds are drilled
with a soil pH of 6.0 to 7.0, at a distance of 1.5 to 2.5 m, x 60 cm x 90 cm
plant spacing. In each pit, two to three cucumber seeds are sown using the ring
or base method. According to Eifediyi and Remison (2010), the bed should be
raised to a height of approximately 5 cm to allow for proper drainage, and its
recommended dimensions are 4 by m or 2 by m with a spacing of 1 m between the
beds.
The growing season for cucumbers is comparatively brief,
lasting more than 70 days for varieties grown in greenhouses and 55 to 60 days
for field-grown varieties. The best soils for growing cucumber seeds are those
that are deep, fertile, rich in humus, free-draining but retain moisture, and
devoid of nematodes. It is recommended to plant the seed on soils with high
rates of water infiltration and moisture retention. Around 10 to 20 male
flowers appear on a typical cucumber plant for every 10 to 20 female flowers
that will bear fruit. At the nodes, flowering is gradually set (Kader, 1994).
1.8 Pest and diseases of cucumber
Many pests and diseases, as well as weeds, can lower
cucumber yields. For instance, this crop is known to be infested by more than
40 different diseases. When appropriate cultural practices have not been
followed and growing conditions are subpar, cucumbers are particularly
vulnerable to pest attacks. Cucumber scab (fungus), angular (bacterium),
powdery mildew (fungus), damping off (fungi), and mosaic (virus) are some of
the common diseases that affect the vegetable. Important pests include cucumber
beetles, cutworms, tarnished plant bugs, seed corn maggots, and two-spotted
spider mites (US Department of Agriculture, 1985).
1.9 Harvest and yield of cucumber
Approximately two-thirds of the national production of
cucumbers is machine-harvested; fresh cucumbers are harvested manually, while
processed cucumbers are also harvested mechanically. As they reach commercial
maturity, cucumbers grown for the fresh market are harvested, allowing for
multiple harvests each season. The primary determinant of postharvest quality
is the maturity at harvest (Kader, 1994). Color, shape, size, and appearance
(free of defects, damage, and decay) are the subjective factors used to
determine a cucumber's maturity (Kader, 1994; U.S. Standards for Grades of
Greenhouse Cucumbers, 1999). The cucumbers are chosen by hand.
1.10 Post-harvest losses of cucumber
i.
Chilling injury: When fruits or
vegetables that are susceptible to chilling are exposed to low but not freezing
temperatures, a physiological condition known as chilling injury can develop
(Eifediyi and Remison, 2010). Although the primary sites of storage disorders,
including chilling injury expression, are thought to be cell membranes, a
biochemical pathway to clarify the mechanism of chilling injury has not yet
been identified. According to Eifediyi and Remison (2010), chilling injury is a
cumulative process whose severity varies with exposure duration and
temperature. Cucumbers must be exposed to chilling temperatures for several
days for chilling injury damage to become visible (Eifediyi and Remison, 2010).
Visual symptoms might not appear until the fruit is moved to a higher storage
temperature. Despite being chilly
ii. Ethylene injury:
In
climacteric fruit, ethylene is necessary to finish the ripening process; in
non-climacteric fruit, it is not. From a marketing perspective, ripening is not
necessary for cucumbers because they produce little to no ethylene after
harvest and do not experience a concurrent rise in respiration rate (Eifediyi
and Remison, 2010; Kader, 2005).
Ethylene causes negative reactions in cucumbers, and the resulting changes are
considered harmful. It is well known that the gaseous plant hormone ethylene controls
a variety of physiological and developmental processes in plants (Kader, 2005;
Huang et al., 2009). Ethylene
influences sex expression in cucumber plants, but its effects on fruit tissue
are considered harmful because they decrease consumer acceptance of the product
as reported by Eifediyi and Remison, (2010).
. 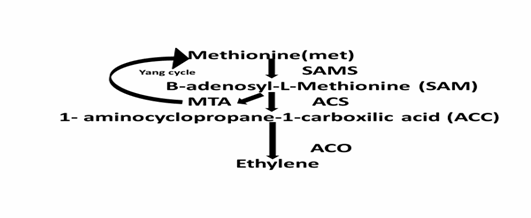
Fig.
1: Ethylene biosynthesis from methionine (Source: Valeria et al., (2014)
1.11 Storage of cucumber
Food and
Agriculture Organization (2008) and USDA (2010) state that a commodity's
shelf-life is reliant on the postharvest treatments it receives because the
quality of the commodity begins to deteriorate after harvest. According to
Kader (2005) and Kitinoja and Gorny (2009), temperature control is typically
the most useful and successful method for extending the postharvest life of a
variety of horticultural commodities, including cucumbers. Cucumbers should be
stored at 7 to 10 ºC and 85 to 95% relative humidity (RH) in the air (Kitinoja
and Gorny, 2009), 8 to 12 ºC in 1 to 4% 02 and 0% CO2, or 10 to 12.5 ºC (Kader,
2005) because of their chilling sensitivity.
Not only will storing the commodity below the recommended
230C storage temperature reduce its quality and shelf life, but it will also
incur unnecessary expenses. Managing the temperature appropriately is also
crucial for food safety and marketing. Food purveyors require suppliers who can
deliver products that are both safe and of consistent quality throughout the
year, partly due to consumer pressure and other competitive market forces.
As a result, growers and food handlers need to make sure
that all actions are in accordance with the suggested protocols for that
specific commodity. Using protective shrink-wrap films, like polyethylene
films, to shield greenhouse-grown cucumbers from excessive water loss and the
ensuing shriveling is another crucial part of cucumber storage. According to
Cong et al. (2007), there is evidence
that the modified atmosphere effect of film wrapping can prolong the shelf life
of certain fruits.
Because wrapped fruit has higher concentrations of 24
polyamines, modified atmosphere packaging has also been shown to lessen the
severity and start of chilling injury in wrapped cucumbers (Cong et al., 2007).
1.12
Waxing and Surface Edible Coatings
The process
of waxing involves applying artificial and natural waxing material to fruits
and vegetables, including tomatoes, cucumbers, watermelons, apples, and garden
eggs (Joyce et al., 1995; Sabir et al., 2004). Saftner (1999) has also
reported on the potential of fruit coating and treatment to enhance the storage
and shelf life qualities of Gala and Golden Delicious apples. Petroleum wax,
paraffin wax oil, and other synthetic waxing materials are examples of
artificial waxing materials. Natural waxing materials include beeswax, carnauba
wax, candelilla wax, montane wax, palm wax, rapeseed wax, soy wax, rice bran
wax, olive wax, sunflower wax, and coconut wax. Waxing is done for two reasons:
to improve appearance and stop water loss, which delays spoiling and shrinking
(Keith, 2003).
Tomato freshness preservation seems to be improved by
using edible coatings in combination with low-temperature storage (Gonzalez-Aguilar
et al., 2010). The creation of a modified environment surrounding the product
acts as a partial barrier to oxygen (O2), carbon dioxide (CO2),
water vapor, and aroma compounds, reducing the rate of fruit respiration and
water loss while maintaining texture and flavor (Olivas and Barbosa-Canovas,
2008). This is the mechanism by which edible coatings preserve fruits and
vegetables. Hydrocolloids (polysaccharides or proteins), hydrophobic substances
(lipids or waxes), or a combination of the two (composite coatings) make up
edible coatings, which may improve the coating's handling characteristics
(Espino-Diaz et al., 2010). Edible
coatings have been used to preserve fruit and vegetables during storage,
according to several studies. To preserve the quality of citrus and apples, and
to a lesser extent, mangos, papayas, pomegranates, cherries, avocados,
cantaloupes, and tomatoes, among others, a variety of edible coatings are
available today (Olivas et al., 2008).
According to Meng et al. (2008),
waxing can be a useful technique for reducing chilling injury because it
creates semi-permeable barriers. Additionally, Chien et al. (2007) reported that waxing could enhance the water content,
titratable acidity, ascorbic acidity, and firmness of tomato fruit.
1.13 Types of food grade wax
Since they
have been applied to a wide range of fruits and vegetables for more than 60
years, waxes are not a recent development. Various natural and synthetic
ingredients are combined to create waxes, which come from different sources
(Chien et al., 2007). Food-grade
waxes are formulated using a variety of raw materials as a base. The materials
that are most frequently used are polyethylene, paraffin, carnauba, and shellac.
Beeswax and candelilla wax are less commonly used wax bases (Ball, 1997;
Postharvest Handling Technical Bulletin, 2004). Mineral oil, paraffin, and
polyethylene are examples of petroleum-based waxes (Ball, 1997). According to
Olivas et al. (2008), these waxes are
frequently used on melons, stone fruits, tropical fruits, and a range of
vegetables.
2.3.2
Importance of waxing
(a) Improved appearance: The post-harvest handling technical bulletin from 2003 states
that waxing tomato fruit will improve its appearance and give it a glossier
appearance. Wax-coated fruits and vegetables are typically more brilliant and
shinier. They also stay colorful and youthful-looking for an extended amount of
time. From a marketing perspective, this is advantageous because consumers tend
to base their acceptance of a product on its outward look. Certain commodities'
internal color can also be improved by waxing (Postharvest Handling Technical
Bulletin, 2004).
(b) Less moisture loss: According to a 2004 postharvest handling technical
bulletin, every fruit and vegetable, including tomatoes, has a natural cuticle
covering that acts as a barrier against moisture loss. Nonetheless, water vapor
can pass through the cuticle's microcracks, pores, and cuticle. A thin layer of
the coating material that adheres tightly is applied to the fruit's surface
during the waxing process. The cuticle's pores are blocked by the wax coating,
which drastically lowers the quantity of water vapor loss.
The product's grade is usually lowered or rendered
entirely unmarketable due to the extent of moisture loss. The importance of
waxing in preventing weight loss increases with the length of time produced is
expected to be stored. A thin layer of wax coating applied can stop product
weight loss from occurring by thirty to forty percent. Waxing reduces or
eliminates pithiness and undesirable textural changes caused by moisture loss.
To minimize these unintended moisture losses, waxing offers a barrier (Postharvest
Handling Technical Bulletin, 2004).
(c) Less economic loss: All fresh fruits and vegetables are primarily made up of
water, which typically makes up 80–90% of the product's fresh weight. After
harvest, the product starts to lose moisture due to transpiration (water
evaporation) and respiration, which causes weight loss. This is not good from
an economic perspective because growers often sell their fresh products based
on weight, and they will receive less money if the product loses weight (Postharvest
Handling Technical Bulletin, 2004).
(d) Reduced postharvest: Disintegration is a barrier created by waxing to prevent
bacteria, fungi, and other pathogens from entering the product. For postharvest
pathogens to proliferate, the skin of the product usually needs a layer of free
moisture. A hydrophobic (non-water compatible) surface is produced by waxing,
and this prevents the growth and development of pathogens. To give the wax even
more defense against deterioration, a fungicide can be added (Postharvest
Handling Technical Bulletin, 2004).
(e) Longer postharvest: Tomatoes and other fruits and vegetables are living
things that keep breathing even after they are harvested, according to reports
published in the Postharvest Handling Technical Bulletin (2004). Waxing
modifies the product's internal atmosphere, increasing the amount of carbon
dioxide and decreasing the amount of oxygen. As a result, the product's
postharvest life increases and its respiration rate decreases. An extended
postharvest life enables farmers and traders to extend their marketing window.
(f) Less susceptibility to chilling injury: Tropical fruits
and vegetables, like tomatoes, are prone to a physiological injury known as
chilling injury (CI), which happens at low temperatures. The temperature and
length of exposure to the low temperature determine how much CI is present.
Depending on the crop, it happens between 13°C (56°F) and 0°C (32°F). Waxing
lessens the severity of CI and permits the damage-free storage of CI-sensitive
goods at slightly lower temperatures. Waxing, however, does not completely
remove CI on commodities that are susceptible (Postharvest Handling Technical
Bulletin, 2004).
2.3.3 Wax Application Methods
The tomato
fruit is normally waxed by manual rubbing and dipping/submergence.
i.
Manual rubbing: By hand, we rubbed the
tomato fruit and spread liquid waxes like Shea butter, coconut oil, and palm
kernel oil evenly over its surface. The wax will be applied to the fruits using
a fine-bristled brush or a soft, absorbent cloth. The tomato fruit will be
applied and then allowed to air dry for approximately fifteen minutes before
being packed (Olivas et al., 2008).
ii. Dipping/submergence: Usually, the tomato fruit is immersed or briefly dipped
in a bath of melted wax, such as Shea butter, paraffin, and beeswax. Almost
immediately after being removed from the melted solution, the wax solidifies.
After dipping, the cucumber fruit is ready to be packed in just one minute
(Olivas et al., 2008).
The research
was undertaken principally to study the effect of waxing on the shelf life of
cucumber fruits with waxed from Moringa and Shea butter oils and their
different formulated composites. The objective of this was to examine the
effect of waxing on the storability of cucumber, thereby reducing ripening
contributing to the existing literature on the effect of waxing on cucumber
using new improved methods. Waxing was done by dipping method in the first day
using sing and composite moringa seed oil and shea butter oil and their
physicochemical parameters were studied for a period of two weeks at interval
of two days.
2.0 MATERIAL AND METHODS
2.1 Materials
The cucumber
fruit (Cucumis sativus L), Moringa seed, and Shea butter were
purchased from Shasha market, along Owo/Akure expressed road, Akure, Ondo
State, and were taken to the Food processing laboratory of the Department of
Food Science and Technology, Rufus Giwa Polytechnic for processing and sample
preparation. All chemicals used for analysis were of analytical grade and
obtained from Delson Pascal Scientific Laboratory chemicals and Equipment in
Alagbaka, Akure, Ondo State, Nigeria.
2.2 Preparation of samples
Firm
cucumbers (Fig. 2), were sorted and washed to remove dirt and sand. Moringa oleifera seeds were de-husked
and roasted. Production of the Moringa seed oil was achieved by mechanical
pressing method (hydraulic press) in the processing laboratory (Figures 3 and
4). The oil was kept in an airtight bottle until when needed to avoid
adulteration while the shea butter was bought directly from local producers
(Fig. 3). These were waxed with different compositions of Moringa oil and Shea
butter (Fig. 4) and the waxing cucumber were placed in baskets and stored for
fifteen days. The samples were picked and blended into slurry each day (Fig. 5)
for analysis after the hardness had been tested.

Fig. 2: Cucumber image

Fig 3: Single oil (Moringa and shea butter
oil)

Fig 4: Composite oil

Fig.5: Blended cucumber fruit.
2.3 Physical properties
Texture, color development, total soluble solids, brix,
and hardness as described by the Association of Official Analytical Chemists (AOAC,2005).
2.4
Chemical composition
The chemical composition
observed were moisture content, ash, titrable acidity and pH as described by
the standard method of the Association of Official Analytical Chemists (AOAC,
2005).
4.0 RESULT
AND DISCUSSION
4.1 Result
Table 1: Physicochemical properties of waxed cucumber
stored for day one and day Three
|
Days
|
Blends
|
Colour
|
Hardness Kg/cm3
|
Ash Content g/100g
|
Moisture Content g/100g
|
Total Solid g/100g
|
Brix
|
PH
|
Titrable A. g/dm3
|
|
Day 1
|
FRC
|
Deep- Green
|
5.300
|
1.700
|
96.400
|
3.600
|
2.500
|
5.450
|
32.600
|
|
Day 3
|
FRC
|
Deep -Green
|
5.300
|
1.500
|
96.120
|
3.180
|
3.000
|
5.600
|
22.100
|
|
|
100%SOC
|
Deep -Green
|
5.300
|
1.150
|
96.080
|
3.220
|
3000
|
5.500
|
34.100
|
|
|
100%MOC
|
Deep -Green
|
5.300
|
1.730
|
96.620
|
3.380
|
2.500
|
6.200
|
30.300
|
|
|
80:20MSC
|
Deep- Green
|
5.300
|
1.230
|
96.000
|
3.180
|
3.000
|
5.400
|
34.500
|
|
|
20:80MSC
|
Deep -Green
|
5.300
|
1.750
|
96.140
|
3.860
|
3.200
|
6.000
|
28.800
|
Keys
1.
FRC: - Fresh cucumber
2.
100% SOC - 100% Shea butter oil for cucumber
3.
100% MOC - 100% Moringa Oil for cucumber
4.
80:20 MSC - 80 % Shea butter oil & 20%
Moringa oil for cucumber
5.
20:80 MSC - 20 % Shea butter oil & 80%
Moringa oil for cucumber
The results
obtained in Table1 for day 1 and 3 showed no significant change in the
physicochemical parameters evaluated for Cucumis
sativus L, however, there is a
significant change in the total titrable acidity, PH and ash
content. The titrable acidity with higher value in 80:20MSC (34.50 g/dm3
and 100%SOC (34.10 g/dm3) and PH (5.4 and 5.5)
respectively. Higher acidity and lower PH indicated the fruit still
retain its freshness for longer post-harvest life according to Jimenez et al., (2002), because ripening
increases with reduced acidity and increased PH of a fruit as explained by Sogo-Temi,
et al., (2014).
Table 2: Physicochemical properties of waxed
cucumber stored for day Five
|
Blends
|
Colour
|
Hardness Kg/cm3
|
Ash Content g/100g
|
Moisture Content
g/100g
|
Total Solid g/100g
|
Brix
|
PH
|
Titrable A. g/dm3
|
|
FRC
|
Green
|
5.300
|
2.300
|
96.540
|
3.860
|
3.500
|
6.700
|
20.800
|
|
100%SOC
|
Deep-Green
|
5.300
|
1.720
|
96.100
|
3.600
|
3.000
|
6.100
|
29.200
|
|
100%MOC
|
Deep- Green
|
5.300
|
2.000
|
96.520
|
4.080
|
3.000
|
6.500
|
27.700
|
|
80:20MSC
|
Deep-Green
|
5.300
|
1.500
|
96.080
|
3.320
|
3.000
|
5.500
|
30.500
|
|
20:80MSC
|
Deep-Green
|
5.300
|
2.300
|
96.340
|
4.160
|
3.200
|
6.500
|
26.100
|
The results
obtained in Table 2 for day showed the values obtained for day five for the
shelve life investigation of Cucumis
sativus L. The colour observed in
FRC was green while others were deep-green, the same hardness was still
observed for all the parameters while the ash content increased significantly
in FRC and 100%MOC (2.30 and 2.000).
According to Sogo-Temi, et al,.
(2014) and Singh et al., (2018)
increase moisture content showed increase in ripening rate and these are
observed in FRC and 100%MOC (96.540 and 96.340g/100g) respectively. A
significant reduction in titrable acidty were noticed at the same blends
(20.800 and 26.100g/dm3) and increase in PH (6.7 and 6.5).
Table 3: Physicochemical properties of waxed cucumber
stored for day Seven
|
Blends
|
Colour
|
Hardness Kg/cm3
|
Ash Content g/100g
|
Moisture Content
g/100g
|
Total Solid g/100g
|
Brix
|
PH
|
Titrable A. g/dm3
|
|
FRC
|
Yellowish-Green
|
4.500
|
2.850
|
96.580
|
4.120
|
5.200
|
7.100
|
18.700
|
|
100%SOC
|
Deep-Green
|
5.300
|
1.900
|
96.100
|
3.900
|
4.300
|
6.500
|
27.200
|
|
100%MOC
|
Green
|
5.000
|
2.130
|
96.520
|
4.740
|
5.400
|
6.900
|
20.500
|
|
80:20MSC
|
Deep-Green
|
5.300
|
1.750
|
96.090
|
3.380
|
4.000
|
6.100
|
27.300
|
|
20:80MSC
|
Green
|
5.000
|
2.380
|
96.540
|
4.360
|
5.100
|
6.700
|
21.500
|
The results obtained in Table 3 for day seven
showed the values obtained for day seven for the shelve life investigation of Cucumis sativus L. Yellowish-Green colour was observed in FRC wile green colour
were observed in 100%MOC and 20:80MSC, deep-green were retained in 80:20MSC and
100%SOC respectively. Ash content and brix increased significantly in FRS as a
result increase in mineral and sugar composition due to ripening. There was a
drastic reduction in titrable acidity in FRC, increase in PH than
the rest, indicating metabolic and physiological changes that irreversibly
alter its characteristics for ripening according to Valeria et al., (2014).
Table 4: Physicochemical properties of waxed cucumber
stored for day Nine
|
Blends
|
Colour
|
Hardness Kg/cm3
|
Ash Content g/100g
|
Moisture Content
g/100g
|
Total Solid g/100g
|
Brix
|
PH
|
Titrable A. g/dm3
|
|
FRC
|
Yellowish-Green
|
3.000
|
2.710
|
96.591
|
4.709
|
7.900
|
8.100
|
15.200
|
|
100%SOC
|
Green
|
5.100
|
1.980
|
96.409
|
5.591
|
4.800
|
6.700
|
24.400
|
|
100%MOC
|
Yellowish-Green
|
4.300
|
2.550
|
96.734
|
3.866
|
6.800
|
7.000
|
16.100
|
|
80:20MSC
|
Deep-Green
|
5.300
|
1.750
|
96.100
|
3.538
|
4.700
|
6.200
|
25.500
|
|
20:80MSC
|
Yellowish-Green
|
4.000
|
2.250
|
96.718
|
4.582
|
7.100
|
6.900
|
15.700
|
Table 4
showed the results obtained in day nine for the shelve life investigation of Cucumis sativus L. The colour showed that only 80:20MSC was Deep-Green, 100%SOC was
green while the rest were Yellowish-Green. The firmness (hardness) was equally
higher in 80:20MSC (5.30) than the rest. Brix, which represented the sugar
content were higher in FRC (7.700) and 20:80MSC (7.100), likewise the PH
(8.100 and 6.900) respectively. The
observed ash content in FRC and 20:80MSC were 2.710 and 2.250g/100g, lower than
the values obtained in day four, but higher in FRC than value recorded for
ripped plantain and the same value with20:80MSC (2.25) for
five days as reported by Baiyeri et
al., (2011).
Table 5: Physicochemical properties of waxed cucumber
stored for day Eleven
|
Blends
|
Colour
|
Hardness Kg/cm3
|
Ash Content g/100g
|
Moisture Content
g/100g
|
Total Solid g/100g
|
Brix
|
PH
|
Titrable A. g/dm3
|
|
FRC
|
Pale-Yellow
|
3.000
|
2.250
|
97.140
|
5.260
|
7.160
|
7.500
|
13.500
|
|
100%SOC
|
Yellowish- Green
|
4.500
|
2.430
|
96.559
|
5.980
|
5.580
|
6.900
|
21.400
|
|
100%MOC
|
Yellow
|
3.500
|
2.100
|
96.850
|
4.106
|
6.850
|
7.500
|
15.000
|
|
80:20MSC
|
Green
|
5.000
|
1.920
|
96.200
|
4.638
|
5.100
|
6.500
|
22.100
|
|
20:80MSC
|
Pale-Yellow
|
3.500
|
2.050
|
96.960
|
4.982
|
7.840
|
6.700
|
14.200
|
Table 5 showed the results obtained in day
eleven for the shelve life investigation of
Cucumis sativus L. Pale yellow
were observed in FRC and 20:80MSC, indicating a drastic denatured by oxidation
of organic component in the peal. A
significant decrease in the total titrable acidity were noticed in FRC, 100%MOC
and 20:80MSC (13.500, 15.000 and 14.200g/dm3), and PH (7.500, 7.500
and 6.700), revealing reduction in fruit quality by fermentation process and decay,
and their resistance to pathogen attack are progressively lost as explained by
Valeria et al., (2014). It is worth
to note that 80:20MSC and 100%SOC still retained their qualities as their titrable
acidity and PH increased gradually.
Table 6: Physicochemical properties of waxed cucumber
stored for day Thirteen
|
Blends
|
Colour
|
Hardness Kg/cm3
|
Ash Content g/100g
|
Moisture Content
g/100g
|
Total Solid g/100g
|
Brix
|
PH
|
Titrable A. g/dm3
|
|
FRC
|
Rotten Yellow
|
3.000
|
1.850
|
97.700
|
5.860
|
3.860
|
7.000
|
15.000
|
|
100%SOC
|
Yellow
|
3.500
|
2.230
|
96.759
|
6.180
|
4.280
|
7.800
|
17.400
|
|
100%MOC
|
Pale-Yellow
|
3.000
|
1.800
|
96.860
|
4.866
|
3.450
|
7.000
|
15.000
|
|
80:20MSC
|
Green
|
4.000
|
2.520
|
96.420
|
4.938
|
4.000
|
7.200
|
19.100
|
|
20:80MSC
|
Rotten-yellow
|
3.000
|
1.750
|
97.282
|
5.182
|
3.500
|
6.700
|
14.200
|
Table 6 showed the results obtained in day
thirteen for the shelve life investigation of Cucumis sativus L. Rotten-yellow
were observed in FRC and 20:80MSC and
3.00 for their hardness respectively. The mineral composition in terms of the
ash content reduced in FRC and 20:80MSC (1.850 and 1.750g/100g) respectively.
Titrable acidity in the two samples (FRC and 20:80MSC) were found to increase due to further fermentation by pathogen attack with progressive lost of their
quality as explained by Valeria et al.,
(2014). In sample 80:20 MSC, Climacteric fruits ripening show a sudden increase
in respiration at the onset of late ripening and increase in the brix content
(7.2) and reduction in titrable acidity (19.100g/dm3), usually in concert with
increased gaseous hormone ethylene production (Sweetman et al., 2009; Valeria et al.,
2014).
Table 7: Physicochemical properties of waxed cucumber stored
for day Fifteen
|
Blends
|
Colour
|
Hardness Kg/cm3
|
Ash Content g/100g
|
Moisture Content
g/100g
|
Total Solid g/100g
|
Brix
|
PH
|
Titrable A. g/dm3
|
|
FRC
|
Rotten-dark
|
1.500
|
0.750
|
97.900
|
5.860
|
3.260
|
6.300
|
18.000
|
|
100%SOC
|
pale-Yellow
|
3.000
|
1.830
|
96.919
|
6.580
|
4.780
|
7.900
|
15.500
|
|
100%MOC
|
Rotten-pale-Yellow
|
2.700
|
0.950
|
96.970
|
4.900
|
3.150
|
6.800
|
17.900
|
|
80:20MSC
|
Yellowish-green
|
4.000
|
2.100
|
96.520
|
4.438
|
4.500
|
7.500
|
16.500
|
|
20:80MSC
|
Rotten-dark
|
2.000
|
0.890
|
97.880
|
4.182
|
3.500
|
6.400
|
18.200
|
The results obtained in Table 7 for day showed the values
obtained for day fifteen for the shelve life investigation of Cucumis sativus L. a complete rotten has set in at FRC and 20:80MSC and increase in
the titrable acidity for complete conversion of the sugar content during
fermentation to acid and increase in increased gaseous hormone
ethylene production (Katz et al.,
2004; Sweetman et al., 2009). Higher level of sugar (brix) were observed in 100%SOC
and 80:20MSC (7.900 and 7.500). However, 80:20MSC retained its good quality at
Yellowish-green ripening with good rigidness and firmness (4.00), followed by
100%SOC (3.00).
CONCLUSION.
The observed preservative measures elucidated in this
research for cucumber post-harvest for fifteen days indicated that there is a
credible measure for preserving Cucumis
sativus L beyond fifteen days
with the application of 80 % Shea butter oil and 20% Moringa oil composition.
However, at 100% Shea butter oil, the process may not exceed the fifteen days
before deterioration sets in. Application of 80 % Shea butter oil and 20%
Moringa oil composition has proved to be the best waxing method for
post-harvest treatment of Cucumis sativus
L.
Author Contributions
G. Aladekoyi and, O. A. Ajala sought study authorization from the
appropriate government institutions. G. Aladekoyi, and O. A.
Shakpo, established the study
methodology which also comprised preparing a checklist that was used in data
collection. G. Aladekoyi and T. A Adesina analyzed the samples and
interpreted the data. O. A. Ajala and G. Aladekoyi undertook the literature
review that included the introductory background information and the
theoretical context. To guarantee accuracy and adherence to the
journal's formatting requirements, all authors revised the work appropriately.
Conflict of Interest
The authors declare that there
are no conflicts of interest regarding the publication of this manuscript. In
addition, the ethical issues; including plagiarism, informed consent, misconduct,
data fabrication and/or falsification, double publication and/or submission,
and redundancy have been completely observed by the authors
REFERENCES
AOAC. (2005). Official Methods of analysis
(15th edition). Association of official analytical chemists. Washington D.C.
Ashby, H. (2000). Protecting Perishable Foods
during Transport by Truck Transportation and Marketing Programs. USDA Handbook. 669: 1-6.
Baiyeri,
K. P.; Aba, S. C.; Otitoju, G. T.; Mbah, O. B.
(20110.The effects of ripening and
cooking method on mineral and
proximate composition of plantain (Musa sp. AAB cv. ‘Agbagba’) fruit pulp. African J. of Biotech., 10(36):
6979-6984, http://www.academicjournals.org/AJB
Ball, J.A. (1997). Evaluation of two
lipid-based edible coatings for their ability to preserve the postharvest
quality of green bell peppers. M. Sc.
Thesis, Blacksburg, Virginia, 89.
Chien, P.; Sheu.; Lin, H. (2007). Coating
citrus (Murcott tangor) fruit with low molecular weight chitosan increases
postharvest quality and shelf life. Food Chem., 100: 1160-1164.
Cohen, Y. (1981) Downy mildew of cucurbits
In the Downy Mildews (Spencer D.M., ed.), London: Academic Press, 341–354.
Cong, F.; Zhang, Y.; Dong, W. (2007). Use of
surface coatings with natamycin to improve the storability of Hami melon at
ambient temperature. Postharvest Biology and Technology 46 (1): 71-75
Cornelius, E.W.; Obeng-Ofori, D. (2008).
Post-harvest science and technology. Smart line Ltd. Accra, Ghana, 17.
Eifediyi, E.K.; Remison, S.U.
(2010). Growth and yield of cucumber (Cucumis sativus l.) as influenced by farm
yard manure and inorganic fertilizer. J. Plant Breed Crop Sci 2: 216-220.
Espino-Diaz, M., J.J; Ornelas-Paz, M.A.;
Martinez-Téllez, C. (2010). Development and characterization of edible films
based on mucilage of Opuntia ficus-indica (L.). J.Food Sci., 75: 347-352.
Food and Agriculture Organization (2008).
Basic Harvest and Post-harvest Handling Considerations for Fresh Fruits and
Vegetables. Postharvest Training CD Rom on Food Processing/FAO manual food
handling and preservation.
Golabadi, M.; Golkar, P;. Eghtedary, A.R.
(2012). Assessment of genetic variation in cucumber (CucumissativusL.)
genotypes.European Journal of Experimental Biology, 2 (5):1382-1388, 2012.
Gonzalez-Aguilar, G.; J. Ayala-Zavala, G.;
Olivas, L.; dela Rosa and E. Alvarez-Parrilla, (2010). Preserving quality of fresh-cut products
using safe technologies. J. Für
Verbraucherschutz und Lebensmittelsicherheit, 5: 65 -72.
Gruda, N.; Tanny, J. (2014). Protected crops,
pp. 327-405. In: G.R,Aldous, D.E.
(Eds.), Horticulture: Plants for People and Places, Volume 1 Dixon. Springer,
Netherlands. https://doi.org/10.1007/978-94-017--5_10.
Huang, S.; Li, R.; Zhang, Z.; Li, L.; Gu, X.
(2009).The genome of the cucumber,Cucumis sativus L. Nat Genet, 41: 1275–1281
Jimenez, A.; Creissen, G.; Kular,
B.; Firmin, J.; Robinson, S.; Verhoeyen, M.; Mullineaux, P. (2002). Changes in
oxidative processes and components of the antioxidant system during tomato
fruit ripening. Planta, 214, 751–758.
Joyce, D. C.; Shorter, A. J.; Jones, P. N.
(1995). Effect of delayed film wrapping and waxing on the shelf life of avocado
fruit. Australian Journal of Experimental Agriculture, 35 (5): 657-659.
Kader, A. A. (1994). Fruit maturity,
ripening, and quality relationships. Perishables Handling Newsletter, 80:2.
Kader, A. A. (2005). Increasing food
availability by reducing postharvest Losses of fresh produce, 2169-2175.
Katz, E., Lagunes, P.M., Riov,
J., Weiss, D., Goldschmidt, E.E., 2004. Molecular and physiological evidence
suggests the existence of a system II-like pathway of ethylene production in
non-climacteric Citrus fruit. Planta 219, 243–252.
Keith, R. E. (1999). Antioxidants and Health.
P: 1-4. Pub: Alabama Cooperative Extension System Pub ID: HE-0778.
Keith, T. (2003). Fruit and Vegetables:
Harvesting, Handling and Storage, [2nd ed. of Postharvest Technology of Fruits
and Vegetables] Oxford: Blackwell / Ames, Iowa: Iowa State, 287. ISBN 9781405106191
Kitinoja, L.; Gorny, J. (2009). Storage
Practices and Structures. Postharvest Technology for Fruit & Vegetable
Produce Marketers, 7. 1.1 –20.6
Meng, X..; Li B.; Liu, J.; Tian, S. (2008).
Physiological responses and quality attributes of table grape fruit to chitosan
pre-harvest spray and postharvest coating during storage. Food Chem., 106: 501-508.
Mrema, C. G.; Rolle, S. R. (2002). Status of
the postharvest sector and its contribution to agricultural development and
economic growth. 9th JIRCAS International
Symposium – Value Addition to Agricultural Product, 13-20.
Obeng-Ofori, D.; Danquah, Y. E.; Ofosu Anim,
J. (2007). Vegetable and spice crop production in West Africa. Smart line publishers, .53-59.
Olivas, G. I..; Davila-Avina, J. E..;
Salas-Salazar, N. A..; Molina, F. J. (2008). Use of edible coatings to preserve
the quality of fruits and vegetables during storage. Stewart Postharvest Rev., 4: 1-9.
Olivas, G.I.; Barbosa-Canovas, G.V. (2008).
Alginate-calcium films: Water vapor permeability and mechanical properties as
affected by plasticizer and relative humidity. LWT Food Sci. Technol., 41:
359-366
Postharvest Handling Technical Bulletin
(2004). Cucumber: Postharvest Care and Market Preparation, PH Bulletin No. 28.
Post-harvest Handling Technical Bulletin,
(2003), Tomato: Postharvest Care and Market Preparation, PH Bulletin No. 9.
Sabir, M. S.; Shah, S. Z. A.; Afzal, A.
(2004). Effect of chemical treatment, wax coating, oil dipping and different
wrapping materials on physio-chemical characteristics and storage behavior of
apple (Malus domestica Borkh). Pakistan
Journal of Nutrition, 3 (2): 122-127.
Saftner, R. A., (1999). The potential of fruit
coating and treatment for improving the storage and shelf life qualities of
Gala and Golden delicious apples. J. of
the American Soc. for Horticultural Sci., 124 (6): 682-689.
Singh, J.,
Bal, J. S.; Singh, S.; Mirza, A. (2018). Assessment of chemicals and growth
regulators on fruit ripening and quality : A review. Plant Archives, 18(2),
1215–1222.
Singh, J.;
Bal, J. S.; Singh, S.; Mirza, A. (2018). Assessment of chemicals and growth
regulators on fruit ripening and quality : A review. Plant Archives, 18(2),
1215–1222.
Smitha, K.; Sunil, K. M. (2016). Influence of
growing environment on growth characters of cucumber (Cucumis sativus L.).
Sogo-Temi,
C. M.; Idowu, O. A.,; Idowu, E. (2014). Effect of biological and chemical
ripening Agents on the nutritional and metal composition of banana (Musa spp). J.
Appl. Sci. Environ. Manage, 18(2), 243–246.
Somarribaa, E.; Cerdaa R, Orozcoa L,
Cifuentesa M, Dávila H (2013). Carbon stocks and cocoa yields in agroforestry
systems of Central America. Agric.
Ecosys,. and Env., 173: 46- 57.
Steven, R.T.; Vermon, R.Y.; and Micheal, C.A. (1985),
“Vitamins and minerals”, In: Fennema, O. (Ed.) Food Chemistry, 2nd Edition,
Marcel Dekker, New York, 523.
Sweetman, C.; Deluc, L.G.;
Cramer, G.R.; Ford, C.M.; Soole, K.L. (2009). Regulation of malate metabolism
in grape berry and other developing fruits. Phytochemistry,
70, 1329–1344.
Umeh, O.A.; Ojiako, F.O. (2018). Limitations
of Cucumber (Cucumis sativus L)
Production for Nutrition Security in Southeast Nigeria. International Journal of Agriculture and Rural Development
21(1): 3437-3443.
US Department of Agriculture (1985). U.S.
Standards for Grades of Greenhouse Cucumbers. (Reprinted - January 1997). 1-14.
USDA (2010). National Nutrient Database for
Standard Reference. SR23 - Reports by Single Nutrients. Dept. of Agric. Agric. Research Service. Release, 23. 1-26. US
Valeria, E. P.; Alejandra, S. M.;
Florencio E. P. (2014).Physiological aspects of fruit ripening: The
mitochondrial connection Mitochondrion.
www.elsevier.com/ locate/mito
|
Cite
this Article: Aladekoyi, G; Ajala, OA; Shakpo, OI; Adesina, TA
(2024). The Effect of Shea Butter Oil and Moringa Seeds Oil Waxing on The
Post-Harvest Life of Cucumber Fruit (Cucumis
sativus L.). Greener
Trends in Food Science and Nutrition, 4(1): 1-11, https://doi.org/10.15580/gtfsn.2024.1.062424088.
|





